Summary
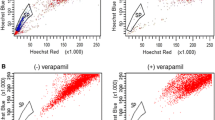
Mounting evidence has shown that side population (SP) cells are enriched for cancer stem cells (CSCs) responsible for cancer malignancy. In this study, SP technology was used to isolate a small subpopulation of SP cells in human gallbladder cancer cell line GBC-SD, and SP cells which had superior potential for proliferation in vitro and tumorigenesis in vivo were identified. Importantly, the abundance of GBC-SD SP cells was increased by a transforming growth factor-β (TGF-β)-induced epithelial-mesenchymal transition (EMT), and this effect was accompanied with a strong up-regulation of ABCG2 mRNA expression, and a decreased sensitivity to mitoxantrone. SP cells were restored upon the removal of TGF-β and the reversion of the cells to an epithelial phenotype, and smad3-specific siRNA reduced SP abundance in response to TGF-β. In conclusion, TGF-β-induced EMT by smad-dependent signaling pathway promotes cancer development and anti-cancer drug resistant phenotype by augmenting the abundance of GBC-SD SP cells, and a better understanding of mechanisms involved in TGF-β-induced EMT may provide a novel strategy for preventing cancer progression.
Similar content being viewed by others
References
Chan KM, Yeh TS, Yu MC, et al. Gallbladder carcinoma with biliary invasion: clinical analysis of the differences from nonbiliary invasion. World J Surg, 2005,29(1):72–75
Kayahara M, Nagakawa T. Recent trends of gallbladder cancer in Japan: an analysis of 4,770 patients. Cancer, 2007,110(3):572–580
Yee K, Sheppard BC, Domreis J, et al. Cancers of the gallbladder and biliary ducts. Oncology (Williston Park), 2002,16(7):939–946, 949; discussion 949–950, 952–953, 956–957
Li C, Heidt DG, Dalerba P, et al. Identification of pancreatic cancer stem cells. Cancer Res, 2007,67(3):1030–1037
O’Brien CA, Pollett A, Gallinger S, et al. A human colon cancer cell capable of initiating tumour growth in immunodeficient mice. Nature, 2007,445(7123):106–110
Reya T, Morrison SJ, Clarke MF, et al. Stem cells, cancer, and cancer stem cells. Nature, 2001,414(6859):105–111
Christiansen JJ, Rajasekaran AK. Reassessing epithelial to mesenchymal transition as a prerequisite for carcinoma invasion and metastasis. Cancer Res, 2006,66(17):8319–8326
Tsuji T, Ibaragi S, Hu GF. Epithelial-mesenchymal transition and cell cooperativity in metastasis. Cancer Res, 2009,69(18):7135–7139
Kudo-Saito C, Shirako H, Takeuchi T, et al. Cancer metastasis is accelerated through immunosuppression during Snail-induced EMT of cancer cells. Cancer Cell, 2009,15(3):195–206
Komuro A, Yashiro M, Iwata C, et al. Diffuse-type gastric carcinoma: progression, angiogenesis, and transforming growth factor beta signaling. J Natl Cancer Inst, 2009,101(8):592–604
Kiyono K, Suzuki HI, Morishita Y, et al. c-Ski overexpression promotes tumor growth and angiogenesis through inhibition of transforming growth factor-beta signaling in diffuse-type gastric carcinoma. Cancer Sci, 2009,100(10):1809–1816
Derynck R, Zhang YE. Smad-dependent and Smad-independent pathways in TGF-beta family signalling. Nature, 2003,425(6958):577–584
Arumugam T, Ramachandran V, Fournier KF, et al. Epithelial to mesenchymal transition contributes to drug resistance in pancreatic cancer. Cancer Res, 2009,69(14): 5820–5828
Moustakas A, Heldin CH. Signaling networks guiding epithelial-mesenchymal transitions during embryogenesis and cancer progression. Cancer Sci, 2007,98(10):1512–1520
Zavadil J, Bottinger EP. TGF-beta and epithelial-to-mesenchymal transitions. Oncogene, 2005,24(37):5764–5774
Savagner P. Leaving the neighborhood: molecular mechanisms involved during epithelial-mesenchymal transition. Bioessays, 2001,23(10):912–923
Goodell MA, Brose K, Paradis G, et al. Isolation and functional properties of murine hematopoietic stem cells that are replicating in vivo. J Exp Med, 1996,183(4): 1797–1806
Yeh YC, Wang CZ, Tang MJ. Discoidin domain receptor 1 activation suppresses alpha2beta1 integrin-dependent cell spreading through inhibition of Cdc42 activity. J Cell Physiol, 2009,218(1):146–156
Wulf GG, Luo KL, Jackson KA, et al. Cells of the hepatic side population contribute to liver regeneration and can be replenished with bone marrow stem cells. Haematologica, 2003,88(4):368–378
Goodell MA. Multipotential stem cells and “side population” cells. Cytotherapy, 2002,4(6):507–508
Thiery JP. Epithelial-mesenchymal transitions in development and pathologies. Curr Opin Cell Biol, 2003,15(6): 740–746
Thiery JP. Epithelial-mesenchymal transitions in tumour progression. Nat Rev Cancer, 2002,2(6):442–454
Ho MM, Ng AV, Lam S, et al. Side population in human lung cancer cell lines and tumors is enriched with stem-like cancer cells. Cancer Res, 2007,67(10):4827–4833
Miettinen PJ, Ebner R, Lopez AR, et al. TGF-beta induced transdifferentiation of mammary epithelial cells to mesenchymal cells: involvement of type I receptors. J Cell Biol, 1994,127(6 Pt 2):2021–2036
Gal A, Sjoblom T, Fedorova L, et al. Sustained TGF beta exposure suppresses Smad and non-Smad signalling in mammary epithelial cells, leading to EMT and inhibition of growth arrest and apoptosis. Oncogene, 2008,27(9): 1218–1230
Roberts AB, Tian F, Byfield SD, et al. Smad3 is key to TGF-beta-mediated epithelial-to-mesenchymal transition, fibrosis, tumor suppression and metastasis. Cytokine Growth Factor Rev, 2006, 17(1–2):19–27
Saika S, Kono-Saika S, Tanaka T, et al. Smad3 is required for dedifferentiation of retinal pigment epithelium following retinal detachment in mice. Lab Invest, 2004,84(10):1245–1258
Saika S, Kono-Saika S, Ohnishi Y, et al. Smad3 signaling is required for epithelial-mesenchymal transition of lens epithelium after injury. Am J Pathol, 2004,164(2):651–663
Sato M, Muragaki Y, Saika S, et al. Targeted disruption of TGF-beta1/Smad3 signaling protects against renal tubulointerstitial fibrosis induced by unilateral ureteral obstruction. J Clin Invest, 2003,112(10):1486–1494
Gauldie J, Kolb M, Ask K, et al. Smad3 signaling involved in pulmonary fibrosis and emphysema. Proc Am Thorac Soc, 2006,3(8):696–702
Zhao J, Shi W, Wang YL, et al. Smad3 deficiency attenuates bleomycin-induced pulmonary fibrosis in mice. Am J Physiol Lung Cell Mol Physiol, 2002,282(3): L585–593
Fukaya R, Ohta S, Yamaguchi M, et al. Isolation of cancer stem-like cells from a side population of a human glioblastoma cell line, SK-MG-1. Cancer Lett, 2010, 291(2):150–157
Kai K, D’Costa S, Yoon BI, et al. Characterization of side population cells in human malignant mesothelioma cell lines. Lung Cancer, 2010,70(2):146–151
Haraguchi N, Utsunomiya T, Inoue H, et al. Characterization of a side population of cancer cells from human gastrointestinal system. Stem Cells, 2006,24(3):506–513
Mani SA, Guo W, Liao MJ, et al. The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell, 2008,133(4):704–715
Mishra L, Derynck R, Mishra B. Transforming growth factor-beta signaling in stem cells and cancer. Science, 2005,310(5745):68–71
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors contributed equally to this work.
This project was supported by a grant from the National Natural Science Foundation of China (No. 30772127).
Rights and permissions
About this article
Cite this article
Zhang, Z., Zhu, F., Xiao, L. et al. Side population cells in human gallbladder cancer cell line GBC-SD regulated by TGF-β-induced epithelial-mesenchymal transition. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 31, 749–755 (2011). https://doi.org/10.1007/s11596-011-0671-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-011-0671-1