Abstract
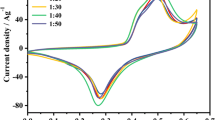
Single-handed helical carbonaceous materials attracted much attention for varieties of potential applications. Herein, single-handed helical 1, 4-phenylene bridged polybissilsesquioxane nanofibers were prepared through a supramolecular templating approach using a pair of enantiomers. After carbonization at 700 °C for 2.0 h and removal of silica using HF aqueous solution, single-handed helical carbonaceous nanofibers were obtained. The obtained samples were characterized using the field-emission scanning electron microscopy, transmission electron microscopy, N2 sorptions, X-ray diffraction, Raman spectroscopy and diffuse reflectance circular dichroism (DRCD). The Raman spectrum indicated that the carbon was amorphous. The DRCD spectra indicated that the carbonaceous nanofibers exhibited optical activity. The surface area of the left-handed helical carbonaceous nanofibers was 907 m2/g. Such material has potential applications as chirality sensor and supercapacitor electrode.
Similar content being viewed by others
References
Qin Y, Yu L, Wang Y, et al. Amorphous Helical Carbon Nanofibers Synthesized at Low Temperature and Their Elasticity and Processablity[J]. Solid State Commun., 2006, 138(1): 5–8
Chen X Q, Yang S M, Motojima S, et al. Morphology and Microstructure of Twisting Nano-Ribbons Prepared Using Sputter-Coated Fe-Base Alloy Catalysts on Glass Substrates[J]. Mater. Lett., 2005, 59(7): 854–858
Allen C S, Zhang C, G Burnell, et al. A Review of Methods for the Accurate Determination of the Chiral Indices of Carbon Nanotubes from Electron Diffraction Patterns[J]. Carbon, 2011, 49: 4961–4971
T Yu, Y Gong, T Lu, et al. Recognition of Carbon Nanotube Chirality by Phage Display[J]. RSC Adv., 2012, 2: 1466–1476
Neihsial S, Periyasamy G, Samanta P K, et al. Understanding the Binding Mechanism of Various Chiral SWCNTs and ssDNA: a Computational Study[J]. J. Phys. Chem. B, 2012, 116(51): 14754–14759
Chung W, Nobusawa K, Kamikubo H, et al. Improving the Second-Order Nonlinear Optical Response of Fluorescent Proteins: The Symmetry Argument[J]. J. Am. Chem. Soc., 2013, 135(10): 4061–4069
Li X L, Tu X M, Zaric S, et al. Selective Synthesis Combined with Chemical Separation of Single-Walled Carbon Nanotubes for Chirality Selection[J]. J. Am. Chem. Soc., 2007, 129(51): 15770–15771
Yang W, Sun W J, Chu W, et al. Synthesis of Carbon Nanotubes Using Scrap Tyre Rubber as Carbon Source[J]. Chin. Chem. Lett., 2012, 23(3): 363–366
Chen Y, Wei L, Wang B, et al. Low-Defect, Purified, Narrowly (n,m)-dispersed Single-walled Carbon Nanotubes Grown from Cobalt-Incorporated MCM-41[J]. ACS Nano, 2007, 1(4): 327–336
Zhang X L, Chu X L, Wang L, et al. Rational Design of a Tetrameric Protein to Enhance Interactions between Self-Assembled Fibers Gives Molecular Hydrogels[J] Angew. Chem. Int. Ed., 2012, 51(18): 4388–4392
Xu Q H, Moreau J J E, Wong Chi Man M J. Influence of Alkylene Chain Length on the Morphology of Chiral Bridged Silsesquioxanes[J]. Sol-Gel Sci. & Technol., 2004, 32(1): 111–115
Shimizu T, Masuda M, Minamikawa H. Supramolecular Nanotube Architectures Based on Amphiphilic Molecules[J]. Cheminform, 2005, 36(32): 1401–1443
Li B Z, Chen Y L, Zhao H, et al. From Branched Self-Assemblies to Branched Mesoporous Silica Nanoribbons[J]. Chem. Commun., 2008, 6366–6368
Wu X J, Ji S J, Li Y, et al. Helical Transfer through Nonlocal Interactions[J]. J. Am. Chem. Soc., 2009, 131(16): 5986–5993
Fan C X, Qiu H B, Ruan J F, et al. Formation of Chiral Mesopores in Conducting Polymers by Chiral-Lipid-Ribbon Templating and “Seeding” Route[J]. Adv. Funct. Mater., 2008, 18(18): 2699–2707
C Y Zhang, S B Wang, H J Huo, et al. Preparation of Helical Mesoporous Tantalum Oxide Nanotubes through a Sol-Gel Transcription Approach[J]. Chem. Asian J., 2013, 8: 709–712
Li H T, Li B Z, Chen Y L, et al. Preparetion of Chiral 4,4-Biphenylene-Silica Nanoribbons[J]. Chin. J. Chem. 2009, 27(10): 1860–1862
Liu S H, Duan Y Y, Feng X J, et al. Synthesis of Enantiopure Carbonaceous Nanotubes with Optical Activity[J]. Angew. Chem. Int. Ed., 2013, 52(27):6858–6862
Zhang C Y, Li Y, Li B Z, et al. Preparation of Single-handed Helical Carbon/silica and Carbonaceous Nanotubes by Using 4, 4-Biphenylene Bridged Polybissilsesquioxane[J]. Chem. Asian J. 2013, 8(11): 2714–2720
Wu Z W, Pang J B, Lu Y F. Synthesis of Highly-ordered Mesoporous Carbon/silica Nanocomposites and Derivative Hierarchically Mesoporous Carbon from a Phenyl-bridged Organosiloxane[J]. Nanoscale, 2009, 1(2): 245–249
Suzuki M, Owa S, Yumoto M, et al. New l-Valine-Based Hydrogelators: Formation of Supramolecular Hydrogels[J]. Tetrahedron Lett., 2004, 45(28): 5399–5402
McCulloch D G, Prawer S, Hoffman A. Structural Investigation of Xenon-Ion-Beam-Irradiated Glassy Carbon[J]. Phys. Rev. B, 1994, 50(9): 5905–5917
Inagaki S, Guan S, Ohsuna T, et al. An Ordered Mesoporous Organosilica Hybrid Material with a Crystal-Like Wall Structure[J]. Nature, 2002, 416(6878): 304–307
Lv Y Y, Zhang F, Dou Y Q, et al. A Comprehensive Study on KOH Activation of Ordered Mesoporous Carbons and Their Supercapacitor Application[J]. J. Mater. Chem., 2012, 22(1): 93–99
Author information
Authors and Affiliations
Corresponding author
Additional information
Funded by the National Natural Science Foundation of China (No. 21574095), the Priority Academic Program Development of Jiangsu High Education Institutions (PAPD)
Rights and permissions
About this article
Cite this article
Xiao, Z., Guo, Y., Li, B. et al. Preparation and characterization of single-handed helical carbonaceous nanofibers using 1,4-phenylene bridged polybissilsesquioxanes. J. Wuhan Univ. Technol.-Mat. Sci. Edit. 31, 1149–1154 (2016). https://doi.org/10.1007/s11595-016-1504-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11595-016-1504-7