Abstract
Purpose
Glucocorticoids (GCs) and external radiotherapy (RT) are used for treating moderate-to-severe Graves’ orbitopathy (GO). We aimed to assess whether GCs and RT were more effective when administered concomitantly or sequentially.
Methods
We retrospectively analyzed clinical outcomes [assessed by Clinical Activity Score (CAS) and NOSPECS classification] in 73 patients treated with both i.v. GCs and RT. The patients were divided in two groups: In group A (53 patients), RT was delivered concomitantly with GCs, and in group B (20 patients) RT was administered subsequently to the end of methylprednisolone.
Results
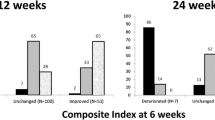
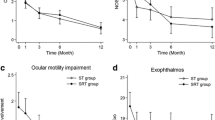
At baseline, CAS (median 4.0) and the percentage of patients encompassing the various grades of the classes 2, 3 and 4 of the NOSPECS score were similar in both groups. Six months after RT, CAS decreased to 2 in both groups (p = 0.0003 vs baseline) as well as NOSPECS class 4 (p < 0.0001 vs baseline). NOSPECS class 2 improved more in group A than in group B (p = 0.016). The median cumulative dose of GCs was lower in group A than in group B (median 4.500 vs 6000 mg, p < 0.007); the overall length of therapy was shorter in group A than in group B (68 vs 106 days, p < 0,02). The most common acute adverse effect was transient conjunctivitis (five in group A and three in group B); seven patients (five in group A and two in group B, age between 60 and 66 years) developed cataract, requiring surgery in five cases.
Conclusions
Concomitant administration of GC and RT showed a favorable effect in moderate-to-severe GO, thus suggesting that RT should be carried out early during steroid therapy, when clinical symptoms do not improve or deteriorate after the first i.v. administrations of GCs.


Similar content being viewed by others
References
Bartalena L, Tanda ML (2009) Cinical practice. Graves’ Orbitopathy. New Engl J Med 360:994–1001
Bartalena L, Baldeschi L, Boboridis K, Eckstein A, Kahaly GJ, Marcocci C, Perros P, Salvi M, Wiersinga WM (2016) The 2016 European Thyroid Association/European Group on Graves’ orbitopathy guidelines for the management of graves’ orbitopathy. Eur Thyroid J 5:9–26
Bartalena L, Pinchera A, Marcocci C (2000) Management of Graves’ ophthalmopathy: reality and perspectives. Endocr Rev 21:168–199
Prummel MF, Mourits MP, Blank L, Berghout A, Koornneef L, Wiersing WM (1993) Randomized double-blind trial of prednisone versus radiotherapy in Graves’ ophthalmopathy. Lancet 342:949–954
Mourits MP, Van Kempen-Hartevel ML, García MBG, Koppeschaar HP, Tick L, Terwee CB (2000) Radiotherapy for Graves’ orbitopathy: randomized placebo-controlled study. Lancet 355:1505–1509
Marquez SD, Lum BL, McDougall IR, Katkuri S, Levin PS, MacManus M, Donaldson SS (2001) Long-term results of irradiation for patients with progressive Graves’ ophthalmopathy. Int J Radiat Oncol Biol Phys 51:766–774
Matthiesen C, Thompsom JS, Thompson D, Farris B, Wilkes B, Ahmad S, Herman T, Bogardus C (2012) The efficacy of radiation therapy in the treatment of graves’ orbitopathy. Int J Radiat Oncol Biol Phys 82:117–123
Prabhu RS, Liebman L, Wojno T, Hall WA, Crocker I (2012) Clinical outcomes of radiotherapy as imitial local therapy for Graves’ ophtalmopathy and predictors of the need for post-radiotherapy decompressive surgery. Radiat Oncol 7:95–102
Tsujino K, Hirota S, Hagiwara M, Fukada S, Takada Y, Hishikawa Y, Kono M, Abe M (2000) Clinical outcomes of orbital irradiation combined with or without systemic high-dose or pulsed corticosteroids for Graves’. Ophtalmopathy 48:857–864
Ohtsuka K, Sato A, Kawaguchi S, Hashimoto M, Suzuki Y (2003) Effect of steroid pulse therapy with and without orbital radiotherapy on Graves’ ophthalmopathy. Am J Ophthalmol 135:285–290
Marcocci C, Bartalena L, Tanda ML, Manetti L, Dell’Unto E, Rocchi R, Barbesino G, Mazzi B, Bartolomei MP, Lepri P, Cartei F, Nardi M, Pinchera A (2001) Comparison of the effectiveness and tolerability of intravenous or oral glucocorticoids associated with orbital radiotherapy in the management of severe Graves’ ophthalmopathy: results of a prospective, single-blind, randomized study. J Clin Endocrinol Metab 86:3562–3567
Abboud M, Arabi A, Salti I, Geara F (2011) Outcome of thyroid associated ophtalmopathy treated by radiation therapy. Radiat Oncol 6:46–51
Sisti E, Menconi F, Leo M, Profilo MA, Mautone T, Mazzi B, Rocchi R, Latrofa F, Nardi M, Vitti P, Marcocci C, Marinò M (2015) Long-term outcome of Graves’ orbitopathy following high-dose intravenous glucocorticoids and orbital radiotherapy. J Endocrinol Invest 38:661–668
Nicosia L, Reverberi C, Agolli L, Marinelli L, De Sanctis V, Minniti G, Valeriani M, Osti MF (2019) Orbital radiotherapy plus concomitant steroids in moderate-to-severe Graves’ Ophtlamopathy: good results after long-term follow-up Int J. Endocrinol Metab 17:e84427
Wakelkamp IMM, Tan H, Saeed P, Schlingemann RO, Verbraak FD, Blank LEC, Prummel MF, Wiersinga WM (2004) Orbital irradiation for Graves’ ophthalmopathy. Ophthalmology 111:1557–1562
Viani GA, Boin AC, De Fendi LI, Fonseca EC, Stefano EJ, de Paula JS (2012) Radiation therapy for Graves’ ophthalmopathy: a systematic review and meta-analysis of randomized controlled trials. Arq Bras Oftalmol 75:324–332
Godfrey KJ, Kazim M (2018) Radiotherapy for active thyroid eye disease. Ophthalmic Plast Reconstr Surg 34:S98–S104
Rajendram R, Bunce C, Lee RW, Morley AM (2012) Orbital radiotherapy for adult thyroid eye disease. Cochrane Database Syst Rev 7:CD007114
Wiersinga WM, Perros P, Kahaly GJ, Mourits MP, Baldeschi L, Boboridis K, Boschi A, Dickinson AJ, Kendall-Taylor P, Krassas GE, Lane CM, Lazarus JH, Marcocci C, Marinò M, Nardi M, Neoh C, Orgiazzi J, Pinchera A, Pitz S, Prummel MF, Sartini MS, Stahl M, Arx V (2006) Clinical assessment of patients with Graves’ orbitopathy: the European Group on Graves’ Orbitopathy recommendations to generalists, specialists and clinical researchers. Eur J Endocrinol 155:387–389
Bartalena L, Baldeschi L, Dickinson A, Eckstein A, Kendall-Taylor P, Marcocci C, Mourits M, Perros P, Boboridis K, Boschi A, Currò N, Daumerie C, Kahaly GJ, Krassas GE, Lane CM, Lazarus JH, Marinò M, Nardi M, Neoh C, Orgiazzi J, Pearce S, Pinchera A, Pitz S, Salvi M, Sivelli P, Stahl M, von Arx G, Wiersinga WM (2008) Consensus statement of the European Group on Graves’ orbitopathy (EUGOGO) on management of GO. Eur J Endocrinol 158:273–285
Hiromatsu Y, Eguchi H, Tani J, Kasaoka M, Teshima Y (2014) Graves’ Ophthalmopathy: epidemiology and Natural History. Int Med 53:353–360
Marcocci C, Marinò M (2012) Treatment of mild, moderate-to-severe and very severe Graves’ orbitopathy. Best Pract Res Clin Endocrinol Metab 26:325–337
Zang S, Ponto KA, Kahaly GJ (2011) Intravenous glucocorticoids for Graves’ orbitopathy: efficacy and morbidity. J Clin Endocrinol Metab 96:320–332
Bartalena L (2013) Graves’ orbitopathy: imperfect treatments for a rare disease. Eur Thyroid J 2:259–269
Tanda ML, Bartalena L (2012) Efficacy and safety of orbital radiotherapy for Graves’ orbitopathy. J Clin Endocrinol Metab 97:3857–3865
Prummel MF, Terwee CB, Gerding MN, Baldeschi L, Mourits MP, Blank L, Dekker FW, Wiersinga WM (2004) A randomized controlled trial of orbital radiotherapy versus sham irradiation in patients with mild graves’ ophthalmopathy. J Clin Endocrinol Metab 89:15–20
Zygulska A (2009) Radiotherapy in the treatment of Graves ophthalmopathy—to do it or not? J Ocul Biol Dis Inform 3:1–11
Gorman CA, Garrity JA, Fatourechi V, Bahn RS, Petersen IA, Stafford SL, Earle JD, Forbes GS, Kline RW, Bergstralh EJ, Offord KP, Rademacher DM, Stanley NM, Bartley GB (2001) A prospective, randomized, double-blind, placebo-controlled study of orbital radiotherapy for Graves’ ophthalmopathy. Ophthalmology 108:1523–1534
Rajendram R, Taylor PN, Wilson VJ, Harris N, Morris OC, Tomlinson M, Yarrow S, Garrott H, Herbert HM, Dick AD, Cook A, Gattamaneni R, Jain R, Olver J, Hurel SJ, Bremner F, Drummond SR, Kemp E, Ritchie DM, Rumsey N, Morris D, Lane C, Palaniappan N, Li C, Pell J, Hills R, Ezra DG, Potts MJ, Jackson S, Rose GE, Plowman N, Bunce C, Uddin JM, Lee RWJ, Dayan CM (2018) Combined immunosuppression and radiotherapy in thyroid eye disease (CIRTED): a multicentre, 2 × 2 factorial, double-blind, randomised controlled trial. Lancet Diabetes Endocrinol 6:299–309
Marcocci C, Bartalena L, Bogazzi F, Bruno-Bossio G, Lepri A, Pinchera A (1991) Orbital radiotherapy combined with high dose systemic glucocorticoids for Graves’ ophthalmopathy is more effective than radiotherapy alone: results of a prospective randomized study. J Endocrinol Invest 14:853–860
Bartalena L, Veronesi G, Krassas GE, Wiersinga WM, Marcocci C, Marinò M, Salvi M, Daumerie C, Bournaud C, Stahl M, Sassi L, Azzolini C, Boboridis KG, Mourits MP, Soeters MR, Baldeschi L, Nardi M, Currò N, Boschi A, Bernard M, von Arx G, Perros P, Kahaly GJ (2012) European Group on Graves’ Orbitopathy (EUGOGO) (2017): Does early response to intravenous glucocorticoids predict the final outcome in patients with moderate-to-severe and active Graves’ orbitopathy? J Endocrinol Invest 40:547–553
Kahaly GJ, Rösler H-P, Pitz S, Hommel G (2000) Low- versus high-dose radiotherapy for graves’ ophthalmopathy: a randomized. Single Blind Trial J Clin Endocrinol Metab 85:102–108
Marcocci C, Bartalena L, Rocchi R, Marinò M, Menconi F, Morabito E, Mazzi B, Mazzeo S, Sartini MS, Nardi M, Cartei F, Cionini L, Pinchera A (2003) Long-term safety of orbital radiotherapy for graves’ ophthalmopathy. J Clin Endocrinol Metab 88:3561–3566
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standards
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Limone, P.P., Bianco, L., Mellano, M. et al. Is concomitant treatment with steroids and radiotherapy more favorable than sequential treatment in moderate-to-severe graves orbitopathy?. Radiol med 126, 334–342 (2021). https://doi.org/10.1007/s11547-020-01244-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11547-020-01244-5