Abstract
Purpose
Percutaneous spinal needle insertion procedures often require proper identification of the vertebral level to effectively and safely deliver analgesic agents. The current clinical method involves “blind” identification of the vertebral level through manual palpation of the spine, which has only 30% reported accuracy. Therefore, there is a need for better anatomical identification prior to needle insertion.
Methods
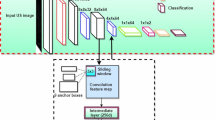
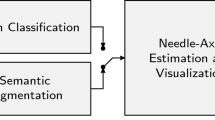
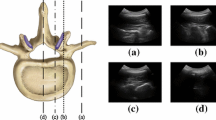
A real-time system was developed to identify the vertebral level from a sequence of ultrasound images, following a clinical imaging protocol. The system uses a deep convolutional neural network (CNN) to classify transverse images of the lower spine. Several existing CNN architectures were implemented, utilizing transfer learning, and compared for adequacy in a real-time system. In the system, the CNN output is processed, using a novel state machine, to automatically identify vertebral levels as the transducer moves up the spine. Additionally, a graphical display was developed and integrated within 3D Slicer. Finally, an augmented reality display, projecting the level onto the patient’s back, was also designed. A small feasibility study \((n=20)\) evaluated performance.
Results
The proposed CNN successfully discriminates ultrasound images of the sacrum, intervertebral gaps, and vertebral bones, achieving 88% 20-fold cross-validation accuracy. Seventeen of 20 test ultrasound scans had successful identification of all vertebral levels, processed at real-time speed (40 frames/s).
Conclusion
A machine learning system is presented that successfully identifies lumbar vertebral levels. The small study on human subjects demonstrated real-time performance. A projection-based augmented reality display was used to show the vertebral level directly on the subject adjacent to the puncture site.







Similar content being viewed by others
References
Brinkmann S, Germain G, Sawka A, Tang R, Vaghadia H (2013) Is there a place for ultrasound in neuraxial anesthesia? Imaging Med 5(2):177–186
Chen H, Ni D, Qin J, Li S, Yang X, Wang T, Heng PA (2015) Standard plane localization in fetal ultrasound via domain transferred deep neural networks. IEEE J Biomed Health Inform 19(5):1627–1636
Ecimovic P, Loughrey J (2010) Ultrasound in obstetric anesthesia: a review of current applications. Int. J Obstet. Anesth 19(3):320–326
Fedorov A, Beichel R, Kalpathy-Cramer J, Finet J, Fillion-Robin JC, Pujol S, Bauer C, Jennings D, Fennessy F, Sonka M, Buatti J, Aylward S, Miller J, Pieper S, Kikinis R (2012) 3D slicer as an image computing platform for the quantitative imaging network. Magn Reson Imaging 30(9):1323–1341
Franklin AD, Hughes EM (2016) Fluoroscopically guided tunneled trans-caudal epidural catheter technique for opioid-free neonatal epidural analgesia. J Anesth 30(3):493–497
Furness G, Reilly M, Kuchi S (2002) An evaluation of ultrasound imaging for identification of lumbar intervertebral level. Anaesthesia 57(3):277–280
Gilad I, Nissan M (1986) A study of vertebra and disc geometric relations of the human cervical and lumbar spine. Spine 11(2):154–157
Goldstein A, Madrazo BL (1981) Slice-thickness artifacts in gray-scale ultrasound. J Clin Ultrasound 9(7):365–375
He K, Zhang X, Ren S, Sun J (2015) Deep residual learning for image recognition. arXiv preprint arXiv:1512.03385
Hetherington J, Pesteie M, Lessoway V, Abolmaesumi P, Rohling R (2017) Identification and tracking of vertebrae in ultrasound using deep networks with unsupervised feature learning. In: SPIE Medical Imaging. International Society for Optics and Photonics
Iandola FN, Moskewicz MW, Ashraf K, Han S, Dally WJ, Keutzer K (2016) Squeezenet: Alexnet-level accuracy with 50x fewer parameters and \(<\)1 mb model size. arXiv preprint arXiv:1602.07360
Jia Y, Shelhamer E, Donahue J, Karayev S, Long J, Girshick R, Guadarrama S, Darrell T (2014) Caffe: convolutional architecture for fast feature embedding. arXiv preprint arXiv:1408.5093
Kerby B, Rohling R, Nair V, Abolmaesumi P (2008) Automatic identification of lumbar level with ultrasound. In: 2008 30th Annual international conference of the IEEE engineering in medicine and biology society, pp 2980–2983. IEEE
Kong B, Zhan Y, Shin M, Denny T, Zhang S (2016) Recognizing end-diastole and end-systole frames via deep temporal regression network. In: International conference on medical image computing and computer-assisted intervention, pp 264–272. Springer
Krizhevsky A, Sutskever I, Hinton GE (2012) Imagenet classification with deep convolutional neural networks. In: Pereira F, Burges CJC, Bottou L Weinberger KQ (eds) Advances in neural information processing systems, Curran Associates, Inc. pp 1097–1105. http://papers.nips.cc/paper/4824-imagenet-classification-with-deep-convolutional-neural-networks.pdf
Kwitt R, Vasconcelos N, Razzaque S, Aylward S (2013) Localizing target structures in ultrasound video-a phantom study. Med Image Anal 17(7):712–722
Lasso A, Heffter T, Rankin A, Pinter C, Ungi T, Fichtinger G (2014) Plus: open-source toolkit for ultrasound-guided intervention systems. IEEE Trans Biomed Eng 61(10):2527–2537
LeCun Y, Boser B, Denker JS, Henderson D, Howard RE, Hubbard W, Jackel LD (1989) Backpropagation applied to handwritten zip code recognition. Neural Comput 1(4):541–551
Oquab M, Bottou L, Laptev I, Sivic J (2014) Learning and transferring mid-level image representations using convolutional neural networks. In: Proceedings of the IEEE conference on computer vision and pattern recognition, pp 1717–1724
Rafii-Tari H, Lessoway VA, Kamani AA, Abolmaesumi P, Rohling R (2015) Panorama ultrasound for navigation and guidance of epidural anesthesia. Ultrasound Med Biol 41(8):2220–2231
Schlotterbeck H, Schaeffer R, Dow W, Touret Y, Bailey S, Diemunsch P (2008) Ultrasonographic control of the puncture level for lumbar neuraxial block in obstetric anaesthesia. Br J Anaesth 100(2):230–234
Soni NJ, Franco-Sadud R, Schnobrich D, Dancel R, Tierney DM, Salame G, Restrepo MI, McHardy P (2016) Ultrasound guidance for lumbar puncture. Neurol Clin Pract 6(4):358–368
Szegedy C, Liu W, Jia Y, Sermanet P, Reed S, Anguelov D, Erhan D, Vanhoucke V, Rabinovich A (2015) Going deeper with convolutions. In: Proceedings of the IEEE conference on computer vision and pattern recognition, pp 1–9
Tran D, Kamani AA, Lessoway VA, Peterson C, Hor KW, Rohling RN (2009) Preinsertion paramedian ultrasound guidance for epidural anesthesia. Anesth Analg 109(2):661–667
Tran DQH, González AP, Bernucci F, Finlayson RJ (2015) Confirmation of loss-of-resistance for epidural analgesia. Reg Anesth Pain Med 40(2):166–173
Ungi T, Lasso A, Fichtinger G (2015) Tracked ultrasound in navigated spine interventions. In: Li S, Yao J (eds) Spinal imaging and image analysis. Springer, pp 469–494. doi:10.1007/978-3-319-12508-4_15
Yu S, Tan KK (2014) Classification of lumbar ultrasound images with machine learning. In: Asia-Pacific conference on simulated evolution and learning, pp 287–298. Springer
Yu S, Tan KK, Sng BL, Li S, Sia ATH (2014) Automatic identification of needle insertion site in epidural anesthesia with a cascading classifier. Ultrasound Med Biol 40(9):1980–1990
Yu S, Tan KK, Sng BL, Li S, Sia ATH (2015) Lumbar ultrasound image feature extraction and classification with support vector machine. Ultrasound Med Biol 41(10):2677–2689
Yu S, Tan KK, Sng BL, Li S, Sia ATH (2015) Real-time automatic spinal level identification with ultrasound image processing. In: 2015 IEEE 12th international symposium on biomedical imaging, pp 243–246. IEEE
Zeiler MD, Fergus R (2014) Visualizing and understanding convolutional networks. In: European conference on computer vision, pp 818–833. Springer
Acknowledgements
The authors would like to thank Mehran Pesteie for his help in data collection.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by the Natural Sciences and Engineering Research Council of Canada (Grant Number RGPIN-2-15-03993), and the Canadian Institutes of Health Research (Grant Number MOP-125935).
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Hetherington, J., Lessoway, V., Gunka, V. et al. SLIDE: automatic spine level identification system using a deep convolutional neural network. Int J CARS 12, 1189–1198 (2017). https://doi.org/10.1007/s11548-017-1575-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11548-017-1575-8