Summary
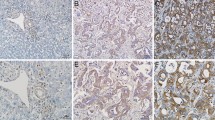
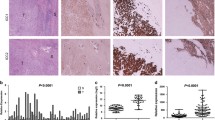
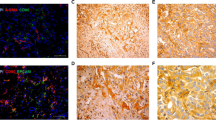
The molecular mechanisms underlying the development of intrahepatic cholangiocarcinoma (ICC) are not clear yet. In this study, we investigated the involvement of Notch1 in the development of ICC. The cDNA microarray analysis showed that Notch1 expression was higher in ICC tissues than in normal biliary epithelial cells. Stable transfection of Notch1 receptor intracellular domain (NICD1) by hydrodynamic tail vein injection induced ICC formation in mice. Western blotting confirmed that Notch1 signaling was activated in human ICC cell lines and mouse ICC tissues. Silencing Notch1 with specific short interfering RNA (siRNA) inhibited the proliferation of ICC cells. Flow cytometry and Western blotting indicated that apoptosis was induced in Notch1-silenced ICC cells compared with controls. Additionally, Notch1 silencing was associated with the inhibition of hairy and enhancer of split-1 (Hes1) and activation of the phosphatase and tensin homolog (PTEN)/p53 pathway. Taken together, these data suggest that Notch1 drives ICC formation and proliferation; downregulation of Notch1 induces apoptosis in ICC cells; Notch1 signaling may serve as a novel therapeutic target for the treatment of ICC.
Similar content being viewed by others
References
Zhao Q, Yu WL, Lu XY, et al. Combined hepatocellular and cholangiocarcinoma originating from the same clone: a pathomolecular evidence-based study. Chin J Cancer, 2016,35(1):82–90
Razumilava N, Gores GJ. Classification, diagnosis, and management of cholangiocarcinoma. Clin Gastroenterol Hepatol, 2013,11(1):13–21
Razumilava N, Gores GJ. Cholangiocarcinoma. Lancet, 2014:2168–2179
Wang FS, Fan JG, Zhang Z, et al. The global burden of liver disease: the major impact of China. Hepatology, 2014,60(6):2099–2108
Esnaola NF, Meyer JE, Karachristos A, et al. Evaluation and management of intrahepatic and extrahepatic cholangiocarcinoma. Cancer, 2016,122(9):1349–1369
Papatheodoridis GV, Lampertico P, Manolakopoulos S, et al. Incidence of hepatocellular carcinoma in chronic hepatitis B patients receiving nucleos(t)ide therapy: a systematic review. J Hepatol, 2010,53(2):348–356
Alves RC, Alves D, Guz B, et al. Advanced hepatocellular carcinoma: Review of targeted molecular drugs. Ann Hepatol, 2011,10(1):21–27
Belina F. Hilar cholangiocarcinoma (Klatskin tumor) - current treatment options. Rozhl Chir, 2013,92:4–15
Yu Y, Wang L, Tang W, et al. RNA interference-mediated knockdown of Notch-1 inhibits migration and invasion, down-regulates matrix metalloproteinases and suppresses NF-kappaB signaling pathway in trophoblast cells. Acta Histochem, 2014,116(5):911–919
Pupo M, Pisano A, Abonante S, et al. GPER activates Notch signaling in breast cancer cells and cancer-associated fibroblasts (CAFs). Int J Biochem Cell Biol, 2014,46:56–67
Dahan S, Rabinowitz KM, Martin AP, et al. Notch-1 signaling regulates intestinal epithelial barrier function, through interaction with CD4+ T cells, in mice and humans. Gastroenterology, 2011,140:550–559
Jensen J, Pedersen EE, Galante P, et al. Control of endodermal endocrine development by Hes-1. Nat Genet, 2000,24(1):36–44
Sun YY, Li L, Liu XH, et al. The spinal notch signaling pathway plays a pivotal role in the development of neuropathic pain. Mol Brain, 2012,5:23–32
Takebe N, Nguyen D, Yang SX. Targeting notch signaling pathway in cancer: clinical development advances and challenges. Pharmacol Ther, 2014,141(2):140–149
Wolpin BM, Mayer RJ. A step forward in the treatment of advanced biliary tract cancer. N Engl J Med, 2010,362(14):1335–1337
Binato M, Schmidt MK, Volkweis BS, et al. Mouse model of diethylnitrosamine-induced gastric cancer. J Surg Res, 2008,148(2):152–157
Yang H, Li TW, Peng J, et al. A mouse model of cholestasis-associated cholangiocarcinoma and transcription factors involved in progression. Gastroenterology, 2011,141(1):378–388
Ju HL, Han KH, Lee JD, et al. Transgenic mouse models generated by hydrodynamic transfection for genetic studies of liver cancer and preclinical testing of anticancer therapy. Int J Cancer, 2016,138(1):1601–1608
Calvisi DF, Wang C, Ho C, et al. Increased lipogenesis, induced by AKT-mTORC1-RPS6 signaling, promotes development of human hepatocellular carcinoma. Gastroenterology, 2011,140(3):1071–1083
Chen X, Calvisi DF. Hydrodynamic transfection for generation of novel mouse models for liver cancer research. Am J Pathol, 2014,184(4):912–923
Wang Y, Wang Y, Zhang L, et al. Endonuclease Restriction-Mediated Real-Time Polymerase Chain Reaction: A Novel Technique for Rapid, Sensitive and Quantitative Detection of Nucleic-Acid Sequence. Front Microbiol, 2016,7:1104–1114
Yuan X, Wu H, Xu H, et al. Notch signaling: an emerging therapeutic target for cancer treatment. Cancer Lett, 2015,369(1):20–27
Kopan R, IIagan MX. The canonical Notch signaling pathway: unfolding the activation mechanism. Cell, 2009,137(2):216–233
Fan B, Malato Y, Calvisi DF, et al. Cholangiocarcinomas can originate from hepatocytes in mice. J Clin Invest, 2012,122(8):2911–2915
O’Neil J, Calvo J, McKenna K, et al. Activating Notch1 mutations in mouse models of T-ALL. Blood, 2006,107(2):781–785
Yan XQ, Sarmiento U, Sun Y, et al. A novel Notch ligand, Dll4, induces T-cell leukemia/lymphoma when overexpressed in mice by retroviral-mediated gene transfer. Blood, 2001,98(13):3793–3799
Allen TD, Rodriguez EM, Jones KD, et al. Activated Notch1 induces lung adenomas in mice and cooperates with Myc in the generation of lung adenocarcinoma. Cancer Res, 2011,71(18):6010–6018
Wang Z, Da Silva TG, Jin K, et al. Notch signaling drives stemness and tumorigenicity of esophageal adenocarcinoma. Cancer Res, 2014,74(21):6364–6374
Gallahan D, Callahan R. The mouse mammary tumor associated gene INT3 is a unique member of the NOTCH gene family (NOTCH4). Oncogene, 1997,14(16):1883–1890
Dill MT, Tornillo L, Fritzius T, et al. Constitutive Notch2 signaling induces hepatic tumors in mice. Hepatology, 2013,57(4):1607–1619
Talora C, Sgroi DC, Crum CP, et al. Specific down-modulation of Notch1 signaling in cervical cancer cells is required for sustained HPV-E6/E7 expression and late steps of malignant transformation. Genes Dev, 2002,16(17):2252–2263
Zweidler-McKay PA, He Y, Xu L, et al. Notch signaling is a potent inducer of growth arrest and apoptosis in a wide range of B-cell malignancies. Blood, 2005,106(12):3898–3906
Aster JC, Pear WS, Blacklow SC. The Varied Roles of Notch in Cancer. Annu Rev Pathol, 2017,12:245–275
Bolos V, Grego-Bessa J, de la Pompa JL. Notch signaling in development and cancer. Endocr Rev, 2008,28(3):339–363
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of Interest Statement
The authors declare that they have no conflict of interest.
This work was supported by the National Natural Science Foundation of China (No. 81801621, No. 81572723, No. 81872253).
Rights and permissions
About this article
Cite this article
Guo, J., Fu, W., Xiang, M. et al. Notch1 Drives the Formation and Proliferation of Intrahepatic Cholangiocarcinoma. CURR MED SCI 39, 929–937 (2019). https://doi.org/10.1007/s11596-019-2125-0
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-019-2125-0