Summary
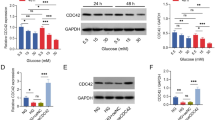
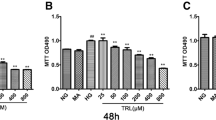
The role of protein kinase C (PKC) activation in advanced glycation end products (AGEs)-induced epithelial-mesenchymal transition in renal proximal tubular epithelial cells was investigated. HKC cells were divided into three groups: normal group, AGE-BSA group (100 mg/L AGE-BSA) and AGE-BSA+PKC inhibitor (10 μmol/L chelerythrine chloride) group. PKC activity was measured by PKC assay kit. The expression of Vimentin, and phosphorylated β-catenin was detected by using Western blotting, and the content of TGF-β1 was examined by ELISA method. The intracellular disposition of Vimentin was observed by fluorescence microscopy. As compared with normal group, PKC activity was increased significantly in AGE-BSA group. The expression of Vimentin, phosphorylated β-catenin, and TGF-β1 was enhanced significantly in AGE-BSA group. The expression of Vimentin, phosphorylated β-catenin, and TGF-β1 was significantly blocked by chelerythrine chloride. High expression of Vimentin, phosphorylated β-catenin, and TGF-β1 induced by AGE-BSA may be mediated via the activation of PKC signal transduction pathway.
Similar content being viewed by others
References
Huebschmann AG, Regensteiner JG, Vlassara H, et al. Diabetes and advanced glycoxidation end products. Diabetes Care, 2006,29(6):1420–1432
Bohlender JM, Franke S, Stein G, et al. Advanced glycation end products and the kidney. Am J Physiol Renal Physiol, 2005,289(4):F645–F659
Simonson MS. Phenotypic transitions and fibrosis in diabetic nephropathy. Kidney Int, 2007,71(9):846–854
Ng YY, Huang TP, Yang WC, et al. Tubular epithelial-myofibroblast transdifferentiation in progressive tubulointerstitial fibrosis in 5/6 nephrectomized rats. Kidney Int, 1998,54(3):864–876
Jinde K, Nikolic-Paterson DJ, Huang XR, et al. Tubular phenotypic change in progressive tubulointerstitial fibrosis in human glomerulonephritis. Am J Kidney Dis, 2001,38(4):761–769
Oldfield MD, Bach LA, Forbes JM, et al. Advanced glycation end products cause epithelial-myofibroblast trans-differentiation via the receptor for advanced glycation end products (RAGE). J Clin Invest, 2001,108(12):1853–1863
Noh H, King GL. The role of protein kinase C activation in diabetic nephropathy. Kidney Int Suppl, 2007(106): S49–S53
Li J, Gobe G. Protein kinase C activation and its role in kidney disease. Nephrology (Carlton), 2006,11(5):428–434
Lee TS, Saltsman KA, Ohashi H, et al. Activation of protein kinase C by elevation of glucose concentration: proposal for a mechanism in the development of diabetic vascular complications. Proc Natl Acad Sci U S A, 1989, 86(13):5141–5145
Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem, 1976, 7(72):248–254.
Ooie T, Takahashi N, Nawata T, et al. Ischemia-induced translocation of protein kinase C-epsilon mediates cardioprotection in the streptozotocin-induced diabetic rat. Circ J, 2003,67(11):955–961
Phillips AO, Steadman R. Diabetic nephropathy: the central role of renal proximal tubular cells in tubulointerstitial injury. Histol Histopathol, 2002,17(1):247–252
Rodriguez-Iturbe B, Johnson RJ, Herrera-Acosta J. Tubulointerstitial damage and progression of renal failure. Kidney Int Suppl, 2005(99):S82–S86
Lane A, Johnson DW, Pat B, et al. Interacting roles of myofibroblasts, apoptosis and fibrogenic growth factors in the pathogenesis of renal tubulo-interstitial fibrosis. Growth Factors, 2002,20(3):109–119
Rastaldi MP. Epithelial-mesenchymal transition and its implications for the development of renal tubulointerstitial fibrosis. J Nephrol, 2006,19(4):407–412
Li JH, Wang W, Huang XR, et al. Advanced glycation end products induce tubular epithelial-myofibroblast transition through the RAGE-ERK1/2 MAP kinase signaling pathway. Am J Pathol, 2004,164(4):1389–1397
Lilien J, Balsamo J, Arregui C, et al. Turn-off, drop-out: functional state switching of cadherins. Dev Dyn, 2002, 224(1):18–29
Lilien J, Balsamo J. The regulation of cadherin-mediated adhesion by tyrosine phosphorylation/dephosphorylation of beta-catenin. Curr Opin Cell Biol, 2005,17(5):459–565
Xu G, Arregui C, Lilien J, et al. PTP1B modulates the association of beta-catenin with N-cadherin through binding to an adjacent and partially overlapping target site. J Biol Chem, 2002,277(51):49 989–49 997
Xu G, Craig AW, Greer P, et al. Continuous association of cadherin with beta-catenin requires the non-receptor tyrosine-kinase Fer. J Cell Sci, 2004,117(Pt 15):3207–3219
Zeng R, Yao Y, Han M, et al. Biliverdin reductase mediates hypoxia-induced EMT via PI3-kinase and Akt. J Am Soc Nephrol, 2008,19(2):380–387
Pantsulaia T. Role of TGF-beta in pathogenesis of diabetic nephropathy. Georgian Med News, 2006(131):13–18
Author information
Authors and Affiliations
Additional information
Shuwang GE and Rui Zeng contributed equally to this work.
This project was supported by grants from National Natural Sciences Foundation of China (No. 30370657, No.30871172), and New Century Excellent Talents Grant (No. NCET004-0712).
Rights and permissions
About this article
Cite this article
Ge, S., Zeng, R., Luo, Y. et al. Role of protein kinase C in advanced glycation end products-induced epithelial-mesenchymal transition in renal proximal tubular epithelial cells. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 29, 281–285 (2009). https://doi.org/10.1007/s11596-009-0303-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-009-0303-1