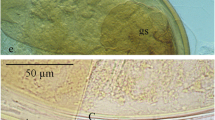
Abstract
Clavulina is an ectomycorrhizal genus of macroscopic basidiomycetes, mostly with clavarioid basidiomata, distributed worldwide. The most commonly reported species are C. amethystina, C. cinerea, C. coralloides, and C. rugosa, which are part of a species complex in temperate ecosystems. In this work, we describe two new species, C. mahiscolorata and C. parvispora, from temperate forests of central Mexico. We also present a geographical range redistribution of C. reae, which displays great macromorphological variation regarding basidiome branching rank, colors, and tip shape. We provide macromorphological, micromorphological, and habitat data for each species, as well as nuclear rDNA ITS, rDNA LSU, and RPB2 sequences. Our findings highlight an underestimation of species richness in temperate ecosystems, as a consequence of assigning existing epithets to taxa without proper morphological and molecular analyses. The designation of neotypes for temperate species of Clavulina is urgent to delimit species concepts for the genus.





Similar content being viewed by others
Change history
31 October 2019
A Correction to this paper has been published: https://doi.org/10.1007/s11557-019-01533-y
References
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic alignment search tool. J Mol Biol 215:403–410. https://doi.org/10.1016/S0022-2836(05)80360-2
Argüelles-Moyao A, Garibay-Orijel R, Márquez-Valdelamar L, Arellano-Torres E (2017) Clavulina-Membranomyces is the most important lineage within the highly diverse ectomycorrhizal fungal community of Abies religiosa. Mycorrhiza 27:53–65. https://doi.org/10.1007/s00572-016-0724-1
Bulliard JBF (1788) Herbier de la France. Tome 8. Paris. https://doi.org/10.5962/bhl.title.5365
Bulliard JBF (1790) Herbier de la France. Tome 10. Paris. https://doi.org/10.5962/bhl.title.5365
Bulliard JBF (1791) Herbier de la France. Tome 11. Paris. https://doi.org/10.5962/bhl.title.5365
Christiansen MP (1967) Clavariaceae Daniae species especially collected in the isle of Zealand. Friesia 8:117–160
CONANP-SEMARNAT (Comisión Nacional de Áreas Naturales Protegidas de la Secretaría de Medio Ambiente y Recursos Naturales) (2006) Programa de Conservación y Manejo Parque Nacional Desierto de Los Leones. Comisión Nacional de Áreas naturales Protegidas, Mexico City
Corner E (1950) A monograph of Clavaria and allied genera. Ann Bot Mem 1:1–740
Corner E (1970) Supplement to “A monograph of Clavaria and allied genera”. Beih Nova Hedwigia 47:22–51
Darriba D, Taboada GL, Doallo R, Posada D (2012) jModelTest 2: more models, new heuristics and parallel computing. Nat Methods 9:1–772. https://doi.org/10.1038/nmeth.2109
Donk MA (1964) A conspectus of the families of Aphyllophorales. Persoonia 3:199–324
Edgar R (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 132:1792–1797. https://doi.org/10.1093/nar/gkh340
García-Palomo A, Zamorano J, López-Miguel C, Galván-García A, Carlos-Valerio V, Ortega R, Macías J (2008) El arreglo morfoestructural de la Sierra de Las Cruces, México central. Rev Mex Cienc Geol 25:158–178
Gardes M, Bruns T (1993) ITS primers with enhanced specificity for basidiomycetes application to the identification of mycorrhizae and rusts. Mol Ecol 2:113–118. https://doi.org/10.1111/j.1365-294X.1993.tb00005.x
He G, Chen S-L, Yan S-Z (2016) Morphological and molecular evidence for a new species in Clavulina from southwestern China. Mycoscience 57:255–263. https://doi.org/10.1016/j.myc.2016.03.002
Henkel TW, Aime C, Uehling JK, Smith ME (2011) New species and distribution records of Clavulina (Cantharellales, Basidiomycota) from the Guiana Shield. Mycologia 103:883–894. https://doi.org/10.3852/10-355
Henkel TW, Meszaros R, Aime C, Kennedy A (2005) New Clavulina species from the Pakaraima Mountains of Guyana. Mycol Prog 4:343–350. https://doi.org/10.1007/s11557-006-0140-6
Hobbie E, Weber N, Trappe J (2001) Mycorrhizal vs. saprotrophic status of fungi: the isotopic evidence. New Phytol 150:601–610. https://doi.org/10.1046/j.1469-8137.2001.00134.x
Hobbie E, Weber N, Trappe J, Van Klinken G (2002) Using radiocarbon to determine the mycorrhizal status of fungi. New Phytol 156:129–136. https://doi.org/10.1046/j.1469-8137.2002.00496.x
Holmskjold T (1790) Beata ruris otia fungis danicis. Vol. 1. Copenhagen, Denmark
Hopple JS, Vilgalys R (1994) Phylogenetic relationships among coprinoid taxa and allies based on data from restriction site mapping of nuclear rDNA. Mycologia 86:96–107. https://doi.org/10.2307/3760723
Hou W, Lian B, Dong H, Jiang H, Wu X (2012) Distinguishing ectomycorrhizal and saprophytic fungi using carbon and nitrogen isotopic compositions. Geosci Front 3:351–356. https://doi.org/10.1016/j.gsf.2011.12.005
Kennedy P, Matheny P, Ryberg K, Henkel T, Uehling J, Smith M (2012) Scaling up: examining the macroecology of ectomycorrhizal fungi. Mol Ecol 21:4151–4154. https://doi.org/10.1111/j.1365-294X.2012.05703.x
Knudsen H, Vesterholt J (1997) Clavulinaceae (Donk) Donk, In: Hansen L & Knudsen H, (ed.), Nordic macromycetes Vol. 3 Heterobasidioid, Aphyllophoroid, and Gastromycetoid Basidiomycetes. Nordsvamp, Copenhagen, pp. 253–255
Larsson A (2014) AliView: a fast and lightweight alignment viewer and editor for large data sets. Bioinformatics 30:3276–3278. https://doi.org/10.1093/bioinformatics/btu531
Lineé CV (1753) Species Plantarum. Vol. 2. p 1182. https://doi.org/10.5962/bhl.title.669
McNeill J (2014) Holotype specimens and type citations: General issues. Taxon 63(5):1112–1113
Miller A, Pfeiffer W, Schwartz T (2010) Creating the CIPRES science gateway for inference of large phylogenetic trees. Proceedings of the Gateway Computing Environments Workshop (GCE), New Orleans, LA pp. 1–8
Moncalvo J, Nilsson R, Koster B, Dunham S, Bernauer T, Matheny P, Porter T, Margaritescu S, Weiss M, Garnica S, Danell E, Langer G, Langer E, Larsson E, Larsson K, Vilgalys R (2006) The cantharelloid clade: dealing with incongruent gene trees and phylogenetic reconstruction methods. Mycologia 98:937–948. https://doi.org/10.1080/15572536.2006.11832623
Olariaga I, Jugo B, García-Etxebarria K, Salcedo I (2009) Species delimitation in the European species of Clavulina (Cantharellales, Basidiomycota) inferred from phylogenetic analyses of ITS region and morphological data. Mycol Res 113:1261–1270. https://doi.org/10.1016/j.mycres.2009.08.008
Olariaga I, Salcedo I (2012) New combinations and notes in clavarioid fungi. Mycotaxon 121:37–44. https://doi.org/10.5248/121.37
Petersen R (1988) The clavarioid fungi of New Zealand. Mycologia 80:571–576
Phookamsak R, Hyde KD, Jeewon R, Jayarama D, Gareth D et al (2019) Fungal diversity notes 929–1035: taxonomic and phylogenetic contributions on genera and species of fungi. Fungal Divers 95:1–273. https://doi.org/10.1007/s13225-019-00421-w
Rea C (1920) New or rare British fungi. Trans Br Mycol Soc 6:61–64. https://doi.org/10.1016/S0007-1536(17)80012-7
Reverchon F, Ortega-Larrocea MDP, Bonilla-Rosso G, Pérez-Moreno J (2012) Structure and species composition of ectomycorrhizal fungal communities colonizing seedlings and adult trees of Pinus montezumae in Mexican Neotropical forests. FEMS Microbiol Ecol 80:479–487. https://doi.org/10.1111/j.1574-6941.2012.01314.x
Rinaldi C, Comandini O, Kuyper T (2008) Ectomycorrhizal fungal diversity: separating the wheat from the chaff. Fungal Divers 33:1–45
Ronquist F, Huelsenbeck JP (2003) MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19(12):1572–1574
Schröter J (1888) Die Pilze Schlesiens. In: Cohn F (ed) Kryptogamen-Flora von Schlesien, 1st edn. J.U. Kern’s Verlag Max Müller, Breslau, pp 442–443. https://doi.org/10.5962/bhl.title.1632
Stafleu FA, Cowan RS (1976) Taxonomic Literature, vol I. A-G. Bohn, Scheltema and Holkema, Utrecht. https://doi.org/10.5962/bhl.title.48631
Stamatakis A, Hoover P, Rougemont J (2008) A rapid bootstrap algorithm for the RAxML web servers. Syst Biol 57:758–771. https://doi.org/10.1080/10635150802429642
Tavaré S (1986) Some probabilistic and statistical problems in the analysis of DNA sequences. Lect Maths Life Sci 17:57–86
Tedersoo L, Smith ME (2013) Lineages of ectomycorrhizal fungi revisited: foraging strategies and novel lineages revealed by sequences from belowground. Fungal Biol Rev 27:83–99. https://doi.org/10.1016/j.fbr.2013.09.001
Tedersoo L, Kõljalg U, Hallenberg N, Larsson K-H (2003) Fine scale distribution of ectomycorrhizal fungi and roots across substrate layers including coarse woody debris in a mixed forest. New Phytol 159:153–165. https://doi.org/10.1046/j.1469-8137.2003.00792.x
Tedersoo L, May T, Smith M (2010) Ectomycorrhizal lifestyle in fungi: global diversity, distribution, and evolution of phylogenetic lineages. Mycorrhiza 20:217–263. https://doi.org/10.1007/s00572-009-0274-x
Tedersoo L, Naadel T, Bahram M, Pritsch K, Buegger F, Leal M, Urmas K, Põldmaa K (2012) Enzymatic activities and stable isotope patterns of ectomycorrhizal fungi in relation to phylogeny and exploration types in an afrotropical rain forest. New Phytol 195:832–843. https://doi.org/10.1111/j.1469-8137.2012.04217.x
Thacker JR, Henkel TW (2004) New species of Clavulina from Guyana. Mycologia 96:650–657. https://doi.org/10.1080/15572536.2005.11832961
Tibpromma S, Hyde J, Jeewon R, Maharachchikumbura S, Liu J et al (2017) Fungal diversity notes 111-252: taxonomic and phylogenetic contributions to fungal taxa. Fungal Divers 83:1–261. https://doi.org/10.1007/s13225-017-0378-0
Trudell SA, Rygiewicz PT, Edmonds RL (2004) Patterns of nitrogen and carbon stable isotope ratios in macrofungi, plants and soils in two old-growth conifer forests. New Phytol 164:317–335. https://doi.org/10.1111/j.1469-8137.2004.01162.x
Turland NJ, Wiersema JH, Barrie FR, Greuter W, Hawksworth DL, Herendeen PS, Knapp S, Kusber W-H, Li D-Z, Marhold K, May TW, McNeill J, Monro AM, Prado J, Price MJ, Smith GF (eds) (2018) International Code of Nomenclature for algae, fungi, and plants (Shenzhen Code) adopted by the Nineteenth International Botanical Congress Shenzhen, China, July 2017. Regnum Vegetabile 159. Koeltz Botanical Books, Glashütten. https://doi.org/10.12705/Code.2018
Uehling J, Henkel T, Aime C, Vilgalys R, Smith M (2012a) New species of Clavulina (Cantharellales, Basidiomycota) with resupinate and effused basidiomata from the Guiana Shield. Mycologia 104:547–556. https://doi.org/10.3852/11-130
Uehling J, Henkel T, Aime C, Vilgalys R, Smith M (2012b) New species and distribution records for Clavulina (Cantharellales, Basidiomycota) from the Guiana Shield, with a key to the lowland neotropical taxa. Fungal Biol 116:1263–1274. https://doi.org/10.1016/j.funbio.2012.09.004
Vilgalys R, Hester M (1990) Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J Bacteriol 172:4238–4246. https://doi.org/10.1128/jb.172.8.4238-4246.1990
Wanscher J, Kornerup A (1991) Farver I Farver Bestemmelse af farver. Politikens Forlag, Denmark
Wartchow F (2012) Clavulina incrustata, a new species from Pernambuco, Brazil. Cryptogam Mycol 33:105–113. https://doi.org/10.7872/crym.v33.iss1.2012.105
Ważny R (2014) Ectomycorrhizal communities associated with silver fir seedlings (Abies alba Mill.) differ largely in mature silver fir stands and in Scots pine forecrops. Ann For Sci 71:801–810. https://doi.org/10.1007/s13595-014-0378-0
White TJ, Bruns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomes RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols: a guide to methods and applications. Academic Press, New York, pp 315–322
Zeller B, Brechet C, Maurice JP, Le Tacon F (2007) 13C and 15N isotopic fractionation in trees, soils and fungi in a natural forest stand and a Norway spruce plantation. Ann Forest Sci 64:419–429. https://doi.org/10.1051/forest:2007019
Acknowledgments
The authors thank Silvia Espinoza Matías for her photographic SEM work, Laura Margarita Márquez Valdelamar and Nelly López at LaNaBio for sample sequencing, and Noemí Matías Ferrer for her collaboration in the generation of sequences. We also thank Aldi de Oyarzabal Salcedo for the illustrations of the species described. We thank three anonymous reviewers and the editor for their commentaries.
Funding
This research was funded by Universidad Nacional Autónoma de México PAPIIT grant IN-223114 and by Consejo Nacional de Ciencia y Tecnología (CONACYT) grant CONACYT- 239266. EP-P received a CONACYT fellowship for graduate studies, number 420208.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Editorial Responsibility: Zhu-Liang Yang
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Eduardo, PP., Margarita, VR., Roberto, GO. et al. Two new species of Clavulina and the first record of Clavulina reae from temperate Abies religiosa forests in central Mexico. Mycol Progress 18, 1187–1200 (2019). https://doi.org/10.1007/s11557-019-01516-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11557-019-01516-z