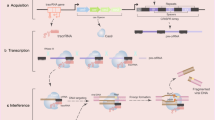
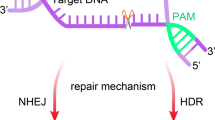
Abstract
Although germline editing has been the subject of debate ever since the 1980s, it tended to be based rather on speculative assumptions until April 2015, when CRISPR/Cas9 technology was used to modify human embryos for the first time. This article combines knowledge about the technical and scientific state of the art, economic considerations, the legal framework and aspects of clinical reality. A scenario will be elaborated as a means of identifying key ethical implications of CRISPR/Cas9 genome editing in humans and possible ways of dealing with them. Unlike most other discussions of CRISPR/Cas9 germline editing, which are generally based on deontological arguments, the focus in this case will be on a consequentialistic argument against certain applications of germline and somatic editing that takes not only the potential benefits and risks but also socioeconomic issues into consideration. The practical need for an indication catalogue, guidelines for clinical trials, and for funding of basic research will be pointed out. It will be argued that this need for regulatory action and discussion does not stem primarily from the fact that CRISPR/Cas9 germline editing is revolutionary in terms of its ethical implications and potential for human therapy, although this is the prevailing view in the current discussion. Understanding the value and interest dependency of arguments put forward by different stakeholders and learning from past debates related to similar technologies might prove a fruitful method of reaching judgments and decisions that come closer to a consensus upon which society as a whole can agree - which after all should be the true goal of an ethical debate and of bioethics.
Similar content being viewed by others
Notes
Transcription activator-like effector nuclease
Zinc-finger nucleases
References
Liang P, Xu Y, Zhang X, Ding C, Huang R, Zhang Z, Lv J, Xie X, Chen Y, Li Y, Sun Y, Bai Y, Songyang Z, Ma W, Zhou C, Huang J (2015) CRISPR/Cas9-mediated gene editing in human tripronuclear zygotes. Protein Cell 6(5):363–372. doi:10.1007/s13238-015-0153-5
Doudna JA, Charpentier E (2014) The new frontier of genome engineering with CRISPR-Cas9. Science 346(6213):1077. doi:10.1126/science.1258096
Cox DBT, Platt RJ, Zhang F (2015) Therapeutic genome editing: prospects and challenges. Nat Med 21(2):121–131. doi:10.1038/nm.3793
Gersbach CA (2014) Genome engineering: the next genomic revolution. Nat Methods 11(10):1009–1011. doi:10.1016/j.tibtech.2013.04.004
Mussolino C, Mlambo T, Cathomen T (2015) Proven and novel strategies for efficient editing of the human genome. Curr Opin Pharmacol 24:105–112. doi:10.1016/j.coph.2015.08.008
Singh P, Schimenti JC, Bolcun-Filas E (2015) A mouse geneticist’s practical guide to CRISPR applications. Genetics 199(1):1–15. doi:10.1534/genetics.114.169771
Ran Le Cong FA, Cox D, Lin S, Barretto R, Habib N, Hsu PD, Wu X, Jiang W, Marraffini LA, Zhang F (2013) Multiplex genome engineering using CRISPR/Cas systems. Science 339(6121):819–823. doi:10.1126/science.1231143
Bosley KS, Botchan M, Bredenoord AL, Carroll D, Charo RA, Charpentier E, Cohen R, Corn J, Doudna J, Feng G, Greely HT, Isasi R, Ji W, Kim JS, Knoppers B, Lanphier E, Li J, Lovell-Badge R, Martin GS, Moreno J, Naldini L, Pera M, Perry ACF, Venter JC, Zhang F, Zhou Q (2015) CRISPR germline engineering—the community speaks. Nat Biotechnol 33(5):478–486. doi:10.1038/nbt.3227
Lanphier E, Urnov F, Haecker SE, Werner M, Smolenski J (2015) Don’t edit the human germ line. Nature 519(7544):410–411. doi:10.1038/519410a
Vogel G (2015) Embryo engineering alarm. Science 347(6228):1301. doi:10.1126/science.347.6228.1301
Norman C (1983) Clerics urge ban on altering germline cells. Science 220(4604):1360–1361
Torgersen H, Schmidt M (2013) Frames and comparators: how might a debate on synthetic biology evolve? Futures 48(100):44–54. doi:10.1016/j.futures.2013.02.002
AJ Newson, A Wrigley (2015) Identifying key developments, issues and questions relating to techniques of genome editing with engineered nucleases. Background paper. http://nuffieldbioethics.org/wp-content/uploads/Genome-Editing-Briefing-Paper-Newson-Wrigley.pdf. Accessed 1 Dec 2015
Sarewitz D (2015) CRISPR: science can’t solve it. Nature 522(7557):413–414. doi:10.1038/522413a
Ledford H (2015) CRISPR the disruptor. Nature 522(7554):20–24. doi:10.1038/522020a
Kaiser J, Normile D (2015) Embryo engineering study splits scientific community. Science 348(6234):486–487. doi:10.1126/science.348.6234.486
Pollack R (2015) Eugenics lurk in the shadow of CRISPR. Science 348(6237):871. doi:10.1126/science.348.6237.871-a
Miller HI (2015) Germline gene therapy: we’re ready. Science 348(6241):1325. doi:10.1126/science.348.6241.1325-a
Krishan K, Kanchan T, Singh B (2016) Human genome editing and ethical considerations. Sci Eng Ethics 22(2):597–599. doi:10.1007/s11948-015-9675-8
Goldim JR (2015) Genetics and ethics: a possible and necessary dialogue. J Community Genet 6(3):193–196. doi:10.1007/s12687-015-0232-6
Sugarman J (2015) Ethics and germline gene editing. EMBO Rep 16(8):879–880. doi:10.15252/embr.201540879
Carroll D, Charo RA (2015) The societal opportunities and challenges of genome editing. Genome Biol 16(1):242. doi:10.1186/s13059-015-0812-0
Araki M, Ishii T (2014) International regulatory landscape and integration of corrective genome editing into in vitro fertilization. Reprod Biol Endocrinol 12:108. doi:10.1186/1477-7827-12-108
Ishii T (2015) Germline genome-editing research and its socioethical implications. Trends Mol Med 21(8):473–481. doi:10.1016/j.molmed.2015.05.006
Merlot J (2015) Umstrittene Experimente: Forscher manipulieren Erbgut menschlicher Embryonen. SPIEGEL, 24 April 2015. http://www.spiegel.de/wissenschaft/medizin/forscher-manipulieren-gene-menschlicher-embryonen-a-1030142.html. Accessed 1 Dec 2015
Focus (2015) Forscher aus China verändern Erbgut von Embryos. Focus, 24 April 2015. http://www.focus.de/wissen/mensch/tabubruch-der-gentechnik-chinesische-forscher-veraendern-erbgut-von-embryos_id_4635723.html. Accessed 1 Dec 2015
Schlütter J (2015) Tagesspiegel, 24 April 2015. Die Weltgemeinschaft sollte über ethische Grenzen diskutieren! http://www.tagesspiegel.de/wissen/gentechnisch-optimierte-embryonen-dieweltgemeinschaft-sollte-ueber-ethische-grenzen-diskutieren/11681026.html. Accessed 1 Dec 2015
Zinkant K (2015) Eine Grenze ist überschritten. Süddeutsche Zeitung, 25 April 2015. http://www.sueddeutsche.de/wissen/genetische-manipulation-eine-grenze-ist-ueberschritten-1.2452395. Accessed 1 Dec 2015
Bahnsen U (2015) Der Mensch kann seine Evolution nun selbst bestimmen. ZEIT, 23 April 2015. http://www.zeit.de/wissen/gesundheit/2015-04/genetik-erbgut-embryo-china. Accessed 1 Dec 2015
Zimmer C (2015) Editing human embryos: so this happened. http://phenomena.nationalgeographic.com/2015/04/22/editing-human-embryos-so-this-happened. Accessed 1 Dec 2015
Economist (2015) Editing Humanity. Economist, 22 August 2015. http://www.economist.com/news/leaders/21661651-new-technique-manipulating-genes-holdsgreat-promise-rules-are-needed-govern-its. Accessed 1 Dec 2015
Baltimore D, Berg P, Botchan M, Carroll D, Charo RA, Church G, Corn JE, Daley GQ, Doudna JA, Fenner M, Greely HT, Jinek M, Martin GS, Penhoet E, Puck J, Sternberg SH, Weissman JS, Yamamoto KR (2015) A prudent path forward for genomic engineering and germline gene modification. Science 348(6230):36–38. doi:10.1126/science.aab1028
Berg P (2008) Meetings that changed the world: Asilomar 1975: DNA modification secured. Nature 455(7211):290–291. doi:10.1038/455290a
Kaebnick GE (2015) A Moratorium on Gene Editing? http://www.thehastingscenter.org/Bioethicsforum/Post.aspx?id=7359&blogid=140. Accessed 1 Dec 2015
Heavey P (2013) Synthetic biology ethics: a deontological assessment. Bioethics 27(8):442–452. doi:10.1111/bioe.12052
Ter Meulen R, Biller-Andorno N, Newson A, Hunter D (2013) How to object to radically new technologies on the basis of justice: the case of synthetic biology. Bioethics 27(8):426–434. doi:10.1111/bioe.12049
Smith K (2013) Synthetic biology: a utilitarian perspective. Bioethics 27(8):453–463. doi:10.1111/bioe.12050
Smith KR, Chan S, Harris J (2012) Human germline genetic modification: scientific and bioethical perspectives. Arch Med Res 43(7):491–513. doi:10.1111/bioe.12050
Pugh J (2015) Autonomy, natality and freedom: a liberal re-examination of habermas in the enhancement debate. Bioethics 29(3):145–152. doi:10.1111/bioe.12082
Sunkara SK, Rittenberg V, Raine-Fenning N, Bhattacharya S, Zamora J, Coomarasamy A (2011) Association between the number of eggs and live birth in IVF treatment: an analysis of 400 135 treatment cycles. Hum Reprod 26(7):1768–1774. doi:10.1093/humrep/der106
Das SK, Menezes ME, Bhatia S, Wang X-Y, Emdad L, Sarkar D, Fisher PB (2015) Gene therapies for cancer: strategies, challenges and successes. J Cell Physiol 230(2):259–271. doi:10.1002/jcp.24791
Ledford H (2015) Mini enzyme moves gene editing closer to the clinic. Nature 520(7545):18. doi:10.1038/520018a
Kendler KS (2013) What psychiatric genetics has taught us about the nature of psychiatric illness and what is left to learn. Mol Psychiatry 18(10):1058–1066. doi:10.1038/mp.2013.50
Lander ES (2015) Brave new genome. N Engl J Med 373(1):5–8. doi:10.1056/NEJMp1506446
Powledge TB (2014) Whole-genome sequencing in your doctor’s office? A reality check, but sooner than later. http://www.geneticliteracyproject.org/2014/03/25/whole-genome-sequencing-in-your-doctors-office-a-reality-check-but-sooner-than-later/. Accessed 1 Dec 2015
Winand R, Hens K, Dondorp W, de Wert G, Moreau Y, Vermeesch JR, Liebaers I, Aerts J (2014) In vitro screening of embryos by whole-genome sequencing: now, in the future or never? Hum Reprod 29(4):842–851. doi:10.1093/humrep/deu005
Hens K, Dondorp W, de Wert G (2015) A leap of faith? An interview study with professionals on the use of mitochondrial replacement to avoid transfer of mitochondrial diseases. Hum Reprod 30(5):1256–1262. doi:10.1093/humrep/dev056
Hammitt JK (2002) QALYs versus WTP. Risk Anal 22(5):985–1001
EMA, Guideline on the quality, non-clinical and clinical aspects of gene therapy medicinal products: Draft. http://www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2015/05/WC500187020.pdf. Accessed 1 Dec 2015
U.S. Department of Health and Human Services- FDA- Center for Biologics Evaluation (2006) Gene therapy clinical trials - observing subjects for delayed adverse events: guidance for industry. Accessed 1 Dec 2015
Europäisches Parlament und Europäischer Rat (2009) RICHTLINIE 2009/120/EG, 2009
Knight FH (1921) Risk, uncertainty and profit. Hart, Schaffner & Marx, Houghton Mifflin, Boston, MA
DACEHTA-Danish Centre for Health Technology Assessment (2015) Health technology assessment handbook. http://www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2015/05/WC500187020.pdf. Accessed 1 Dec 2015
Nature Medicine (2015) Germline editing: time for discussion. Nat Med 21(4):295. doi:10.1038/nm.3845
Morrison C (2015) $1-million price tag set for glybera gene therapy. Nat Biotechnol 33(3):217–218. doi:10.1038/nbt0315-217
ECDC & EMEA (2009) The bacterial challenge - time to react: a call to narrow the gap between multidrug-resistant bacteria in the EU and development of new antibacterial agents, 2009. Accessed 1 Dec 2015
Kuhrt N (2013) Was darf ein Monat Leben kosten? Frankfurter Allgemeine Zeitung, 20 January 2010. http://www.faz.net/aktuell/wissen/medizin/krebstherapie-was-darf-ein-monat-leben-kosten-1907448.html. Accessed 1 Dec 2015
Wirth T, Parker N, Ylä-Herttuala S (2013) History of gene therapy. Gene 525(2):162–169. doi:10.1016/j.gene.2013.03.137
Clinicaltrials.gov. Accessed 1 Dec 2015
Knoppers BM, Chadwick R (2005) Human genetic research: emerging trends in ethics, nature reviews. Genetics 6(1):75–79. doi:10.1038/nrg1505
Cressey D, Abbott A, Ledford H (2015) UK scientists apply for licence to edit genes in human embryos. http://www.scientificamerican.com/article/scientists-apply-for-license-to-edit-genes-in-human-embryos. Accessed 8 Dec 2015
Tu Z, Yang W, Yan S, Guo X, Li X-J (2015) CRISPR/Cas9: a powerful genetic engineering tool for establishing large animal models of neurodegenerative diseases. Mol Neurodegener 10:35. doi:10.1186/s13024-015-0031-x
Combes RD, Balls M (2014) Every silver lining has a cloud: the scientific and animal welfare issues surrounding a new approach to the production of transgenic animals. ATLA Altern Lab Anim 42(2):137–145
Reardon S (2015) NIH reiterates ban on editing human embryo DNA. http://www.nature.com/news/nih-reiterates-ban-on-editing-human-embryo-dna-1.17452. Accessed 8 Dec 2015
Fateh-Moghadam B (2011) Rechtliche Aspekte der somatischen Gentherapie. In: Fehse B, Domasch S (eds) Gentherapie in Deutschland. Dornburg: Wissenschaftlicher Verlag, pp 151–184
Reich J (ed) (2015) Genomchirurgie beim Menschen - zur verantwortlichen Bewertung einer neuen Technologie: eine Analyse der interdisziplinären Arbeitsgruppe Gentechnologiebericht. BBAW, Berlin
Johnson MH, Franklin SB, Cottingham M, Hopwood N (2010) Why the medical research council refused Robert Edwards and Patrick Steptoe support for research on human conception in 1971. Hum Reprod 25(9):2157–2174
Bundesministerium für Gesundheit. PID. http://www.bmg.bund.de/glossarbegriffe/p-q/praeimplantationsdiagnostik.html. Accessed 1 Dec 2015
van Montfoort APA, Hanssen LLP, de Sutter P, Viville S, Geraedts JPM, de Boer P (2012) Assisted reproduction treatment and epigenetic inheritance. Hum Reprod Update 18(2):171–197
Wang J, Sauer MV (2006) In vitro fertilization (IVF): a review of 3 decades of clinical innovation and technological advancement. Ther Clin Risk Manag 2(4):355–364
Karen Geraghty, Protecting the Public: Profile of Dr. Frances Oldham Kelsey. http://journalofethics.ama-assn.org/2001/07/prol1-0107.html. Accessed 1 Dec 2015
Review of scientific methods to avoid mitochondrial disease (2014) http://www.hfea.gov.uk/8807.html. Accessed 1 Dec 2015
Europäisches Parlament und Rat zur Schaffung eines Gemeinschaftskodexes für Humanarzneimittel im Hinblick auf Arzneimittel für neuartige Therapien, zur Änderung der Richtlinie 2001/83/EG des Europäischen Parlaments und des Rates zur Schaffung eines Gemeinschaftskodexes für Humanarzneimittel im Hinblick auf Arzneimittel für neuartige Therapien: RICHTLINIE 2009/120/EG, 2009.
Asher Mullard (2015) Use of personalized cancer drugs runs ahead of the science. http://www.nature.com/news/use-of-personalized-cancer-drugs-runs-ahead-of-the-science-1.18389. Accessed 1 Dec 2015
Nuffield Council (2012) Novel techniques for the prevention of mitochondrial DNA disorders: an ethical view. Nuffield Council on Bioethics, London
Mitalipov S, Wolf DP (2014) Clinical and ethical implications of mitochondrial gene transfer. Trends Endocrinol Metab 25(1):5–7. doi:10.1016/j.tem.2013.09.001
Tachibana M, Sparman M, Sritanaudomchai H, Ma H, Clepper L, Woodward J, Li Y, Ramsey C, Kolotushkina O, Mitalipov S (2009) Mitochondrial gene replacement in primate offspring and embryonic stem cells. Nature 461(7262):367–372. doi:10.1038/nature08368
Jesse Reynolds (2009) Monkeys, mitochondria, and the human germline. http://www.thehastingscenter.org/Bioethicsforum/Post.aspx?id=3904&blogid=140. Accessed 1 Dec 2015
Kouros N (2013) Eugenics concerns over mitochondrial replacement. Monash Bioeth Rev 31(2):5–6
Ball P (2014) The art of medicine: unnatural reactions. Lancet 383:1964–1965
Smajdor A, Ives J, Baldock E, Langlois A (2008) Getting from the ethical to the empirical and back again: the danger of getting it wrong, and the possibilities for getting it right. Health Care Anal 16(1):7–16. doi:10.1007/s10728-007-0079-z
Nordmann A, Rip A (2009) Mind the gap revisited. Nat Nanotechnol 4(5):273–274. doi:10.1038/nnano.2009.26
Deutscher Ethikrat (2014) Sollten Vorkern- und Spindeltransfer bei mitochondrialen Erkrankungen in Deutschland zulässig sein? http://www.ethikrat.org/dateien/pdf/jt-22-05-2014-vorkern-und-spindeltransfer.pdf. Accessed 1 Dec 2015
Engelhardt HT (2011) Confronting moral pluralism in posttraditional Western societies: bioethics critically reassessed. J Med Philos 36(3):243–260
Patenaude J, Legault GA, Béland J-P, Parent M, Boissy P (2011) Moral arguments in the debate over nanotechnologies: are we talking past each other? NanoEthics 5(3):285–293. doi:10.1007/s11569-011-0132-0
Macer D (1995) International perceptions and approval of gene therapy. Hum Gene Ther 6:791–803
Robillard JM (2015) Communicating in context: a priority for gene therapy researchers. Expert Opin Biol Ther 15(3):315–318. doi:10.1517/14712598.2015.1001735
Robillard JM, Roskams-Edris D, Kuzeljevic B, Illes J (2014) Prevailing public perceptions of the ethics of gene therapy. Hum Gene Ther 25(8):740–746. doi:10.1089/hum.2014.030
Madhusoodanan J (2015) Bioethics accused of doing more harm than good. Nature 524(7564):139
Pinker S (2015) The moral imperative for bioethics. https://www.bostonglobe.com/opinion/2015/07/31/the-moral-imperative-for-bioethics/JmEkoyzlTAu9oQV76JrK9N/story.html. Accessed 1 Dec 2015
Niu Y, Shen B, Cui Y, Chen Y, Wang J, Wang L, Kang Y, Zhao X, Si W, Li W, Xiang AP, Zhou J, Guo X, Bi Y, Si C, Hu B, Dong G, Wang H, Zhou Z, Li T, Tan T, Pu X, Wang F, Ji S, Zhou Q, Huang X, Ji W, Sha J (2014) Generation of gene-modified cynomolgus monkey via Cas9/RNA-mediated gene targeting in one-cell embryos. Cell 156(4):836–843. doi:10.1016/j.cell.2014.01.027
Wu Y, Liang D, Wang Y, Bai M, Tang W, Bao S, Yan Z, Li D, Li J (2013) Correction of a genetic disease in mouse via use of CRISPR-Cas9. Cell Stem Cell 13(6):659–662. doi:10.1016/j.stem.2013.10.016
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The author declares that she has no conflict of interest.
Rights and permissions
About this article
Cite this article
Baumann, M. CRISPR/Cas9 genome editing – new and old ethical issues arising from a revolutionary technology. Nanoethics 10, 139–159 (2016). https://doi.org/10.1007/s11569-016-0259-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11569-016-0259-0