Summary
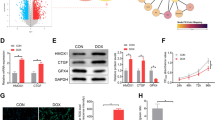
In order to investigate the influence of silencing soluble epoxide hydrolase (sEH) with double-stranded small interfering RNA (siRNA) on cardiomyocytes apoptosis induced by doxorubicin (DOX), two plasmids containing siRNA sequences specific to sEH were constructed and transfected into the primary cultured cardiomyocytes by using FuGENE HD transfection agents. The mRNA and protein expression levels of sEH were detected by semiquantitative RT-PCR and Western blotting respectively, and the plasmids that silenced sEH most significantly were selected, and renamed EH-R. The plasmids carrying a nonspecific siRNA coding sequence (PCN) served as the negative control. Cardiomyocytes were divided into four groups: control group, DOX group, PCN+DOX group, and EH-R+DOX group. Apoptosis of cardiomyocytes was induced by DOX at a concentration of 1 μmol/L. Apoptosis rate of cardiomyocytes was determined by flow cytometery. The protein expression levels of Bcl-2 and Bax were detected by Western blotting. The results showed that the expression of sEH was down-regulated by EH-R plasmid. The expression levels of sEH mRNA and protein in the EH-R+DOX group were significantly decreased as compared with other groups (P<0.01). As compared with the control group, the apoptosis rate of cardiomyocytes in three DOX-treated groups was obviously increased, the expression levels of Bax increased, and those of Bcl-2 decreased (P<0.01). However, the expression levels of Bax were decreased, those of Bcl-2 increased and the apoptosis rate of cardiomyocytes obviously decreased in EH-R+DOX group when compared with those in the DOX group and the PCN+DOX group (P<0.01 for each). It was concluded that the recombinant plasmids could be successfully constructed, and transfected into the primary cultured cardiomyocytes. They could ameliorate the DOX-induced cardiomyocytes apoptosis by selectively inhibiting the expression of sEH with RNAi and increasing the expression of Bcl-2.
Similar content being viewed by others
References
Chatterjee K, Zhang JQ, Honbo N, et al. Doxorubicin cardiomyopathy. Cardiology, 2010,115(2):155–162
Singal PK, Iliskovic N. Doxorubicin-induced cardiomyopathy. New Engl J Med, 1998,339(13):900–905
Takemura G, Fujiwara H. Doxorubicin-induced cardiomyopathy from the cardiotoxic mechanisms to management. Prog Cardiovasc Dis, 2007,49(5):330–352
Carvalho C, Santos RX, Cardoso S, et al. Doxorubicin: The good, the bad and the ugly effect. Curr Med Chem, 2009,16(25):3267–3285
Imig JD, Hammock BD. Soluble epoxide hydrolase as a therapeutic target for cardiovascular diseases. Nat Rev Drug Discov, 2009,8(10):794–805
Rossi JJ. Expression strategies for short hairpin RNA interference triggers. Hum Gene Ther, 2008,19(4):313–317
Hannon GJ. RNA interference. Nature, 2002,418(6894): 244–251
Arola OJ, Saraste A, Pulkki K, et al. Acute doxorubicin cardiotoxicity involves cardiomyocyte apoptosis. Cancer Res, 2000,60(7):1789–1792
Gilleron M, Marechal X, Montaigne D, et al. NADPH oxidases participate to doxorubicin-induced cardiac myocyte apoptosis. Biochem Bioph Res Commun, 2009,388(4):727–731
Chua CC, Liu XW, Gao JP, et al. Multiple actions of pifithrin-alpha on doxorubicin-induced apoptosis in rat myoblastic H9c2 cells. Am J Physiol Heart Circ Physiol, 2006,290(6):H2606–H2613
Seubert JM, Sinal CJ, Graves J, et al. Role of soluble epoxide hydrolase in postischemic recovery of heart contractile function. Circ Res, 2006,99(4):442–450
Xu D, Li N, He Y, et al. Prevention and reversal of cardiac hypertrophy by soluble epoxide hydrolase inhibitors. Proc Natl Acad Sci USA, 2006,103(49):18733–18738
Monti J, Fischer J, Paskas S, et al. Soluble epoxide hydrolase is a susceptibility factor for heart failure in a rat model of human disease. Nat Genet, 2008,40(5): 529–537
Nithipatikom K, Gross GJ. Epoxyeicosatrienoic acids: novel mediators of cardioprotection. J Cardiovasc Pharmacol Ther, 2010,15(2):112–119
Zordoky BNM, Anwar-Mohamed A, Aboutabl ME, et al. Acute doxorubicin cardiotoxicity alters cardiac cytochrome P450 expression and arachidonic acid metabolism in rats. Toxicol Appl Pharm, 2010,242(1):38–46
Huang M, Chan DA, Jia FJ, et al. Short hairpin RNA interference therapy for ischemic heart disease. Circulation, 2008,118(14):S226–U289
Kurreck J. RNA interference: from basic research to therapeutic applications. Angew Chem Int Edit, 2009, 48(8):1378–1398
Xiao C, Qiu L, Zen Q. Construction of shRNA targeted to the rat angiotensin II type 1 receptors and its RNAi in cytoplasma. J Huazhong Univ Sci Technol Med Sci, 2006,26(1):4–8
Reif RD, Martinez MM, Wang K, et al. Simultaneous cell capture and induction of apoptosis using an anti-CD95 affinity microdevice. Anal Bioanal Chem, 2009,395(3): 787–795
Gustafsson AB, Gottlieb RA. Bcl-2 family members and apoptosis, taken to heart. Am J Physiol Cell Physiol, 2007,292(1):C45–C51
Dong JW, Zhu HF, Zhu WZ, et al. Intermittent hypoxia attenuates ischemia/reperfusion induced apoptosis in cardiac myocytes via regulating Bcl-2/Bax expression. Cell Res, 2003,13(5):385–391
van Empel VPM, De Windt LJ. Myocyte hypertrophy and apoptosis: a balancing act. Cardiovasc Res, 2004, 63(3):487–499
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Du, G., Lv, J., He, L. et al. Influence of silencing soluble epoxide hydrolase with RNA interference on cardiomyocytes apoptosis induced by doxorubicin. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 31, 324–328 (2011). https://doi.org/10.1007/s11596-011-0375-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-011-0375-6