Summary
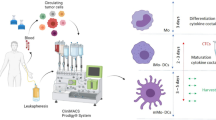
In order to develop a protocol for clinical grade generation of dendritic cells (DCs) for cancer immumotherapy, aphereses were performed with the continuous flow cell separator and materials were derived from 10 leukemia patients that had achieved complete remission. Peripheral blood monocytes were cultured in vitro with GM-CSF, IL-4 for 6 days, then TNF-(the TNF-group) or TNF-, IL-1, IL-6, PGE2 (the cytokine mixture group) were added to promote maturation. Cell number was counted by hematology analyzer, and phenotype study (CD1a, CD14, CD83) was carried out by flow cytometry, and the function of DCs was examined by mixed lymphocyte reaction. The results showed that (0.70±0.13)×107/mL (the TNF-α group) and (0.79±0.04)×107/mL (the cytokine mixture group) DCs were generated respectively in peripheral blood obtained by leucapheresis. The phenotypes were as follows: CD1a+ (74.65±4.45)%, CD83+ (39.50±4.16)%, CD14+ (2.90±1.76)% in TNF-α group, and CD1a+ (81.86±5.87)%, CD83+ (81.65±6.36)%, CD14+ (2.46±1.68)% in the cytokine mixture group. It was concluded that leucapheresis may be a feasible way to provide large number of peripheral blood monocytes for DC generation, and combined administration of TNF-, IL-1, IL-6, and PGE2 may greatly promote maturity.
Similar content being viewed by others
References
Tkachenko N, Wojas K, Tabarkiewicz J et al. Generation of dendritic cells from human peripheral blood monocytes—comparison of different culture media. Folia Histochem Cytobiol, 2005,43(1):25–30
Zheng X F, Yin Q S, Wu W et al. Early immunologic status of patients following hand allotransplantation. Chin J Clin Rehabil (Chinese), 2006,10(45):203–205
Brown R D, Pope B, Murray A. Dendritic cells from patients with myeloma are numerically normal but functionally defective as they fail to up-regulate CD80(B7-1) expression after huCD40LT stimulation because of inhibition by transforming growth factor-betal and interleukin-10. Blood, 2001,98:2992–2998
Santini S M, Di Pucchio T, Lapenta C et al. A new type I IFN-mediated pathway for the rapid differentiation of monocytes into highly active dendritic cells. Stem Cells, 2003,21:357–362
Bai L, Feuerer M, Beckhove P et al. Generation of dendritic cells from human bone marrow mononuclear cells: advantages for clinical application in comparison to peripheral blood monocyte derived cells. Int J Oncol, 2002,20:247–253
Elias M, van Zanten J, Hospers G A et al. Closed system generation of dendritic cells from a single blood volume for clinical application in immunotherapy. J Clin Apher, 2005,20(4):197–207
Labeur M S, Roters B, Pers B et al. Generation of tumor immunity by bone marrow-derived dendritic cells correlates with dendritic cell maturation stage. J Immunol, 1999,162:168–175
Obermaier B, Dauer M, Hertenl J et al. Development of a new protocol for 2-day generation of mature dendritic cells from human monocytes. Biol Proced Online, 2003,5(1):197–203
Author information
Authors and Affiliations
Additional information
TANG Duozhuang, male, born in 1978, Postgraduate
This work was supported by a grant from the National Natural Science Foundation of China (No. 30400186).
Rights and permissions
About this article
Cite this article
Tang, D., Tao, S., Cao, Y. et al. Clinical grade of gerneration of dendritic cells for immunotherapy. J. Huazhong Univ. Sc. Technol. 27, 265–268 (2007). https://doi.org/10.1007/s11596-007-0312-x
Received:
Issue Date:
DOI: https://doi.org/10.1007/s11596-007-0312-x



