Abstract
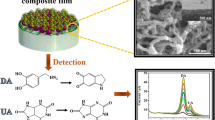
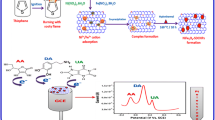
In recent years, metal–organic framework (MOF) has become a hot electrode material for electrochemistry. However, it has rarely been studied in uric acid detection. In this paper, cubic Ni-based MOF has been synthesized by Ni(NO3)2·6H2O and H3BTC in room temperature; then, Au nanoparticles (Au NPs) were decorated on Ni-MOF by in situ synthesis method. This electrochemical sensor can response to uric acid (UA) in solution and enhance response signal of UA detection. Next, some experiment conditions including pH, scan rate, and proportion between Au NPS and Ni-MOF have been optimized with cyclic voltammetry (CV) and differential pulse voltammetry (DPV). And then, the Au/Ni-MOF has been charactered with scanning electron microscope, X-ray diffraction, and X-ray photoelectron spectroscopy. Experiment finding, this electrochemical sensor has a wide linearity range from 15 to 500 μM with a low detection limit of 5.6 μM (S/N = 3). Besides, the sensor constructed also has excellent selectivity and stability. Finally, the Au/Ni-MOF sensor was successfully applied in uric acid detection in the body’s serum.









Similar content being viewed by others
Change history
17 September 2022
A Correction to this paper has been published: https://doi.org/10.1007/s11581-022-04742-w
References
Arif D, Hussain Z, Sohail M, Liaqat MA, Khan MA, Noor T (2020) A non-enzymatic electrochemical sensor for glucose detection based on Ag@TiO2@ metal-organic framework (ZIF-67) Nanocomposite. Front Chem 8:573510. https://doi.org/10.3389/fchem.2020.573510
Du J, Yue R, Yao Z, Jiang F, Du Y et al (2013) Nonenzymatic uric acid electrochemical sensor based on graphene-modified carbon fiber electrode. Colloid Surface A 419:94–99. https://doi.org/10.1016/j.colsurfa.2012.11.060
Mahmoudian MR, Basirun WJ, Sookhakian M, Woi PM, Zalnezhad E et al (2019) Synthesis and characterization of α-Fe2O3/polyaniline nanotube composite as electrochemical sensor for uric acid detection. Adv Powder Technol 30:384–392. https://doi.org/10.1016/j.apt.2018.11.015
Azeredo NFB, Goncalves JM, Rossini PO, Araki K, Wang J, Angnes L (2020) Uric acid electrochemical sensing in biofluids based on Ni/Zn hydroxide nanocatalyst. Mikrochim Acta 187:379. https://doi.org/10.1007/s00604-020-04351-2
Guo X, Yue H, Song S, Huang S, Gao X et al (2020) Simultaneous electrochemical determination of dopamine and uric acid based on MoS2nanoflowers-graphene/ITO electrode. Microchem J 154:104527. https://doi.org/10.1016/j.microc.2019.104527
Bi Y-S, Liu B, Liu X-Y, Qin Y, Zou B-X (2020) A h-BCN for electrochemical sensor of dopamine and uric acid. J Nanomater 2020:1–9. https://doi.org/10.1155/2020/4604820
Lu S, Hummel M, Chen K, Zhou Y, Kang S, Gu Z (2020) Synthesis of Au@ZIF-8 nanocomposites for enhanced electrochemical detection of dopamine. Electrochem Commun 114:106715. https://doi.org/10.1016/j.elecom.2020.106715
Liu T, Zhou M, Pu Y, Liu L, Li F et al (2021) Silver nanoparticle-functionalized 3D flower-like copper (II)-porphyrin framework nanocomposites as signal enhancers for fabricating a sensitive glutathione electrochemical sensor. Sens Actuators B 342:130047. https://doi.org/10.1016/j.snb.2021.130047
Li Y, Xie M, Zhang X, Liu Q, Lin D et al (2019) Co-MOF nanosheet array: A high-performance electrochemical sensor for non-enzymatic glucose detection. Sens Actuators B 278:126–132. https://doi.org/10.1016/j.snb.2018.09.076
Goncalves JM, Martins PR, Rocha DP, Matias TA, Juliao MSS et al (2021) Recent trends and perspectives in electrochemical sensors based on MOF-derived materials. J Mater Chem C 9:8718–8745. https://doi.org/10.1039/d1tc02025k
Qiu Z, Yang T, Gao R, Jie G, Hou W (2019) An electrochemical ratiometric sensor based on 2D MOF nanosheet/Au/polyxanthurenic acid composite for detection of dopamine. J Electroanal Chem 835:123–129. https://doi.org/10.1016/j.jelechem.2019.01.040
Stephenson CJ, Whitford CL, Stair PC, Farha OK, Hupp JT (2016) Chemoselective hydrogenation of crotonaldehyde catalyzed by an Au@ZIF-8 composite. ChemCatChem 8:855–860. https://doi.org/10.1002/cctc.201501171
Jiang X, Zhao C, Zhong C, Li J (2017) The electrochemical sensors based on MOF and their applications. Progress in Chemistry 29:1206–1214. https://doi.org/10.7536/pc170619
Ma Z-Z, Ma Y, Liu B, Xu L, Jiao H (2021) A high-performance Co-MOF non-enzymatic electrochemical sensor for glucose detection. New J Chem 45:21350–21358. https://doi.org/10.1039/d1nj04480j
Rani S, Sharma B, Malhotra R, Kumar S, Varma RS, Dilbaghi N (2020) Sn-MOF@CNT nanocomposite: an efficient electrochemical sensor for detection of hydrogen peroxide. Environ Res 191:110005. https://doi.org/10.1016/j.envres.2020.110005
Tang J, Liu Y, Hu J, Zheng S, Wang X et al (2020) Co-based metal-organic framework nanopinnas composite doped with Ag nanoparticles: a sensitive electrochemical sensing platform for simultaneous determination of dopamine and acetaminophen. Microchem J 155:104759. https://doi.org/10.1016/j.microc.2020.104759
Wu Z, Zhao DY, Dong Y, Li Y (2020) Raman-tag labelled Au@ZIF-8 for cell metabolism monitoring in vitro. Clin Hemorheol Microcirc 75:489–498. https://doi.org/10.3233/CH-200861
Yang H (2019) Highly sensitive electrochemical biosensor assembled by Au nanoparticle/MOF-5 composite electrode for DNA detection. Int J Electrochem Sci 14:5491–507. https://doi.org/10.20964/2019.06.49
Huang X, Huang D, Chen J, Ye R, Lin Q, Chen S (2020) Fabrication of novel electrochemical sensor based on bimetallic Ce-Ni-MOF for sensitive detection of bisphenol A. Anal Bioanal Chem 412:849–860. https://doi.org/10.1007/s00216-019-02282-3
Xue Z, Jia L, Zhu R-R, Du L, Zhao Q-H (2020) High-performance non-enzymatic glucose electrochemical sensor constructed by transition nickel modified Ni@Cu-MOF. J Electroanal Chem 858:113783. https://doi.org/10.1016/j.jelechem.2019.113783
Chen J, Yin H, Zhou J, Wang L, Gong J et al (2020) Efficient nonenzymatic sensors based on Ni-MOF microspheres decorated with Au nanoparticles for glucose detection. J Electron Mater 49:4754–4763. https://doi.org/10.1007/s11664-020-08191-x
Zhang X, Xu Y, Ye B (2018) An efficient electrochemical glucose sensor based on porous nickel-based metal organic framework/carbon nanotubes composite (Ni-MOF/CNTs). J Alloys Compd 767:651–656. https://doi.org/10.1016/j.jallcom.2018.07.175
Zeraati M, Alizadeh V, Kazemzadeh P, Safinejad M, Kazemian H, Sargazi G (2021) A new nickel metal organic framework (Ni-MOF) porous nanostructure as a potential novel electrochemical sensor for detecting glucose. J Porous Mater. https://doi.org/10.1007/s10934-021-01164-3
Guo H, Zhang T, Wang M, Sun L, Zhang J et al (2021) Electrochemical behavior of MOF-801/MWCNT-COOH/AuNPs: a highly selective electrochemical sensor for determination of guanine and adenine. Colloid Surf A 627:127195. https://doi.org/10.1016/j.colsurfa.2021.127195
Xiao L, Xu R, Yuan Q, Wang F (2017) Highly sensitive electrochemical sensor for chloramphenicol based on MOF derived exfoliated porous carbon. Talanta 167:39–43. https://doi.org/10.1016/j.talanta.2017.01.078
Kurniawan F, Kiswiyah NSA, Madurani KA, Tominaga M (2018) Electrochemical sensor based on single-walled carbon nanotubes-modified gold electrode for uric acid detection. J Electrochem Soc 165:B515–B522. https://doi.org/10.1149/2.0991811jes
Murugan N, Chan-Park MB, Sundramoorthy AK (2019) Electrochemical detection of uric acid on exfoliated nanosheets of graphitic-like carbon nitride (g-C3N4) based sensor. J Electrochem Soc 166:B3163–B3170. https://doi.org/10.1149/2.0261909jes
Zheng C, Pei F, Feng S, Wu Y, Ding Y, Lei W (2020) Electrochemical synthesis of nickel–copper alloy nanocomposite to fabricate an electrochemical sensor for uric acid. NANO 15:2050153. https://doi.org/10.1142/s1793292020501532
Li G, Liu S, Liu D, Zhang N (2021) MOF-derived porous nanostructured Ni2P/C material with highly sensitive electrochemical sensor for uric acid. Inorg Chem Commun 130:108713. https://doi.org/10.1016/j.inoche.2021.108713
Lv J, Li C, Feng S, Chen S-M, Ding Y et al (2019) A novel electrochemical sensor for uric acid detection based on PCN/MWCNT. Ionics 25:4437–4445. https://doi.org/10.1007/s11581-019-03010-8
Buledi JA, Ameen S, Memon SA, Fatima A, Solangi AR et al (2021) An improved non-enzymatic electrochemical sensor amplified with CuO nanostructures for sensitive determination of uric acid. Open Chem 19:481–491. https://doi.org/10.1515/chem-2021-0029
Eryigit M, Kurt Urhan B, Dogan HO, Ozer TO, Demir U (2022) ZnO nanosheets-decorated ERGO layers: an efficient electrochemical sensor for non-enzymatic uric acid detection. IEEE Sens J 22:5555–5561. https://doi.org/10.1109/jsen.2022.3150088
Pan Y, Zuo J, Hou Z, Huang Y, Huang C (2020) Preparation of electrochemical sensor based on zinc oxide nanoparticles for simultaneous determination of AA, DA, and UA. Front Chem 8:592538. https://doi.org/10.3389/fchem.2020.592538
Mazzara F, Patella B, Aiello G, O’Riordan A, Torino C et al (2021) Electrochemical detection of uric acid and ascorbic acid using r-GO/NPs based sensors. Electrochim Acta 388:138652. https://doi.org/10.1016/j.electacta.2021.138652
Funding
This work was sponsored by the Chongqing Natural Science Foundation of China (CSTC-2018JSCX-MSYBX014).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: In the originally published article, the Abstract section was incorrectly captured during typesetting. The correct Abstract section is given here.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, F., Liu, L., Liu, T. et al. Ni-MOF nanocomposites decorated by au nanoparticles: an electrochemical sensor for detection of uric acid. Ionics 28, 4843–4851 (2022). https://doi.org/10.1007/s11581-022-04712-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-022-04712-2