Abstract
Purpose
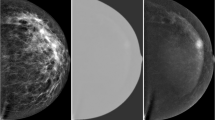
Contrast-enhanced spectral mammography (CESM) is a recently developed breast imaging technique. CESM relies on dual-energy acquisition following contrast agent injection to improve mammography sensitivity. CESM is comparable to contrast-enhanced MRI in terms of sensitivity, at a fraction of the cost. However, since lesion variability is large, even with the improved visibility provided by CESM, differentiation between benign and malignant enhancement is not accurate and a biopsy is usually performed for final assessment. Breast biopsies can be stressful to the patient and are expensive to healthcare systems. Moreover, as the biopsies results are most of the time benign, a specificity improvement in the radiologist diagnosis is required. This work presents a deep learning-based decision support system, which aims at improving the specificity of breast cancer diagnosis by CESM without affecting sensitivity.
Methods
We compare two analysis approaches, fine-tuning a pretrained network and fully training a convolutional neural network, for classification of CESM breast mass as benign or malignant. Breast Imaging Reporting and Data Systems (BIRADS) is a radiological lexicon, used with breast images, to categorize lesions. We improve each classification network by incorporating BIRADS textual features as an additional input to the network. We evaluate two ways of BIRADS fusion as network input: feature fusion and decision fusion. This leads to multimodal network architectures. At classification, we also exploit information from apparently normal breast tissue in the CESM of the considered patient, leading to a patient-specific classification.
Results
We evaluate performance using fivefold cross-validation, on 129 randomly selected breast lesions annotated by an experienced radiologist. Each annotation includes a contour of the mass in the image, biopsy-proven label of benign or malignant lesion and BIRADS descriptors. At 100% sensitivity, specificity of 66% was achieved using a multimodal network, which combines inputs at feature level and patient-specific classification.
Conclusions
The presented multimodal network may significantly reduce benign biopsies, without compromising sensitivity.






Similar content being viewed by others
References
Carney PA, Miglioretti DL, Yankaskas BC, Kerlikowske K, Rosenberg R, Rutter CM, Geller BM, Abraham LA, Taplin SH, Dignan M, Cutter G (2003) Individual and combined effects of age, breast density, and hormone replacement therapy use on the accuracy of screening mammography. Ann Intern Med 138(3):168–75
Drukteinis JS, Mooney BP, Flowers CI, Gatenby RA (2013) Beyond mammography: new frontiers in breast cancer screening. Am J Med 126(6):472–479
Lobbes MBI, Smidt ML, Houwers J, Tjan-Heijnen VC, Wildberger JE (2013) Contrast enhanced mammography: techniques, current results, and potential indications. Clin Radiol 68(9):935–944
Li L, Roth R, Germaine P, Ren S, Lee M, Hunter K, Tinney E, Liao L (2017) Contrast-enhanced spectral mammography (CESM) versus breast magnetic resonance imaging (MRI): a retrospective comparison in 66 breast lesions. Diagn Interv Imaging 98:113123
Fallenberg EM, Dromain C, Diekmann F, Engelken F, Krohn M, Singh JM, Ingold-Heppner B, Winzer KJ, Bick U, Renz AD (2014) Contrast-enhanced spectral mammography versus MRI: initial results in the detection of breast cancer and assessment of tumour size. Eur Radiol 24(1):256–264
Luczyska E, Heinze-Paluchowska S, Dyczek S, Blecharz P, Rys J, Reinfuss M (2014) Contrast-enhanced spectral mammography: comparison with conventional mammography and histopathology in 152 women. Korean J Radiol 15(6):689–696
Brake GM, Karssemeijer N, Hendriks JH (2000) An automatic method to discriminate malignant masses from normal tissue in digital mammograms1. Phys Med Biol 45(10):2843
Sampat MP, Markey MK, Bovik AC (2005) Computer-aided detection and diagnosis in mammography. Handb Image Video Process 2(1):1195–1217
Vasantha M, Bharathi DVS, Dhamodharan R (2010) Medical image feature, extraction, selection and classification. IJEST 1(2):2071–2076
Jalalian A, Mashohor SB, Mahmud HR, Saripan MIB, Ramli ARB, Karasfi B (2013) Computer-aided detection/diagnosis of breast cancer in mammography and ultrasound: a review. Clin Imaging 37(3):420–426
Mateos MJ, Gastelum A, Mrquez J, Brandan ME (2016) Texture analysis of contrast-enhanced digital mammography (CEDM) images. International Workshop on Digital Mammography. LNCS 9699, Springer, pp 585–592
Cheung YC, Tsai HP, Lo YF, Ueng SH, Huang PC, Chen SC (2016) Clinical utility of dual-energy contrast-enhanced spectral mammography for breast microcalcifications without associated mass: a preliminary analysis. Eur Radiol 26(4):1082–1089
Patel BK, Ranjbar S, Wu T, Pockaj BA, Li J, Zhang N, Lobbes M, Zhang B, Mitchell JR (2018) Computer-aided diagnosis of contrast-enhanced spectral mammography: a feasibility study. Eur J Radiol 98:207–213
Pudil P, Novoviov J, Kittler J (1994) Floating search methods in feature selection. Pattern Recognit Lett 15(11):1119–1125
Cortes C, Vapnik V (1995) Support-vector networks. Mach Learn 20(3):273–297
American College of Radiology, ACR BI-RADS Atlas 5th Edition, 125–143 (2013)
Knogler T, Homolka P, Hoernig M, Leithner R, Langs G, Waitzbauer M, Pinker-Domenig K, Leitner S, Helbich TH (2017) Application of BI-RADS descriptors in contrast-enhanced dual-energy mammography: comparison with MRI. Breast Care 12:212–216
Ngiam J, Khosla A, Kim M, Nam J, Lee H, Ng AY (2011) Multimodal deep learning. In: Proceedings of the 28th ICML
Wu D, Pigou L (2016) Deep dynamic neural networks for multimodal gesture segmentation and recognition. IEEE Trans Pattern Anal Mach Intell 38(8):1583–1597
Cao Y, Steffey S, He J, Xiao D, Tao C, Chen P, Mller H (2014) Medical image retrieval: a multimodal approach. Cancer Inf 13:CIN-S14053
Krizhevsky A, Sutskever I, Hinton GE (2012) Imagenet classification with deep convolutional neural networks. NIPS 25:1097–1105
Tajbakhsh N, Shin JY, Gurudu SR, Hurst RT, Kendall CB, Gotway MB, Liang J (2016) Convolutional neural networks for medical image analysis: Full training or fine tuning? IEEE TMI 35(5):1299–1312
Karpathy A, Toderici G, Shetty S, Leung T, Sukthankar R, Fei-Fei L (2014) Large-scale video classification with convolutional neural networks. In: IEEE conference CVPR, pp 1725–1732
Ioffe S, Szegedy C (2015) Batch normalization: accelerating deep network training by reducing internal covariate shift. In: ICML, pp 448–456
Kingma D, Adam JB (2014) A method for stochastic optimization. arXiv preprint arXiv:1412.6980
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
For this type of study formal consent is not required.
Rights and permissions
About this article
Cite this article
Perek, S., Kiryati, N., Zimmerman-Moreno, G. et al. Classification of contrast-enhanced spectral mammography (CESM) images. Int J CARS 14, 249–257 (2019). https://doi.org/10.1007/s11548-018-1876-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11548-018-1876-6