Abstract
Purpose
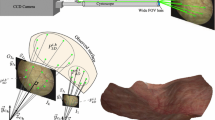
Organ-level registration is critical to image-guided therapy in soft tissue. This is especially important in organs such as the kidney which can freely move. We have developed a method for registration that combines three-dimensional locations from a holographic conoscope with an endoscopically obtained textured surface. By combining these data sources clear decisions as to the tissue from which the points arise can be made.
Methods
By localizing the conoscope’s laser dot in the endoscopic space, we register the textured surface to the cloud of conoscopic points. This allows the cloud of points to be filtered for only those arising from the kidney surface. Once a valid cloud is obtained we can use standard surface registration techniques to perform the image-space to physical-space registration. Since our methods use two distinct data sources we test for spatial accuracy and characterize temporal effects in phantoms, ex vivo porcine and human kidneys. In addition we use an industrial robot to provide controlled motion and positioning for characterizing temporal effects.
Results
Our initial surface acquisitions are hand-held. This means that we take approximately 55 s to acquire a surface. At that rate we see no temporal effects due to acquisition synchronization or probe speed. Our surface registrations were able to find applied targets with submillimeter target registration errors.
Conclusion
The results showed that the textured surfaces could be reconstructed with submillimetric mean registration errors. While this paper focuses on kidney applications, this method could be applied to any anatomical structures where a line of sight can be created via open or minimally invasive surgical techniques.













Similar content being viewed by others
References
Drucker BJ (2005) Renal cell carcinoma: current status and future prospects. Cancer Treat Rev 3:536–545. doi:10.1016/j.ctrv.2005.07.009
Patel SG, Penson DF, Clark PE, Cookson MS, Chang SS, Herrell SD, Smith JA, Borocase DA (2012) National trends in the use of partial nephrectomy: a rising tide that has not lifted all boats. J Urol 287(3):816–821. doi:10.1016/j.juro.2011.10.173
Cooperberg MR, Mallin K, Kane CJ, Carroll PR (2011) Treatment trends for stage 1 renal cell carcinoma. J Urol 186:394–399. doi:10.1016/j.juro.2011.03.130
Novick AC (2004) Laparoscopic and partial nephrectomy. Clin Cancer Res 10:6322S. doi:10.1158/1078-0432.CCR-050003
Orvieto M, Chien GW, Tolhurst SR, Rapp DE, Steinberg GD, Mikhail AA, Brendler CB, Shalhav AL (2005) Simplifying laparoscopic partial nephrectomy: technical considerations for reproducible outcomes. Adult Urol 66:976–980. doi:10.1016/j.urology.2005.05.013
Fergany AF, Hafez KC, Novick AC (2000) Long-term results of nephron sparing surgery for localized renal cell carcinoma: 10-year followup. J Urol 163:442–445. doi:10.1016/S0022-5347(05)67896-2
Godley PA, Ataga KI (2000) Renal cell carcinoma. Curr Opin Oncol 12:260–264
Vogelzang NJ, Stadler WM (1998) Kidney cancer. Lancet 352:1691–1696. doi:10.1016/S0140-6736(98)01041-1
Nosnik IP, Mouraviev V, Nelson R, Polascik TJ (2006) Multiple nephron-sparing procedures in solitary kidney with recurrent, metachronous, nonfamilial renal cell carcinoma. Urology 68(6):1343. doi:10.1016/j.urology.2006.09.004
Lau WK, Blute ML, Weaver AL, Torres VE, Zincke H (2000) Matched comparison of radical nephrectomy vs nephron-sparing surgery in patients with unilateral renal cell carcinoma and a normal contralateral kidney. Mayo Clin Proc 75:1236–1242
Becker F, Van Poppel H, Hakenberg OW, Stief C, Gill I, Guazzoni G, Montorsi F, Russo P, Stöckle M (2009) Assessing the impact of ischaemia time during partial nephrectomy. Eur Urol 56(4):625–634. doi:10.1016/j.eururo.2009.07.016
Pomara G, Campo G, Francesca F (2009) Intraoperative and postoperative complications of nephron sparing surgery: prevention and possible treatments. Arch Ital Urol Androl 81(2):80–85
Kercher KW, Heniford PT, Matthews BD, Smith TI, Lincourt AE, Hayes DH, Eskind LB, Irby PB, Teigland CM (2003) Laparoscopic versus open nephrectomy in 210 consecutive patients: outcomes, cost, and changes in practice patterns. surg endosc 17(12):1889–1895
Negrete-Pulido O, Molina-Torres M, Castaño-Tostado E, Loske AM, Gutiérrez-Aceves J (2010) Percutaneous renal access: the learning curve of a simplified approach. J Endourol 24(3):457–460. doi:10.1089/end.2009.0210
Ong R, Glisson C, Altamar H, Viprakasit D, Clark P, Herrell SD, Galloway RL (2010) Intraprocedural registration for image-guided kidney surgery. IEEE Trans Mechatron 15(6):847–852. doi:10.1109/TMECH.2010.2066985
Benincasa AB, Clements LW, Herrell SD, Galloway RL (2008) Feasibility study for image-guided kidney surgery: assessment of required intraoperative surface for accurate physical to image space registrations. Med Phys 35:4251–4261. doi:10.1118/1.2969064
Puerto-Souza GA, Mariottini GL (2013) A fast and accurate feature-matching algorithm for minimally invasive endoscopic images. IEEE-TMI 32(7):1201–1214. doi:10.1109/TMI.2013.223930
Glisson CL, Ong R, Simpson AL, Burgner J, Lathrop RA, Webster RJ, Galloway RL (2011) Registration methods for gross motion correction during image-guided kidney surgery. Comput Assist Radiol Surg 6:S159–S163. doi:10.1109/TBME.2012.2215033
Cash DM, Sinha TK, Chapman WC, Terawaki H, Dawant BM, Galloway RL, Miga MI (2005) Incorporation of a laser range scanner into image-guided liver surgery: surface acquisition, registration, and tracking. Med Phys 30:1671–1682. doi:10.1118/1.1578911
Stoyanov D, Darzi A, Yang GZ (2005) A practical approach towards accurate dense 3D depth recovery for robotic laparoscopic surgery. Comput Aided Surg 10(4):199–208. doi:10.1080/10929080500230379
Langø T, Tangen GA, Mårvik R, Ystgaard B, Yavuz Y, Kaspersen JH, Solberg OV, Hernes TAN (2008) Navigation in laparoscopy–prototype research platform for improved image-guided surgery. Minim Invasive Ther Allied Technol 17(1):17–33. doi:10.1080/13645700701797879
Mersmann S, Müller M, Seitel A, Arnegger F, Tetzlaff R, Dinkel J, Baumhauer M, Schmied B, Meinzer H-P, Maier-Hein L (2011) Time-of-flight camera technique for augmented reality in computer-assisted interventions. In: Proceeding of SPIE 7964, medical imaging 2011: visualization. Image-guided procedures, and modeling. doi:10.1117/12.878149
Maier-Hein L, Groch A, Bartoli A, Bodenstedt S, Guillaume B, Chang P, Clancy N, Elson ND, Haase S (2014) Comparative validation of single-shot optical techniques for laparoscopic 3D surface reconstruction. IEEE-TMI 33(10):1913–1930. doi:10.1109/TMI.2014.2325607
Ding S, Miga MI, Noble JH, Cao A, Dumpuri P, Thompson RC, Dawant BM (2009) Semiautomatic registration of pre- and post brain tumor resection laser range data: method and validation. IEEE Trans Biomed Eng 56:770–780. doi:10.1109/TBME.2008.2006758
Dumpuri P, Clements LW, Dawant BM, Miga MI (2010) Model-updated image-guided liver surgery: preliminary results using surface characterization. Prog Biophys Mol Biol 103:197–207. doi:10.1016/j.pbiomolbio.2010.09.014
Clements LW, Chapman WC, Dawant BM, Galloway RL, Miga MI (2008) Robust surface registration using salient anatomical features for image-guided liver surgery: algorithm and validation. Med Phys 35:2528–2540. doi:10.1118/1.2911920
Li P, Wang W, Song Z, An Y, Zhang C (2014) A framework for correcting brain retraction based on an eXtended Finite Element Method using a laser range scanner. Int J CARS 9:669–681. doi:10.1007/s1548-013-0958-8
Sirat GY (1992) Conoscopic holography. 1. Basic principles and physical basis. J Opt Soc Am Opt Image Sci Vis 9:70–83. doi:10.1364/JOSAA.9.000070
Sirat GY, Psaltis D (1988) Conoscopic holograms. Opt Commun 65:243–249. doi:10.1016/0030-4018(88)90160-5
Lathrop RA, Hackworth D, Webster RJ (2010) Minimally invasive holographic surface scanning for soft tissue image registration. IEEE Trans Biomed Eng 57(6):1497–1506. doi:10.1109/TBME.2010.2040736
Burgner J, Simpson AL, Fitzpatrick JM, Lathrop RA, Herrell SD, Miga MI, Webster RJ (2012) A study on the theoretical and practical accuracy of conoscopic holography-based surface measurements: toward image registration in minimally invasive surgery. Int J Med Robot 9(2):190–203. doi:10.1002/rcs.1446
Simpson AL, Burgner J, Glisson CL, Herrell SD, Ma B, Pheiffer TS, Webster RJ, Miga MI (2013) Comparison study of intraoperative surface acquisition methods for surgical navigation. IEEE Trans Biomed Eng 60:1090–1099
Glisson C, Ong R, Clark P, Herrell D, Galloway R (2011) The use of virtual fiducials in image-guided kidney surgery. In: Proceedings of SPIE 7964, medical imaging 2011: visualization. Image-guided procedures, and modeling. doi:10.1117/12.877092
Danilchenko A (2011) Fiducial-based registration with anisotropic localization error. Doctoral dissertation, Vanderbilt University
Szpala S, Wierzbicki M, Guiraudon G, Peters TM (2005) Real-time fusion of endoscopic views with dynamic 3-D cardiac images: a phantom study. IEEE Trans Med Imaging 24:1207–1215. doi:10.1109/TMI.2005.853639
Sielhorst T, Sa W, Khamene A, Sauer F, Navab N (2007) Measurement of absolute latency for video see through augmented reality. In: Proceedings of the 2007 6th IEEE and ACM internationalsymposium on mixed and augmented reality. pp 1–4, 2007. doi:10.1109/ISMAR.2007.4538850
https://msdn.microsoft.com/en-us/library/windows/desktop/dd757629(v=vs.85).aspx
Acknowledgments
This work is funded in part by the National Institutes of Health: Grant R01 CA162477 from the National Cancer Institute, R01 NS049251 of the National Institute for Neurological Disorders and Stroke, and R44 DK081240 National Institute of Diabetes and Digestive and Kidney Diseases.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Rowena Ong, Courtenay Glisson, Jessica Burgner-Kahrs, Amber Simpson, Andrei Danilchenko, Ray Lathrop, Duke Herrell, Robert Webster III, Michael Miga, and Robert L. Galloway declare that they have no conflict of interest.
Human and animal participants
The work used ex vivo kidneys obtained post-euthanasia from animals killed under IACUC-approved protocols. No human data were used.
Rights and permissions
About this article
Cite this article
Ong, R., Glisson, C.L., Burgner-Kahrs, J. et al. A novel method for texture-mapping conoscopic surfaces for minimally invasive image-guided kidney surgery. Int J CARS 11, 1515–1526 (2016). https://doi.org/10.1007/s11548-015-1339-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11548-015-1339-2