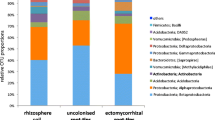
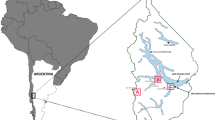
Abstract
Trechispora (Hydnodontaceae) is considered as a soil-inhabiting fungus. However, some species in the genus are frequently forming basidiomes on soil, a typical feature of ectomycorrhizal fungi. Ectomycorrhizal basidiomes are found in neotropical and subtropical region, but taxonomical information and DNA sequences of root fungi and basidiomes from native Atlantic Rainforest are poorly reported. Basidiomes and soil samples including roots, humus layer, and mineral soil were collected in the Atlantic Rainforest, in Florianópolis (South of Brazil). Sequences of the ITS region were obtained from all sample types and subjected to phylogenetic reconstruction. Two sequences amplified from apparently ectomycorrhizal roots belonged to Trechispora and suggested a root-associated ecology, at least biotrophic and possibly ectomycorrhizal. The analysis of isotope abundance in the same Brazilian site and in French Guiana showed that Trechispora thelephora has high 15N abundance and is often intermediate between ectomycorrhizal and saprotrophic species in 13C abundance. This is congruent with a plant biotrophic ecology, perhaps ectomycorrhizal. Future investigations in subtropical regions are needed to determine whether such a mode of nutrition is widespread among Trechispora.



Similar content being viewed by others
References
Agerer R (ed) (1997) Colour Atlas of Ectomycorrhizae. Einhorn-Verlag Eduard Dietenberger GmbH, Schwäbisch-Gmünd, Germany
Agerer R, Christian J, Mayr C, Hobbie E (2012) Isotopic signatures and trophic status of Ramaria. Mycol Prog 11(2):47–59
Albee-Scott S, Kropp BR (2010) A phylogenetic study of Trechispora thelephora. Mycotaxon 114:395–399
Alexander I, Selosse MA (2009) Mycorrhizas in tropical forests: a neglected research imperative. New Phytol 182:14–16
Alvarez-Manjarrez J, Villegas-Ríos M, Garibay-Orijel R, Contreras-Pacheco M, Kõljalg U (2016) Tomentella brunneoincrustata, the first described species of the Pisonieae-associated Neotropical Tomentella clade, and phylogenetic analysis of the genus in Mexico. Mycol Prog 15(1):1–11
Alvarez-Manjarrez J, Garibay-Orijel R, Smith ME (2018) Caryophyllales are the main hosts of a unique set of ectomycorrhizal fungi in a Neotropical dry forest. Mycorrhiza 28(2):103–115
Andrade ACS, Queiroz MH, Hermes RAL, Oliveira VL (2000) Mycorrhizal status of some plants of the Araucaria forest and the Atlantic rainforest in Santa Catarina, Brazil. Mycorrhiza 10(3):131–136
Brundrett MC, Tedersoo L (2018) Evolutionary history of mycorrhizal symbioses and global host plant diversity. New Phytol 220(4):1108–1115
Brundrett M, Tedersoo L (2019) Misdiagnosis of mycorrhizas and inappropriate recycling of data can lead to false conclusions. New Phytol 221:18–24
Corrales A, Henkel TW, Smith ME (2018) Ectomycorrhizal associations in the tropics–biogeography, diversity patterns and ecosystem roles. New Phytol 220(4):1076–1091
Darriba D, Taboada GL, Doallo R, Posada D (2012) jModelTest 2: more models, new heuristics and parallel computing. Nat Methods 9(8):772–772
Doyle JJ, Doyle JL (1987) A rapid isolation procedure for small quantities of fresh tissue. Phytochem Bull 19:11–15
Dunham SM, Larsson KH, Spatafora JW (2007) Species richness and community composition of mat-forming ectomycorrhizal fungi in old-and second growth Douglas-fir forests of the HJ Andrews experimental Forest, Oregon, USA. Mycorrhiza 17(8):633–645
Falkenberg DB (1999) Aspectos da flora e da vegetação secundária da restinga de Santa Catarina, sul do Brasil. INSULA Rev Bot 28:01
Gardes M, Bruns TD (1993) ITS primers with enhanced specificity for basidiomycetes-application to the identification of mycorrhizae and rusts. Mol Ecol 2(2):113–118
Giachini AJ, Oliveira VL, Castellano MA, Trappe JM (2000) Ectomycorrhizal fungi in Eucalyptus and Pinus plantations in southern Brazil. Mycologia 92(6):1166–1177
Giachini AJ, Souza LA, Oliveira VL (2004) Species richness and seasonal abundance of ectomycorrhizal fungi in plantations of Eucalyptus dunnii and Pinus taeda in southern Brazil. Mycorrhiza 14(6):375–381
Góes-Neto A, Loguercio-Leite C, Guerrero R (2005) DNA extraction from frozen field-collected and dehydrated herbarium fungal basidiomata: performance of SDS and CTAB-based methods. Biotemas 18(2):19–32
Guindon S, Gascuel O (2003) A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst Biol 52(5):696–704
Halbwachs H, Easton GL, Bol R, Hobbie EA, Garnett MH, Peršoh D, Dixon L, Ostle N, Karasch P, Griffith GW (2018) Isotopic evidence of biotrophy and unusual nitrogen nutrition in soil-dwelling Hygrophoraceae. Environ Microbiol 20(10):3573–3588
Halling RE (2001) Ectomycorrhizae: co-evolution, significance, and biogeography. Ann Mo Bot Gard 5:13
Haug I, Weiß M, Homeier J, Oberwinkler F, Kottke I (2005) Russulaceae and Thelephoraceae form ectomycorrhizas with members of the Nyctaginaceae (Caryophyllales) in the tropical mountain rain forest of southern Ecuador. New Phytol 165(3):923–936
Hayward J, Hynson NA (2014) New evidence of ectomycorrhizal fungi in the Hawaiian Islands associated with the endemic host Pisonia sandwicensis (Nyctaginaceae). Fungal Ecol 12:62–69
Heijden MG, Martin FM, Selosse MA, Sanders IR (2015) Mycorrhizal ecology and evolution: the past, the present, and the future. New Phytol 205(4):1406–1423
Henkel TW, Aime MC, Chin MM, Miller SL, Vilgalys R, Smith ME (2012) Ectomycorrhizal fungal sporocarp diversity and discovery of new taxa in Dicymbe monodominant forests of the Guiana Shield. Biodivers Conserv 21(9):2195–2220
Henry C, Raivoarisoa JF, Razafimamonjy A, Ramanankierana H, Andrianaivomahefa P, Ducousso M, Selosse MA (2017) Transfer to forest nurseries significantly affects mycorrhizal community composition of Asteropeia mcphersonii wildings. Mycorrhiza 27(4):321–330
Hibbett DS, Bauer R, Binder M, Giachini AJ, Hosaka K, Justo A, Larsson E, Larsson KH, Lawrey JD, Miettinen O, Nagy LG, Nilsson RH, Weiss M, Thorn RG (2014) 14 Agaricomycetes. In: Systematics and evolution. Springer, Berlin, pp 373–429
Hobbie EA, Sánchez FS, Rygiewicz PT (2012) Controls of isotopic patterns in saprotrophic and ectomycorrhizal fungi. Soil Biol Biochem 48:60–68
Hosaka K, Bates ST, Beever RE, Castellano MA, Colgan W 3rd, Domínguez LS, Nouhra ER, Geml J, Giachini AJ, Kenney SR, Simpson NB, Spatafora JW, Trappe JM (2006) Molecular phylogenetics of the gomphoid-phalloid fungi with an establishment of the new subclass Phallomycetidae and two new orders. Mycologia 98(6):949–959
Kariman K, Barker SJ, Jost R, Finnegan PM, Tibbett M (2014) A novel plant–fungus symbiosis benefits the host without forming mycorrhizal structures. New Phytol 201(4):1413–1422
Katoh K, Standley DM (2013) MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol Biol Evol 30(4):772–780
Kearse M, Moir R, Wilson A, Stones-Havas S, Cheung M, Sturrock S, Thierer T (2012) Geneious basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 28(12):1647–1649
Kirk PM, Cannon PF, David JC, Stalpers JA (eds) (2008) Ainsworth and Bisby’s dictionary of the Fungi, 10th edn. Wallingford, CABI Publishing
Largent DL, Johnson D, Watling R (1977) How to identify mushrooms to genus III: microscopic features. Mad River Press Inc., California
Larsson KH (1994) Poroid species in Trechispora and the use of calcium oxalate crystals for species identification. Mycol Res 98(10):1153–1172
Larsson KH (1996) New species and combinations in Trechispora (Corticiaceae, Basidiomycotina). Nord J Bot 16(1):83–98
Liberta AE (1973) The genus Trechispora (Basidiomycetes, Corticiaceae). Can J Bot 51(10):1871–1892
Mayor JR, Schuur EA, Henkel TW (2009) Elucidating the nutritional dynamics of fungi using stable isotopes. Ecol Lett 12(2):171–183
Miller MA, Pfeiffer W, Schwartz T (2010) Creating the CIPRES Science Gateway for inference of large phylogenetic trees. In: Proceedings of the Gateway Computing Environments Workshop (GCE). San Diego Supercomput. Center, New Orleans, LA. CA pp 1–8
Moyersoen B (2006) Pakaraimaea dipterocarpacea is ectomycorrhizal, indicating an ancient Gondwanaland origin for the ectomycorrhizal habit in Dipterocarpaceae. New Phytol 172(4):753–762
Mueller GM, Bills GF, Foster MS (2004) Biodiversity of fungi: inventory and monitoring methods. Elsevier Academic Press
Rinaldi AC, Comandini O, Kuyper TW (2008) Ectomycorrhizal fungal diversity: separating the wheat from the chaff. Fungal Divers 33:1–45
Ronquist F, Huelsenbeck JP (2003) MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19(12):1572–1574
Rosenthal LM, Larsson KH, Branco S, Chung JA, Glassman SI, Liao HL, Peay KG, Smith DP, Talbot JM, Taylor JW, Vellinga EC, Vilgalys R, Bruns TD (2017) Survey of corticioid fungi in North American pinaceous forests reveals hyperdiversity, underpopulated sequence databases, and species that are potentially ectomycorrhizal. Mycologia 109(1):115–127
Roy M, Schimann H, Braga-Neto R, Da Silva RA, Duque J, Frame D, Wartchow F, Neves MA (2016) Diversity and distribution of ectomycorrhizal fungi from Amazonian lowland white-sand forests in Brazil and French Guiana. Biotropica 48(1):90–100
Roy M, Vasco-Palacios A, Geml J, Buyck B, Delgat L, Giachini A, Grebenc T, Harrower E, Kuhar F, Magnago A, Rinaldi AC, Schimann H, Selosse M-A, Sulzbacher MA, Wartchow F, Neves M-A (2017) The (re)discovery of ectomycorrhizal symbioses in Neotropical ecosystems sketched in Florianópolis. New Phytol 214:920–923
Schimann H, Roy M, Jaouen G (2019). Fungi of French Guiana. Version 1.3. Unité Mixte de Recherche EcoFoG (Ecologie des Forêts de Guyane). Occurrence dataset https://doi.org/10.15468/ymvlrp. Accessed via GBIF.org on 22 July 2019
Schoch CL, Seifert KA, Huhndorf S, Robert V, Spouge JL, Levesque CA, Miller AN (2012) Nuclear ribosomal internal transcribed spacer (ITS) region as a universal DNA barcode marker for Fungi. Proc Natl Acad Sci 109(16):6241–6246
Selosse MA, Martos F (2014) Do chlorophyllous orchids heterotrophically use mycorrhizal fungal carbon? Trends Plant Sci 19(11):683–685
Selosse MA, Richard F, He X, Simard SW (2006) Mycorrhizal networks: des liaisons dangereuses? Trends Ecol Evol 21(11):621–628
Selosse MA, Dubois MP, Alvarez N (2009) Do Sebacinales commonly associate with plant roots as endophytes? Mycol Res 113(10):1062–1069
Selosse MA, Schneider-Maunoury L, Martos F (2018) Time to re-think fungal ecology? New Phytol 217(3):968–972
Singer R, Araujo IDJDS (1979) Litter decomposition and ectomycorrhiza in Amazonian forests. 1. A comparison of litter decomposing and ectomycorrhizal basidiomycetes in latosol-terra-firme rain forest and white podzol campinarana. Acta Amazon 9(1):25–42
Smith SE, Read DJ (2008) In: 3rd (ed) Mycorrhizal symbiosis. Academic Press, New York
Stamatakis A (2006) RAxML-VI-HPC: maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed models. Bioinformatics 22(21):2688–2690
Sulzbacher MA, Grebenc T, García MÁ, Silva BD, Silveira A, Antoniolli ZI, Marinho P, Münzenberger B, Telleria MT, Baseia IG, Martín MP (2016) Molecular and morphological analyses confirm Rhizopogon verii as a widely distributed ectomycorrhizal false truffle in Europe, and its presence in South America. Mycorrhiza 26:377–388
Sulzbacher MA, Grebenc T, Giachini AJ, Baseia IG (2017) Sclerotium-forming fungi from soils of the Atlantic rainforest of northeastern Brazil. Plant Ecol Evolut 150(3):358–362
Suvi T, Tedersoo L, Abarenkov K, Beaver K, Gerlach J, Koljalg U (2010) Mycorrhizal symbionts of Pisonia grandis and P. sechellarum in Seychelles: identification of mycorrhizal fungi and description of new Tomentella species. Mycologia 102(3):522–533
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30(12):2725–2729
Tedersoo L, Smith ME (2013) Lineages of ectomycorrhizal fungi revisited: foraging strategies and novel lineages revealed by sequences from belowground. Fungal Biol Rev 27:83–99
Tedersoo L, May TW, Smith ME (2010a) Ectomycorrhizal lifestyle in fungi: global diversity, distribution, and evolution of phylogenetic lineages. Mycorrhiza 20(4):217–263
Tedersoo L, Sadam A, Zambrano M, Valencia R, Bahram M (2010b) Low diversity and high host preference of ectomycorrhizal fungi in Western Amazonia, a neotropical biodiversity hotspot. ISME J 4(4):465–471
Tello SA, Silva-Flores P, Agerer R, Halbwachs H, Beck A, Peršoh D (2014) Hygrocybe virginea is a systemic endophyte of Plantago lanceolata. Mycol Prog 13(3):471–475
Vohnik M, Sadowsky JJ, Kohout P, Lhotáková Z, Nestby R, Kolařík M (2012) Novel root-fungus symbiosis in Ericaceae: sheathed ericoid mycorrhiza formed by a hitherto undescribed basidiomycete with affinities to Trechisporales. PLoS One 7(6):e39524
Wang B, Qiu YL (2006) Phylogenetic distribution and evolution of mycorrhizas in land plants. Mycorrhiza 16(5):299–363
White TJ, Bruns T, Lee SJWT, Taylor JW (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In Innis MA, Gelfand DH, Sninsky JJ, and White TJ (Eds) PCR protocols: A guide to methods and applications, Academic Press, San Diego, CA, pp 315–322
Wilson D (1995) Endophyte: the evolution of a term, and clarification of its use and definition. Oikos 73(2):274–276
Yokomizo NKS (1986) Micorrizas em essenciais florestas. Anais da I reunião brasileira sobre micorrizas. Lavras, Brasil, UFLA, p 112
Zeller B, Brechet C, Maurice J-P, Le Tacon F (2007) 13C and 15N isotopic fractionation in trees, soils and fungi in a natural forest stand and a Norway spruce plantation. Ann For Sci 64:419–429
Acknowledgments
The first author thanks the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) for the master’s scholarship. We thank Lina Ribeiro Venturieri and Cony Decock for help on collecting field trips, Professor Mayara Caddah for help in Guapira opposita field identification, and one anonymous reviewer for insightful comments on an earlier version of this paper. Isotopic analyses were made at SSMIM (MNHN Paris) with technical assistance of Denis Fiorillo (UMR 7209 CNRS).
Funding
The sampling in French Guiana was funded by Nouragues field station projects and by Labex grants CEBA (ANR 10-LABX-0025) and TULIP (ANR 10-LABX-0041).
Author information
Authors and Affiliations
Corresponding author
Additional information
Section Editor: Marc Stadler
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Vanegas-León, M.L., Sulzbacher, M.A., Rinaldi, A.C. et al. Are Trechisporales ectomycorrhizal or non-mycorrhizal root endophytes?. Mycol Progress 18, 1231–1240 (2019). https://doi.org/10.1007/s11557-019-01519-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11557-019-01519-w