Abstract
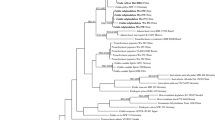
Phylloporia is shown to be monophyletic according to molecular sequence data. Morphologically, Phylloporia is characterized by annual or perennial basidiomata, a monomitic to dimitic hyphal system, generally presenting a duplex context, absence of setae, and abundant tiny, thick-walled and colored basidiospores. In this study, specimens from China and Brazil a priori identified to Phylloporia species were revised. Phylogenetic analysis based on the nuc 28S rDNA showed that they nested within the Phylloporia clade, in which they formed eight new terminal lineages. According to this result and additional morphological and ecological data (host relationships), these eight lineages are described as eight new species: Phylloporia atlantica, P. cystidiolophora, P. montana, P. perangusta, P. rattanicola, P. solicola, P. splendida, and P. subpulla. Detailed descriptions with illustrations are provided for these new species, and their relationships with similar or allied species are discussed. An identification key for all accepted 56 species with a synoptic description is provided, and two keys to neotropical and Asian Phylloporia species are also provided.










Similar content being viewed by others
References
Berkeley MJ (1856) Decades of fungi. Decades 54-55. Rio Negro fungi. Hooker’s J Bot Kew Gard Misc 8:169–177
Chen JJ, Cui BK, Zhou LW, Korhonen K, Dai YC (2015) Phylogeny, divergence time estimation, and biogeography of the genus Heterobasidion (Basidiomycota, Russulales). Fungal Divers 71:185–200. https://doi.org/10.1007/s13225-014-0317-2
Chen JJ, Cui BK, Dai YC (2016) Global diversity and molecular systematics of Wrightoporia s.l. (Russulales, Basidiomycota). Persoonia 37:21–36. https://doi.org/10.3767/003158516X689666
Chen YY, Zhu L, Xing JH, Cui BK (2017) Three new species of Phylloporia (Hymenochaetales) with dimitic hyphal systems from tropical China. Mycologia 109:951–964. https://doi.org/10.1080/00275514.2017.1410692
Cui BK, Yuan HS, Dai YC (2010) Two new species of Phylloporia (Basidiomycota, Hymenochaetaceae) from China. Mycotaxon 113:171–178. https://doi.org/10.5248/113.171
Dai YC (2010) Hymenochaetaceae (Basidiomycota) in China. Fungal Divers 45:131–343. https://doi.org/10.1007/s13225-010-0066-9
Dai YC (2012) Polypore diversity in China with an annotated checklist of Chinese polypores. Mycoscience 53:49–80. https://doi.org/10.1007/s10267-011-0134-3
Darriba D, Taboada GL, Doallo R, Posada D (2012) jModelTest 2: more models, new heuristics and parallel computing. Nat Methods 9:772. https://doi.org/10.1038/nmeth.2109
Decock C, Amalfi M, Robledo G, Castillo G (2013) Phylloporia nouraguensis, an undescribed species on Myrtaceae from French Guiana. Cryptogam Mycol 34:15–27. https://doi.org/10.7872/crym.v34.iss1.2013.15
Decock C, Yombiyeni P, Memiaghe H (2015) Hymenochaetaceae from the Guineo-Congolian rainforest: Phylloporia flabelliforma sp. nov. and Phylloporia gabonensis sp. nov., two undescribed species from Gabon. Cryptogam Mycol 36:449–467. https://doi.org/10.7872/crym/v36.iss4.2015.449
Ferreira-Lopes V, Robledo GL, Reck MA, Góes-Neto A, Drechsler-Santos ER (2016) Phylloporia spathulata sensu stricto and two new South American stipitate species of Phylloporia (Hymenochaetaceae). Phytotaxa 257(2):133–148. https://doi.org/10.11646/phytotaxa.257.2.3
Gafforov Y, Tomšovský M, Langer E, Zhou LW (2014) Phylloporia yuchengii sp. nov. (Hymenochaetales, Basidiomycota) from Western Tien Shan Mountains of Uzbekistan based on phylogeny and morphology. Cryptogam Mycol 35:313–322. https://doi.org/10.7872/crym.v35.iss4.2014.313
Gilbertson RL, Ryvarden L (1986–1987) North American polypores 1–2. Fungiflora, Oslo, pp 1–885
Góes-Neto A, Loguercio-Leite C, Guerrero RT (2005) DNA extraction from frozen field-collected and dehydrated herbarium fungal basidiomata: performance of SDS and CTAB-based methods. Biotemas 18(2):19–32
Guindon S, Gascuel O (2003) A simple, fast and accurate method to estimate large phylogenies by maximum-likelihood. Syst Biol 52:696–704. https://doi.org/10.1080/10635150390235520
Hall TA (1999) Bioedit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser 41:95–98. https://doi.org/10.1021/bk-1999-0734.ch008
Hattori T, Sakayaroj J, Jones EBG, Suetrong S, Preedanon S, Klaysuban A (2014) Three species of Fulvifomes (Basidiomycota, Hymenochaetales) associated with rots on mangrove tree Xylocarpus granatum in Thailand. Mycoscience 55:344–354. https://doi.org/10.1016/j.myc.2014.01.001
Ipulet P, Ryvarden L (2005) New and interesting polypores from Uganda. Synop Fung 20:87–99
Ji XH, He SH, Chen JJ, Si J, Wu F, Zhou LW, Vlasák J, Tian XM, Dai YC (2017a) Global diversity and phylogeny of Onnia (Hymenochaetaceae) species on gymnosperms. Mycologia 109:27–34. https://doi.org/10.1080/00275514.2016.1274619
Ji XH, Vlasák J, Zhou LW, Wu F, Dai YC (2017b) Phylogeny and diversity of Fomitiporella (Hymenochaetales, Basidiomycota). Mycologia 109:308–322. https://doi.org/10.1080/00275514.2017.1305943
Ji XH, Vlasák J, Tian XM, Dai YC (2018) Three new species of Fomitiporella (Hymenochaetales, Basidiomycota) based on the evidence from morphology and DNA sequence data. MycoKeys 30:73–89. https://doi.org/10.3897/mycokeys.30.23109
Larkin MA, Blackshields G, Brown NP, Chenna R, McGettigan PA, McWilliam H, Valentin F, Wallace IM, Wilm A, Lopez R, Thompson JD, Gibson TJ, Higgins DG (2007) Clustal W and Clustal X 2.0. Bioinformatics 23:2947–2948. https://doi.org/10.1093/bioinformatics/btm404
Montagne JPFC (1854) Cryptogamia Guyanensis seu plantarum cellularium in Guyana gallica annis 1835-1849 a cl. Leprieur collectarum enumeratio universalis. Ann Sci Nat Bot 1:91–144
Murrill WA (1904) The Polyporaceae of North America 9. Bull Torrey Bot Club 31:593–610. https://doi.org/10.2307/2478499
Núñez M, Ryvarden L (2000) East Asian polypores 1. Ganodermataceae and Hymenochaetaceae. Synop Fung 13:1–168.
Núñez M, Ryvarden L (2001) East Asian polypores 2. Synop Fung 14:165–522
Patouillard NT (1891) Contributions a la flore mycologique du Tonkin 2. J Bot (Morot) 5:306–321
Pattengale ND, Alipour M, Bininda-Emonds ORP, Moret BME, Stamatakis A (2010) How many bootstrap replicates are necessary? J Comput Biol 17:337–354. https://doi.org/10.1007/978-3-642-02008-7_13
Petersen JH (1996) Farvekort. The Danish Mycological Society’s colour-chart. Foreningen til Svampekundskabens Fremme, Greve, pp 1–6
Qin WM, Wang XW, Sawahata T, Zhou LW (2018) Phylloporia lonicerae (Hymenochaetales, Basidiomycota), a new species on Lonicera japonica from Japan and an identification key to worldwide species of Phylloporia. MycoKeys 30:17–30. https://doi.org/10.3897/mycokeys.30.23235
Ren GJ, Wu F (2017) Phylloporia lespedezae sp. nov. (Hymenochaetaceae, Basidiomycota) from China. Phytotaxa 299(2):243–251. https://doi.org/10.11646/phytotaxa.299.2.8
Ryvarden L (1972) A critical checklist of the Polyporaceae in tropical East Africa. Nor J Bot 19:229–238
Ryvarden L (1982) Type studies in the Polyporaceae 11. Species described by J.F. C. Montagne, either alone or with other authors. Nor J Bot 2:75–84
Ryvarden L (1983) Type studies in the Polyporaceae 14. Species described by Patouillard, either alone or with other authors. Occ Pap Farlow Herb Crypt Bot 18:1–39
Ryvarden L (1984) Type studies in the Polyporaceae 16. Species described by J.M. Berkeley, either alone or with other mycologists from 1856 to 1886. Mycotaxon 20:329–363
Ryvarden L (1991) Genera of polypores. Nomenclature and taxonomy. Synop Fung 5:1–363
Ryvarden L, Gilbertson RL (1993) European polypores 1. Synop Fung 6:1–387
Ryvarden L, Gilbertson RL (1994) European polypores 2. Synop Fung 7:388–743
Ryvarden L, Johansen I (1980) A preliminary polypore flora of East Africa. Fungiflora, Oslo, pp 1–636
Silvestro D, Michalak I (2012) raxmlGUI: a graphical front-end for RAxML. Org Divers Evol 12:335–337. https://doi.org/10.1007/s13127-011-0056-0
Stamatakis A (2006) RAxML-VI-HPC: maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed models. Bioinformatics 22:2688–2690. https://doi.org/10.1093/bioinformatics/btl446
Swofford DL (2002) PAUP*: Phylogenetic analysis using parsimony (*and other methods). Version 4.0b10. Sinauer Associates, Sunderland, Massachusetts
Valenzuela R, Raymundo T, Cifuentes J, Castillo G, Amalfi M, Decock C (2011) Two undescribed species of Phylloporia from Mexico based on morphological and phylogenetic evidence. Mycol Prog 10:341–349. https://doi.org/10.1007/s11557-010-0707-0
Vilgalys R, Hester M (1990) Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J Bacteriol 172:4238–4246. https://doi.org/10.1128/jb.172.8.4238-4246.1990
Wagner T, Ryvarden L (2002) Phylogeny and taxonomy of the genus Phylloporia (Hymenochaetales). Mycol Prog 1:105–106. https://doi.org/10.11646/1007/s11557-006-0009-8
Weinmann JA (1828) Enumeratio Fungorum agro pawlowskiensi praeecipue crescentium. Syll Pl Nov Ratisb 2:99–118
White TJ, Bruns T, Lee S, Taylor JW (1990) Amplification and direct sequencing of ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR Protocols, a Guide to Methods and Applications. Academic Press, New York, pp 315–322
Yombiyeni P, Decock C (2017) Hymenochaetaceae (Hymenochaetales) from the Guineo-Congolian phytochorion: Phylloporia littoralis sp. nov. from coastal vegetation in Gabon, with an identification key to the local species. Plant Ecol Evol 150:160–172. https://doi.org/10.5091/plecevo.2017.1289
Yombiyeni P, Balezi A, Amalfi M, Decock C (2015) Hymenochaetaceae from the Guineo-Congolian rainforest: three new species of Phylloporia based on morphological, DNA sequences and ecological data. Mycologia 107:996–1011. https://doi.org/10.3852/14-298
Zhou LW (2013) Phylloporia tiliae sp. nov. from China. Mycotaxon 124:361–365. https://doi.org/10.5248/124.361
Zhou LW (2014a) Fomitiporella caviphila sp. nova (Hymenochaetaceae Basidiomycota) from Eastern China, with a preliminary discussion on the taxonomy of Fomitiporella. Ann Bot Fenn 51:279–284. https://doi.org/10.5735/085.051.0503
Zhou LW (2014b) Fulvifomes hainanensis sp. nov. and F. indicus comb. nov. (Hymenochaetales, Basidiomycota) evidenced by a combination of morphology and phylogeny. Mycoscience 55:70–77. https://doi.org/10.1016/j.myc.2013.05.006
Zhou LW (2015a) Four new species of Phylloporia (Hymenochaetales, Basidiomycota) from tropical China with a key to Phylloporia species worldwide. Mycologia 107:1184–1192. https://doi.org/10.3852/14-254
Zhou LW (2015b) Phylloporia osmanthi and P. terrestris spp. nov. (Hymenochaetales, Basidiomycota) from Guangxi, South China. Nova Hedwigia 100:239–249. https://doi.org/10.1127/nova_hedwigia/2014/0220
Zhou LW (2016) Phylloporia minutipora and P. radiata spp. nov. (Hymenochaetales, Basidiomycota) from China and a key to worldwide species of Phylloporia. Mycol Prog 15:57. https://doi.org/10.1007/s11557-016-1200-1
Zhou LW, Dai YC (2012) Phylogeny and taxonomy of Phylloporia (Hymenochaetales): new species and a worldwide key to the genus. Mycologia 104:211–222. https://doi.org/10.3852/11-093
Acknowledgments
We are grateful to Prof. Teuvo Ahti (Helsinki, Finland) who helped us to translate some literature and to Renata Chikowski (Recife, Brazil) for the drawings of the Brazilian specimens.
Funding
The research is financed by the National Natural Science Foundation of China (Project No. 31530002), Conselho Nacional de Desenvolvimento Científico e Tecnológico (PQ 1D 307601/2015-3), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES/SIU 008/13), and Fundação de Amparo à Ciência e Tecnologia de Pernambuco (APQ 0375-2.03/15). LWZ is financially supported by the Youth Innovation Promotion Association CAS (No. 2017240).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Section Editor: Marc Stadler
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wu, F., Ren, GJ., Wang, L. et al. An updated phylogeny and diversity of Phylloporia (Hymenochaetales): eight new species and keys to species of the genus. Mycol Progress 18, 615–639 (2019). https://doi.org/10.1007/s11557-019-01476-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11557-019-01476-4