Abstract
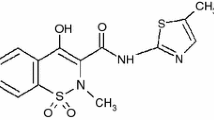
The polyoxometalate (CPFX·HCl)3H3PW12O40·8H2O was prepared and characterized by elemental analysis, IR spectra and TG-DTA-DTG. The thermal decomposition mechanism and non-isothermal kinetic parameters of the polyoxometalate were obtained from the analysis of TG-DTG data using the Achar equation, Coats-Redfern equation (CR), Madhusudanan-Krishnan-Ninan equation (MKN) and Horowitz-Metzger equation (HM). And their mathematical expressions of the kinetic compensation effect were also calculated.
Similar content being viewed by others
References
Zhang L J, Zhou Y S, Yu Z, et al. Synthesis and Structural Characterization of Two New Polyoxometalate-based Organic/Inorganic Charge-transfer Hybrids[J]. J. Mol. Struc., 2001,570: 83–90
Jin S R, Zhang L M, Liu S Z, et al. Hydrothermal Synthesis and Characterization of Inorganic/Organic Hybrid Complex Containing Tunstophosphate[J]. J. Wuhan University of Technology-Mater. Sci. Ed., 2004, 19(4), 21–23
Koga H, Itoh A, Murayama S, et al. Structure-Activity Relationships of Antibacterial 6,7-and 7,8-disubsituted 1-alkyl-1,4-dihydro-4-oxoquinoline-3-carboxylic acids[J]. J. Med. Chem., 1980, 23(9): 1358–1363
López-Gresa M P, Ortiz R, Perelló L, et al. Interactions of Metal Ions with Two Quinolone Antibacterial Agents[J]. J. Inorg. Biochem., 2002, 92(1): 65–71
Juan R A, Caredmy T. Synthesis and Antibacterial Activity of Metal Complexes of Ciprofloxacin[J]. Transit. Metal Chem., 2001, 26(2): 228–236
Liu S X, Liu Y Y, Wang E B, et al. Synthesis of Heteropoly Blue of Tungstophosphate with Keggin Structure and Studies on Their Anti-HIV Activity[J]. Chem. J. Chin. Univ., 1996, 17:1188–1192 (in Chinese)
Liu S X, Wang L, Liu Y Y, et al. Studies on Synthesis and Anti-HIV Activity of Heteropoly Complexes of Tungstotianophospha tes[J]. Chin. J. Rare Earth Soc., 1997, 15(1): 59–63 (in Chinese)
Wang D J, Fang Z D, Han D Y. Thermal Decomposition Mechanism and Non-isothermal Kinetics of the Polyoxometalate of Ciprofloxacin with 12-tungstoboric Acid[J]. Rare Met., 2005, 24(1): 15–21
Wang D J, Fang Z D, Han D Y. Thermal Behaviour of Some New Polyoxometalate Complexes of Ciprofloxacin with Keggintype Heteropoly Acids[J]. Chem. Res. Chin. Univ., 2005, 21(3):258–263
Claude R D, Michel F, Raymonde F, et al. Vibrational Investigations of Polyoxometalates[J]. Inorg. Chem., 1983, 22(2): 207–216
Jose A G, Fatima A S C, Cristina M N T, et al. Investigation of the Thermal Decomposition of Some Metal-substituted Keggin Tungstophosphates[J]. Thermochim. Acta, 1999, 326: 165–174
Wang Z P, Niu J Y, Xu L, et al. Study on Thermal Property of Keggin Heteropoly Acids[J]. Acta Chimica Sinica, 1995, 53(5):757–761 (in Chinese)
Powder Diffraction File. PCPDFWIN, Version 2.02, PDFnumber 47-1320, USA: International Centre for Diffraction Data, 1999
Achar B N N, Bridley G W, Sharp J H. Thermal Decomposition Kinetics of Some New Unsaturated Polyesters[J]. Proc. Int. Clay Conf., 1966, 1: 67–73
Coats A W, Redfern J P. Thermal Studies on Some Metal Complexes of Hexamethyl Leniminecarbo-dithioate[J]. Nature, 1964, 201(1): 67–72
Liu Z H. Handbook of Analytical Chemistry, the 2nd edition, Thermal Analysis, Part 8[M], Beijing: Chemical Industry Press, 2000, 372 (in Chinese)
Horowitz H H, Metzger G. Analysis of Thermogravimetric Traces[J]. Anal. Chem., 1963, 35: 1465–1473
Madhusudhanan P M, Krishnan K, Ninan K N. New Approximation for the P(x) Function in the Evaluation of Non-isothermal Kinetic Data[J]. Thermochim. Acta, 1986, 97: 189–195
Zsako J. Kinetics analysis of Thermogravimetric Data[J]. J. Therm. Anal., 1976, 9(1): 101–108
Author information
Authors and Affiliations
Additional information
Funded by the Natural Science Foundation of the Educational Commission of Hubei Province (No. J200522002 and Z200622001)
Rights and permissions
About this article
Cite this article
Wang, D., Fang, Z. & Lu, L. Thermal behavior and non-isothermal kinetics of the polyoxometalate of ciprofloxacin with tungstophosphoric acid. J. Wuhan Univ. Technol. 22, 240–244 (2007). https://doi.org/10.1007/s11595-005-2240-6
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11595-005-2240-6