Abstract
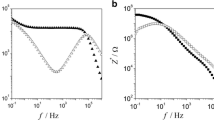
Dielectric properties of cross-linked poly(ethylene oxide) (PEO) with different mesh sizes, doped with lithium salt lithium bis(trifluoromethanesulfonyl)imide (LiTFSI), have been studied in frequency region between 0.1 and 107 Hz and in broad temperature range. Results were compared with linear PEO of 1000 g/mol. Dielectric responses of the systems are dependent on frequency and thermally activated. Systems exhibit different responses in semi-crystalline and molten state. Increase of temperature promotes polarization; whereas, increase of frequency lessens it. In other words, polarization is thermally activated and local conductivity reduced. Generally, one observes enhanced dc conductivity in linear PEO as compared to cross-linked PEO at high temperature and the opposite at low temperature. Resonance responses are observed in low-molecular cross-linked PEO and in linear PEO at low temperature. These responses lead to splitting of polarization relaxation at frequencies beyond low-frequency range. Salt-comprising systems display only relaxation-type dielectric response. Imaginary parts of response spectra show distribution of relaxation times. It turns out that this distribution is independent of temperature in the low-frequency range, but depends on concentration of salt in the cross-linked polymer. In both systems, neat cross-linked and linear polymer of low-molecular mass, one observes coexistence of non-local and local motions of charged entities even at very low temperature.











Similar content being viewed by others
References
Bruce PG, Vincent CA (1993) Polymer electrolytes. J Chem Soc Faraday Trans 89:3187–3191
Shi J, Vincent CA (1993) Dc polarization of polymer electrolytes. Solid State Ionics 60:11–18
Bernson A, Lindgren J, Huang W, Frech R (1995) Coordination and conformation in PEO, PEGM and PEG systems containing lithium or lanthanum triflate. Polymer 36:4471–4478
Marzantowicz M, Dygas JR, Krok F, Florjanczyk Z, Zygadlo-Monikowska E (2006) Influence of crystallization on dielectric properties of PEO:LiTFSI polymer electrolyte. J Non-Cryst Solids 35:5216–5223
Polu AR, Kumar R (2011) Impedance spectroscopy and FTIR studies of PEG-based polymer electrolyte. E-J Chem 8:347–353
Sun J, Stone GM, Belsara NB, Zuckermann RN (2012) Structure-conductivity relationships for peptoid-based PEO-mimetic polymer electrolytes. Macromolecules 45:5151–5156
Florjanczyk Z, Zygadlo-Monikowska E, Ostrowska J, Frydrych A (2014) Solid polymer electrolytes based on ethylene oxide polymers. Polimery 59:80–87
Das S, Ghosh A (2015) Ionic conductivity and dielectric permittivity of PEO-LiClO4 solid polymer electrolytes plasticized with polypropylene carbonate. AIP Adv 5:027125
Liu W, Liu N, Sun J, Hsu PC, Li Y, Lee HW, Cui Y (2015) Ionic conductivity enhancement of polymer electrolytes with ceramic nanowire fillers. Nano Lett 15:2740–2745
Golodnitzky D, Strauss E, Peled E, Greenbaum S (2015) Review on order and disorder in polymer electrolytes. J Electrochem Soc 162:A2551–A2566
Chan CH, Kammer HW (2017) Impedance spectra of polymer electrolytes. Ionics 23:2327–2337
Shen M, Lin Y, Li M, Nan CW (2007) High dielectric performance of polymer composite films induced by a percolating inter-particle barrier. Adv Mater 19:1418–1422
Smith RC, Liang C, Landry M, Nelson JK, Schadler LS (2008) The mechanism leading to useful electric properties of polymer dielectrics. IEEE Dielct El In 15:187–196
Lin W, Song MS, Kong B, Cui Y (2017) Flexible and stretchable energy storage: advances and future perspectives. Adv Mater 29(1-34):1603436
Carvalho M, Guegan P, Cheradame H, Gomes AS (2000) Variation of the mesh size of PEO-based networks filled with TFSILi: from an Arrhenius to WLF type conductivity behavior. Eur Polym J 36:401–409
Samiullah MH, Reichert D, Zinkevich T, Kressler J (2013) NMR characterization of PEG networks synthsized by CuAAC using reactive oligomers. Macromolecules 46:6922–6930
Pulst M, Kressler J (2014) Characterization of linear and cross-linked poly(ethylene oxide), MSc Thesis M. Pulst, Martin-Luther University Halle-Wittenberg, Halle, Germany
Fetters LF, Lohse DJ, Millner ST, Grasley WW (1999) Packing length in linear polymer melts on the entanglement. Macromolecules 32:6847–6851
Nazmul H (2016) Master thesis, Martin-Luther-University, Halle/Saale
Cheng S, Smith DM, Li CY (2015) Anisotropic ion transport in poly(ethylene oxide)/LiClO4 solid state electrolyte tempered by grapheme oxide. Macromolecules 48:4503–4510
Cheng S, Smith DM, Pan QW, Wang SJ, Li CY (2015) Anisotropic ion transport in nano-structured solid polymer electrolytes. RSC Adv 5:48793–48810
Debye P (1929) Polar Molecules. Chemical Catalog Company, New York reprinted by Dover Publishers New York, 1945
Liang YH, Wang CC, Chen CY (2008) Synthesis and characterization of a new network polymer electrolyte containing polyether in the main chains and side chains. Eur Polym J 44:2376–2384
Brandrup J, Immergut EH, Eds (1989) Polymer handbook 3rd ed., PEO. Wiley Interscience, New York, p 545
Walter R, Selser JC, Smith M, Bogoslovov R, Piet G (2002) Network viscoelastic behavior in poly(ethylene oxide) melts: effects of temperature and dissolved LiClO4 on network structure and dynamic behavior. J Chem Phys 117:427–440
Acknowledgements
The presentation, analysis, and discussion of impedance spectra are based on data provided by Professor J. Kressler and his students, Martin-Luther University Halle-Wittenberg, Germany. The author expresses his thanks for collecting and granting of the data.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kammer, H.W. Dielectric relaxation in PEO-based polymer electrolytes. Ionics 24, 1415–1428 (2018). https://doi.org/10.1007/s11581-017-2290-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-017-2290-8