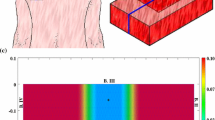
Abstract
Keloids are fibroproliferative disorders described by excessive growth of fibrotic tissue, which also invades adjacent areas (beyond the original wound borders). Since these disorders are specific to humans (no other animal species naturally develop keloid-like tissue), experimental in vivo/in vitro research has not led to significant advances in this field. One possible approach could be to combine in vitro human models with calibrated in silico mathematical approaches (i.e., models and simulations) to generate new testable biological hypotheses related to biological mechanisms and improved treatments. Because these combined approaches do not really exist for keloid disorders, in this brief review we start by summarising the biology of these disorders, then present various types of mathematical and computational approaches used for related disorders (i.e., wound healing and solid tumours), followed by a discussion of the very few mathematical and computational models published so far to study various inflammatory and mechanical aspects of keloids. We conclude this review by discussing some open problems and mathematical opportunities offered in the context of keloid disorders by such combined in vitro/in silico approaches, and the need for multi-disciplinary research to enable clinical progress.


Similar content being viewed by others
References
Abaid N, Eftimie R, Hutt A, Veen L (2022) Editorial: Modelling collective motion across scales. Front Appl Math Stat 8:932364
Abergel RP, Pizzurro D, Meeker CA, Lask G, Matsuoka LY, Minor RR, Chu M-L, Uitto J (1985) Biochemical composition of the connective tissue in keloids and analysis of collagen metabolism in keloid fibroblast cultures. J Invest Dermatol 84(5):384–390
Addison T (1854) On the keloid of Alibert and on true keloid. Med Chir Trans 37:27–47
Adebayo OE, Urcun S, Rolin G, Bordas SPA, Trucu D, Eftimie R (2023) Mathematical investigation of normal and abnormal wound healing dynamics: local and non-local models. Math Biosci Eng 20(9):17446–17498
Agbenorkul PT, Kus H, Myczcowski T (1995) The keloid triad hypothesis (KHT): a basis for keloid etiopathogenesis and clues for prevention. Eur J Plast Surg 18:301–304
Akaishi S, Ogawa R, Hyakusoku H (2008a) Keloid and hypertrophic scar: neurogenic inflammation hypotheses. Med Hypotheses 71(1):32–38
Akaishi S, Akimoto M, Ogawa R, Hyakusoku H (2008b) The relationship between keloid growth pattern and stretching tension: visual analysis using the finite element method. Ann Plast Surg 60(4):445–451
Alibert JL (1806) Description des Maladies de la Peau Observées À l’Hôpital Saint-Louis. Chez Barrois l’ainé et fils, Paris
Alibert JL (1822) Précis Théorique et Pratique sur les Maladies de la Peau. Tome 1. (2 ed.). Caille et Ravier, Paris, Paris
Alnaes MS, Blechta J, Hake J, Johansson A, Kehlet B, Logg A, Richardson C, Ring J, Rognes M, Wells GN (2015) The FEniCS project version 1.5. Arch Numer Softw 3(100):9–23
Altrock PM, Liu LL, Michor F (2015) The mathematics of cancer: integrating quantitative models. Nat Rev Cancer 15:730–745
Anderson ARA, Chaplain MAJ, Newman EL, Steele RJC, Thompson AM (2000) Mathematical modelling of tumour invasion and metastasis. Comput Math Methods Med 2:129–154
Aoki M, Miyake K, Ogawa R, Dohi T, Akaishi S, Hyakusoku H, Shimada T (2014) siRNA knockdown of tissue inhibitor of metalloproteinase-1 in keloid fibroblasts leads to degradation of collagen type I. J Invest Dermatol 134:818–826
Araujo RP, McElwain DLS (2004) A history of the study of solid tumour growth: the contribution of mathematical modelling. Bull Math Biol 66:1039–1091
Arciero J, Swingdon D (2013) Equation-based models of wound healing and collective cell migration. In: Vodovotz Y, An G (eds) Complex systems and computational biology approaches to acute inflammation. Springer, New York, pp 185–207, Chap. 11
Babu M, Diegelmann R, Oliver N (1992) Keloid fibroblasts exhibit an altered response to TGF-\(\beta \). J Invest Dematol 99:650–655
Bayat A, Arscott G, Ollier WER, Grouther DAM, Ferguson MWJ (2005) Keloid disease: clinical relevance of single versus multiple site scars. Br J Plast Surg 58:28–37
Bekisz S, Geris L (2020) Cancer modeling: from mechanistic to data-driven approaches, and from fundamental insights to clinical applications. J Comput Sci 46:101198
Bettinger DA, Yager DR, Diegelmann RF, Cohen IK (1996) The effect of TGF-\(\beta \) on keloid fibroblast proliferation and collagen synthesis. Plast Reconstr Surg 98:827–833
Bianca C (2011) Mathematical modeling for keloid formation triggered by virus: Malignant effects and immune system competition. Math Models Methods Appl Sci 21:389–419
Bianca C, Fermo L (2011) Bifurcation diagrams for the moments of a kinetic type model of keloid-immune system competition. Comput Math Appl 61:277–288
Bienias W, Miȩkoś-Zydek B, Kaszuba A (2011) Current views on the etiopathogenesis of keloids. Post Dermatol Alergol 28(6):467–475
Bordas S, Duflot M (2007) Derivative recovery and a posteriori error estimate for extended finite elements. Comput Methods Appl Mech Eng 196(35–36):3381–3399
Bordas S, Duflot M, Le P (2008) A simple error estimator for extended finite elements. Commun Numer Methods Eng 24(11):961–971
Bowden LG, Byrne HM, Maini PK, Moulton DE (2016) A morphoelastic model for dermal wound closure. Biomech Model Mechanobiol 15(3):663–681
Bran GM, Goessler UR, Hormann K, Riedel F, Sadick H (2009) Keloids: current concepts of pathogenesis (review). Int J Mol Med 24:283–293
Buganza Tepole A, Kuhl E (2013) Systems-based approaches toward wound healing. Pediatr Res 73(2):553–563
Bui HP, Tomar S, Courtecuisse H, Cotin S, Bordas SP (2017) Real-time error control for surgical simulation. IEEE Trans Biomed Eng 65(3):596–607
Bui HP, Tomar S, Courtecuisse H, Audette M, Cotin S, Bordas SP (2018) Controlling the error on target motion through real-time mesh adaptation: applications to deep brain stimulation. Int J Numer Methods Biomed Eng 34(5):2958
Butler PD, Longaker MT, Yang GP (2008) Current progress in keloid research and treatment. J Am Coll Surg 206:731–741
Buttenschön A, Edelstein-Keshet L (2020) Bridging from single to collective cell migration: a review of models and links to experiments. PLoS Comput Biol 16(12):1008411
Camley BA, Rappel W-J (2017) Physical models of collective cell motility: from cell to tissue. J Phys D Appl Phys 50(11):113002
Carrillo JA, D’Orsogna MR, Panferov V (2009) Double milling in self-propelled swarms from kinetic theory. Kinetic Realted Models 2(2):363–378
Carthy JM (2018) TGF\(\beta \) signaling and the control of myofibroblast differentiation: implications for chronic inflammatory disorders. J Cell Physiol 233:98–106
Chambert J, Jacquet E, Remache D (2012) Numerical analysis of keloid scar in the presternal area. Comput Methods Biomech Biomed Engin 15(1):23–24
Chambert J, Lihoreau T, Joly S, Chatelain B, Sandoz P, Humbert P, Jacquet E, Rolin G (2019) Multimodal investigation of a keloid scar by combining mechanical tests in vivo with diverse imaging techniques. J Mech Behav Biomed Mater 99:206–215
Chaplain MA, McDougall SR, Anderson AR (2006) Mathematical modeling of tumor-induced angiogenesis. Annu Rev Biomed Eng 8:233–257
Chen L, Painter K, Surulescu C, Zhigun A (2020) Mathematical models for cell migration: a non-local perspective. Philos Trans R Soc B 375:1807
Chuang Y-L, D’Orsogna MR, Marthaler D, Bertozzi AL, Chayes LS (2007) State transitions and the continuum limit for a 2D interacting, self-propelled particle system. Physica D 232:33–47
Cobbold CA, Sherratt JA (2000) Mathematical modelling of nitric oxide activity in wound healing can explain keloid and hypertrophic scarring. J Theor Biol 204(2):257–288
Cumming BD, McElwain DL, Upton Z (2010) A mathematical model of wound healing and subsequent scarring. J R Soc Interface 7:19–34
Dallon JC, Sherratt JA, Maini PK (1999) Mathematical modelling of extracellular matrix dynamics using discrete cells: fiber orientation and tissue regeneration. J Theor Biol 199:449–471
Dallon JC, Sherratt JA, Maini PK, Ferguson M (2000) Biological implications of discrete mathematical model for collagen deposition and alignment in dermal wound repair. IMA J Math Appl Med Biol 17:379–393
Desmouliére A, Geinoz A, Gabbiani F, Gabbiani G (1993) Transforming growth factor-\(\beta \)1 induces \(\alpha \)-smooth muscle actin expression in granulation tissue myofibroblasts and in quiescent and growing cultured fibroblasts. J Cell Biol 122:103–111
Deutsch A, Friedl P, Preziosi L, Theraulaz G (2020) Multi-scale analysis and modelling of collective migration in biological systems. Philos Trans R Soc B 375(1807):20190377
Diesboeck TS, Wang Z, Macklin P, Cristini V (2011) Multiscale cancer modelling. Annu Rev Biomed Eng 13:127–155
Du H, Bartleson JM, Butenko S, Alonso V, Liu WF, Winer DA, Butte MJ (2023) Tuning immunity through tissue mechanotransduction. Nat Rev Immunol 23:174–188
Duddu R, Bordas S, Chopp D, Moran B (2008) A combined extended finite element and level set method for biofilm growth. Int J Numer Meth Eng 74(5):848–870
Duprez M, Bordas SPA, Bucki M, Bui HP, Chouly F, Lleras V, Lobos C, Lozinski A, Rohan P-Y, Tomar S (2020) Quantifying discretization errors for soft tissue simulation in computer assisted surgery: a preliminary study. Appl Math Model 77:709–723
Dvorak HF (1986) Tumors: wounds that do not heal: similarities between tumor stroma generation and wound healing. N Engl J Med 315:1650–1659
Eftimie R, Barelle C (2021) Mathematical investigation of innate immune responses to lung cancer: the role of macrophages with mixed phenotypes. J Theor Biol 524:110739
Eftimie G, Eftimie R (2022) Quantitative predictive approaches for Dupuytren disease: a brief review and future perspectives. Math Biosci Eng 19(3):2876–2895
Eftimie R, Bramson JL, Earn DJD (2011) Interactions between the immune system and cancer: a brief review of non-spatial mathematical models. Bull Math Biol 73(1):2–32
Eftimie R, Mavrodin A, Bordas SPA (2022) From digital control to digital twins in medicine: a brief review and future pespectives. Adv Appl Mech 56:1–49
Enderling H, Chaplain MAJ (2014) Mathematical modelling of tumour growth and treatment. Curr Pharm Des 20(30):4934–4940
Escuin-Ordinas H, Li S, Xie MW, Sun L, Hugo W, Huang RR, Jiao J, de-Faria FM, Realegeno S, Azhdam PKA, Komenan SMD, Atefi M, Comin-Anduix B, Pellegrini M, Cochran AJ, Modlin RL, Herschman HR, Lo RS, McBride WH, Segura T, Ribas A (2016) Cutaneous wound healing through paradoxical MAPK activation by BRAF inhibitors. Nat Commun 7:12348
Flegg JA, Menon SN, Maini PK, McElwain DLS (2015) On the mathematical modeling of wound healing angiogenesis in skin as a reaction-transport process. Front Physiol 6:1–17
Fujiwara M, Muragaki Y, Ooshima A (2005) Upregulation of transforming growth factor-beta1 and vascular endothelial growth factor in cultured keloid fibroblasts: relevance to angiogenic activity. Arch Dermatol Res 297:161–169
Gaffney EA, Pugh K, Maini PK, Arnold F (2002) Investigating a simple model of cutaneous wound healing angiogenesis. J Math Biol 45:337–374
Gauglitz GG, Korting HC, Pavicic T, Ruzicka T, Jeschke MG (2011) Hypertrophic scarring and keloids: pathomechanisms and current and emerging treatment strategies. Mol Med 17:113–125
Ghazawi FM, Zargham R, Gilardino MS, Sasseville D, Jafarian F (2018) Insights into the pathophysiology of hypertrophic scars and keloids: how do they differ? Adv Skin Wound Care 31(1):582–594
Greenspan HP (1976) On the growth and stability of cell cultures and solid tumours. J Theor Biol 56:229–242
Haisa M, Okochi H, Grotendorst GR (1994) Elevated levels of PDGF alpha receptors in keloid fibroblasts contribute to an enhanced response to PDGF. J Invest Dermatol 103:560–563
Hajdu SI (2011) A note from history: landmarks in history of cancer, part 1. Cancer 117(5):1097–1102
He Y, Merin MR, Sharon VR, Maverakis E (2011) Eruptive keloids associated with breast cancer: a paraneoplastic phenomenon? Acta Derm Venereol 91:480–481
Huang C, Ogawa R (2022) Roles of inflammasomes in keloids and hypertrophic scars—lessons learned from chronic diabetic wounds and skin fibrosis. Int J Mol Sci 23:6820
Hunt CA, Ropella GEP, Park S, Engelberg J (2008) Dichotomies between computational and mathematical models. Nat Biotechnol 26:737–738
Jagadeesan J, Bayat A (2007) Transforming growth factor beta (TGF\(\beta \)) and keloid disease. Int J Surg Lond Engl 5:278–285
Jarrett AM, Hormuth DA, Wu C, Kazerouni AS, Ekrut DA, Virostko J, Sorace AG, DiCarlo JC, Kowalski J, Patt D, Goodgame B, Avery S, Yankeelov TE (2020) Evaluating patient-specific neoadjuvant regimens for breast cancer via a mathematical model constrained by quantitative magnetic resonance imaging data. Neoplasia 22(12):820–830
Jones SG, Edwards R, Thomas DW (2004) Inflammation and wound healing: the role of bacteria in the immuno-regulation of wound healing. Int J Low Extrem Wounds 3(4):201–208
Jorgensen SN, Sanders JR (2016) Mathematical models of wound healing and closure: a comprehensive review. Med Biol Eng Comput 54(9):1297–1316
Jumper N, Paus R, Bayat A (2015) Functional histopathology of keloid disease. Histol Histopathol 30:1033–1057
Kather JN, Poleszczuk J, Suarez-Carmona M, Krisam J, Charoentong P, Valous NA, Weis C-A, Tavernar L, Leiss F, Herpel E, Klupp F, Ulrich A, Schneider M, Jäger AM, Halama N (2017) In silico modeling of immunotherapy and stroma-targeting therapies in human colorectal cancer. Cancer Res 77(22):6442–6452
Kim SW (2021) Management of keloid scars: noninvasive and invasive treatments. Arch Plast Surg 48(2):149–157
Kim Y, Stolarska MA, Othmer HG (2007) A hybrid model for tumor spheroid growth in vitro i: theoretical development and early results. Math Models Methods Appl Sci 17(supp01):1773–1798
Kim MJ, Reed D, Rejniak K (2014) The formation of tight tumour clusters affects the efficacy of cell cycle inhibitors: a hybrid model study. J Theor Biol 352:31–50
Kimura S, Tsuji T (2021) Mechanical and immunological regulation in wound healing and skin reconstruction. Int J Mol Sci 22:5474
Kimura K, Inadomi T, Yamauchi W, Yoshida Y, Kashimura T, Terui T (2014) Dermatofibrosarcoma protuberans on the chest with a variety of clinical features masquerading as a keloid: is the disease really protuberant? Ann Dermatol 26:643–645
Kirshtein A, Akbarinejad S, Hao W, Le T, Su S, Aronow RA, Shahriyari L (2020) Data driven mathematical model of colon cancer progression. J Clin Med 9:3947
Koppenol DC, Vermolen FJ (2017) Biomedical implications from a morphoelastic continuum model for the simulation of contracture formation in skin grafts that cover excised burns. Biomech Model Mechanobiol 16:1187–1206
Koppenol DC, Vermolen FJ, Niessen FB, Zuijlen PPM, Vuik K (2017) A mathematical model for the simulation of the formation and the subsequent regression of hypertrophic scar tissue after dermal wounding. Biomech Model Mechanobiol 16:15–32
Kuehlmann B, Bonham CA, Zucal I, Prantl L, Gurtner GC (2020) Mechanotransduction in wound healing and fibrosis. J Clin Med 9:1423
Laird AK, Tyler SA, Barton AD (1965) Dynamics of normal growth. Growth 21:233–248
Leaper D, Assadian O, Edmiston CE (2015) Approach to chronic wound infections. Br J Dermatol 173(2):351–358
Lee S, Rauch J, Kolch W (2020) Targeting MAPK signaling in cancer: mechanisms of drug resistance and sensitivity. Int J Mol Sci 21(3):1102
Li X, Wang Y, Yuan B, Yang H, Qiao L (2017) Status of M1 and M2 type macrophages in keloid. Int J Clin Exp Pathol 10(11):11098–11105
Li Q, Chen H, Liu Y, Bi J (2023) Osteomodulin contributes to keloid development by regulating p38 MAPK signaling. J Dermatol 50(7):895–905
Limandjaja GC, Al LJ (2018) Reconstructed human keloid models show heterogeneity within keloid scars. Arch Dermatol Res 310:815–826
Limandjaja GC, Nielsen FB, Scheper RJ, Gibbs S (2020) The keloid disorder: heterogeneity, histopathology, mechanisms and models. Front Cell Dev Biol 8:360
Limandjaja GC, Niessen FB, Scheper RJ, Gibbs S (2021) Hypertrophic scars and keloids: overview of the evidence and practical guide for differentiating between these abnormal scars. Exp Dermatol 30:146–161
Liu X, Fang J, Huang S, Wu X, Xie X, Wang J, Liu F, Zhang M, Peng Z, Hu N (2021) Tumour-on-a-chip: from bioinspired design to biomedical application. Microsyst Nanoeng 7:50
Lonardo AD, Nasi S, Pulciani S (2015) Cancer: we should not forget the past. J Cancer 6(1):29–39
Lu F, Gao J, Ogawa R, Hyakusoku H, Ou C (2007) Biological differences between fibroblasts derived from peripheral and central areas of keloid tissues. Plast Reconstr Surg 120:625–630
Lu Y-Y, Tu H-P, Wu C-H, Hong C-H, Yang K-C, Yang HJ, Chang K-L, Lee C-H (2021) Risk of cancer development in patients with keloids. Sci Rep 11:9390
Macnamara CK (2021) Biomechanical modelling of cancer: agent-based force-based models of solid tumours within the context of the tumour microenvironment. Comput Syst Oncol 1:1018
Maini PK, Baker RE (2014) Modelling collective cell motion in biology. In: Ansari AR (ed) Advances in applied mathematics. Springer proceedings in mathematics & statistics, vol 87, pp 1–11
Marie N, Lejeune A, Chouly F, Chambert J, Jacquet E (2022) DWR error estimator for the biomechanics of the skin with a keloid scar. FigShare repository. https://doi.org/10.6084/m9.figshare.19372280.v1
Maroudas-Sacks Y, Zemel A (2018) theoretical analysis of stress distribution and cell polarisation surrounding a model wound. Biophys J 115(2):398–410
Marttala J, Andrews JP, Rosenbloom J, Uitto J (2016) Keloids: animal models and pathological equivalents to study tissue fibrosis. Matrix Biol 51:47–54
McDougall S, Dallon J, Sherratt JA, Maini P (2006) Fibroblast migration and collagen deposition during dermal wound healing: mathematical modelling and clinical implications. Philos Trans R Soc A 364:1385–1405
Metzcar J, Wang Y, Heiland R, Macklin P (2019) A review of cell-based computational modelling in cancer biology. Clin Cancer Inf 3:1–13
Mi Q, Riviere B, Clermont G, Steed DL, Vodovotz Y (2007) Agent-based model of inflammation and wound healing: insights into diabetic foot ulcer pathology and the role of transforming growth factor-beta1. Wound Repair Regen 15:671–682
Monfared GS, Ertl P, Rothbauer M (2021) Microfluidic and lab-on-a-chip systems for cutaneous wound healing studies. Pharmaceutics 13:793
Monte UD (2009) Does the cell number \(10^{9}\) still really fit one gram of tumour tissue? Cell Cycle 8(3):505–506
Nicholas, R.S., Stodell, M.: An important case of misdiagnosis: keloid scar or high-grade soft-tissue sarcoma? BMJ Case Rep 1–2 (2014)
Niessen FB, Spauwen PH, Schalkwijk J, Kon M (1999) On the nature of hypertrophic scars and keloids: a review. Plastic Reconstr Surg 104:1435–1458
Ogawa R (2011) Mechanobiology of scarring. Wound Repair Regen 19(Suppl. 1):2–9
Ogawa R (2017) Keloid and hypertrophic scars are the result of chronic inflammation in the reticular dermis. Int J Mol Sci 18(3):606
Ogawa R, Okai K, Tokumura F, Mori K, Ohmori Y, Hyakusoku H, Huang C, Akaishi S (2012) The relationship between skin stretching/contraction and pathologic scarring: the important role of mechanical forces in keloid generation. Wound Repair Regen 20:149–157
Olsen L, Sherratt JA, Maini PK (1995) A mechanochemical model for adult dermal wound contraction and the permanence of the contracted tissue displacement profile. J Theor Biol 177:113–128
Olsen L, Sherratt JA, Maini PK (1996) A mathematical model for fibro-proliferative wound healing disorders. Bull Math Biol 58(4):787–808
Pappalardo F, Palladini A, Pennisi M, Castiglione F, Motta S (2012) Mathematical and computational models in tumour immunology. Math Model Nat Phenomena 7(3):186–203
Pettet G, Chaplain MAJ, McElwain DLS, Byrne HM (1996) On the role of angiogenesis in wound healing. Proc R Soc Lond B 263(1376):1487–1493
Poleszczuk J, Enderling H (2014) A high-performance cellular automaton model of tumour growth with dynamically growing domains. Appl. Math. (Irvine) 5(1):144–152
Posta F, Chou T (2010) A mathematical model of intercellular signaling during epithelial wound healing. J Theor Biol 266(1):70–78
Quaranta V, Rejniak KA, Gerlee P, Anderson ARA (2008) Invasion emerges from cancer cell adaptation to competitive microenvironments: quantitative predictions from multiscale mathematical models. Semin Cancer Biol 18(5):338–348
Raimúndez E, Keller S, Zwingenberger G, Ebert K, Hug S, Theis FJ, Maier D, Luber B, Hasenauer J (2020) Model-based analysis of response and resistance factors of cetuximab treatment in gastric cancer cell lines. PLoS Comput Biol 16(3):1007147
Rejniak KA, Anderson ARA (2011) Hybrid models of tumour growth. Wiley Interdiscip Rev Syst Biol Med 3(1):115–125
Renardy M, Hult C, Evans S, Linderman JJ, Kirschner DE (2019) Global sensitivity analysis of biological multiscale models. Curr Opin Biomed Eng 11:109–116
Requena L, Martin L, Na MCF, Escalonilla P (1996) Keloidal basal cell carcinoma. A new clinicopathological variant of basal cell carcinoma. Br J Dermatol 134(5):953–957
Roose T, Chapman SJ, Maini PK (2007) Mathematical models of avascular tumour growth. SIAM Rev 49(2):179–208
Schäffer MR, Efron PA, Thornton FJ, Klingel K, Gross SS, Barbul A (1997a) Nitric oxide, an autocrine regulator of wound fibroblast synthetic function. J Immunol 158(5):2375–2381
Schäffer MR, Tantry U, Wesep RA, Barbul A (1997b) Nitric oxide metabolism in wounds. J Surg Res 71(1):25–31
Sfakianakis N, Chaplain MAJ (2021) Mathematical modelling of cancer invasion: a review. In: Suzuki T, Poignard C, Chaplain M, Quaranta V (eds.) Methods of mathematical oncology. MMDS 2020. Springer proceedings in mathematics & statistics, vol 370, pp 153–172
Shan M, Liu H, Song K, Liu S, Hao Y, Wang Y (2022) Immune-related gene expression in skin, inflamed and keloid tissue from patients with keloids. Oncol Lett 23:72
Sherratt JA, Dallon JC (2002) Theoretical models of wound healing: past successes and future challenges. C R Biologies 325:557–564
Sherratt JA, Murray JD (1990) Models of epidermal wound healing. Proc R Soc Lond B 241:29–36
Shi CK, Zhao Y-P, Ge P, Huang G-B (2019) Therapeutic effect of interleukin-10 in keloid fibroblasts by suppression of TGF-\(\beta \)/Smad pathway. Eur Rev Med Pharmacol Sci 23:9085–9092
Shuttleworth R, Trucu D (2019) Multiscale modelling of fibres dynamics and cell adhesion within moving boundary cancer invasion. Bull Math Biol 81:2176–2219
Stern JR, Christley S, Zaborina O, Alverdy JC, An G (2012) Integration of TGF-\(\beta \) and EGFR based signaling pathways using an agent based model of epithelial restitution. Wound Repair Regen 20(6):862–871
Stolarska MA, Kim Y, Othmer HG (2009) Multi-scale models of cell and tissue dynamics. Philos Trans R Soc A Math Phys Eng Sci 367(1902):3525–3553
Supp DM (2019) Animal models or studies of keloid scarring. Adv Wound Care (New Rochelle) 8(2):77–89
Suttho D, Mankhetkorn S, Binda D, Pazart L, Humbert P, Rolin G (2017) 3D modelling of keloid scars in vitro by cell and tissue engineering. Arch Dermatol Res 309:55–62
Sutula D, Elouneg A, Sensale M, Chouly F, Chambert J, Lejeune A, Baroli D, Hauseux P, Bordas S, Jacquet E (2020) An open source pipeline for design of experiments for hyperelastic models of the skin with applications to keloids. J Mech Behav Biomed Mater 112:103999
Suveges S, Eftimie R, Trucu D (2020) Directionality of macrophages movement in tumour invasion: a multiscale moving-boundary approach. Bull Math Biol 82(12):148
Suveges S, Chamseddine I, Rejniak KA, Eftimie R, Trucu D (2021) Collective cell migration in a fibrous environment: a hybrid multiscale modelling approach. Front Appl Math Stat 7:680029
Suveges S, Eftimie R, Trucu D (2022) Re-polarisation of macrophages within collective tumour cell migration: a multiscale moving boundary approach. Front Appl Math Stat 7:799650
Tan S, Khumalo N, Bayat A (2019) Understanding keloid pathobiology from a quasi-neoplastic perspective: less of a scar and more of a chronic inflammatory disease with cancer-like tendencies. Front Immunol 10:1664–3224
Tanzer ML (2006) Current concepts of extracellular matrix. J Orthop Sci 11:326–331
Tepole AB, Kuhl E (2016) Computational modeling of chemo-bio-mechanical coupling: a systems-biology approach toward wound healing. Comput Methods Biomech Biomed Engin 19(1):13–30
Thackham JA, McElwain DLS, Turner IW (2009) Computational approaches to solving equations arising from wound healing. Bull Math Biol 71:211–246
Thomlinson RH, Gray LH (1955) The histological structure of some human lung cancers and the possible implications for radiotherapy. Br J Cancer 9:539–549
Tongdee E, Touloei K, Shitabata PK, Shareef S, Maranda EL (2016) Keloidal atypical fibroxanthoma: case and review of the literature. Case Rep Dermatol 8:156–163
Tranquillo RT, Murray JD (1993) Mechanistic model of wound contraction. J Surg Res 55:233–247
Ulrich D, Ulrich F, Unglaub F, Piatkowski A, Pallua N (2010) Matrix metalloproteinases and tissue inhibitors of metalloproteinases in patients with different types of scars and keloids. J Plast Reconstr Aesthet Surg 63:1015–1021
Urcun S, Rohan P-Y, Skalli W, Nassoy P, Bordas SPA, Sciumè G (2021) Digital twinning of cellular capsule technology: emerging outcomes from the perspective of porous media mechanics. PLoS ONE 16(7):1–30
Venkatesh KP, Raza MM, Kvedar JC (2022) Heath digital twins as tools for precision medicine: consideration for computation, implementation, and regulation. NPJ Digital Med 5:150
Vincent AS, Phan TT, Mukhopadhyay A, Lim HY, Halliwell B, Wong KP (2008) Human skin keloid fibroblasts display bioenergetics of cancer cells. J Invest Dermatol 128:702–709
Vodovotz Y, Clermont G, Chow C, An G (2004) Mathematical models of acute inflammatory response. Curr Opin Crit Care 10:383
Wang Z-C, Zhao W-Y, Cao Y, Liu Y-Q, Sun Q, Shi P, Cai J-Q, Shen XZ, Tan W-Q (2020) The roles of inflammation in keloid and hypertrophic scars. Front Immunol 11:603187
Waugh HV, Sherratt JA (2006) Macrophage dynamics in diabetic wound healing. Bull Math Biol 68:197–207
Webb G (2022) The force of cell-cell adhesion in determining the outcome in a nonlocal advection diffusion model of wound healing. Math Biosci Eng 19(9):8689–8704
WHO I (2022) ICD-11 for Mortality and Morbidity Statistics. https://icd.who.int/browse11/l-m/en#/http%3a%2f%2fid.who.int%2ficd%2fentity%2f831995767 Accessed 02/2022
Xu X, Gu S, Huang X, Ren J, Gu Y, Wei C, Lian X, Li H, Gao Y, Jin R, Gu B, Zan T, Wang Z (2020) The role of macrophages in the formation of hypertrophic scars and keloids. Burns Trauma 8:006
Xue M, Jackson CJ (2015) Extracellular matrix reorganization during wound healing and its impact on abnormal scarring. Adv Wound Care 4:119–136
Yankeelov TE, Atuegwu N, Hormuth D, Weis JA, Barnes SL, Miga MI, Rericha EC, Quaranta V (2013) Clinically relevant modelling of tumour growth and treatment response. Sci Transl Med 5(187):187–9
Yin A, Moes DJAR, Hasselt JGC, Swen JJ, Guchelaar H-J (2019) A review of mathematical models for tumour dynamics and treatment resistance evolution of solid tumours. CPT Pharmacometrics Syst Pharmacol 8(10):720–737
Zhang T, Wang X-F, Wang Z-C, Lou D, Fang Q-Q, Hu Y-Y, Zhao W-Y, Zhang L-Y, Wu L-H, Tan W-Q (2020) Current potential therapeutic strategies targeting the TGF-\(\beta \)/Smad signaling pathway to attenuate keloid and hypertrophic scar formation. Biomed Pharmacother Biomed Pharmacother 129:110287
Zhou Y, Sun Y, Hou W, Ma L, Tao Y, Li D, Xu C, Bao J, Fan W (2020) The JAK2/STAT3 pathway inhibitor, AG490, suppresses the abnormal behavior of keloid fibroblasts in vitro. Int J Mol Med 46:191–200
Zhou B, Gao Z, Liu W, Wu X, Wang W (2022) Important role of mechanical microenvironment on macrophage dysfunction during keloid pathogenesis. Exp Dermatol 31(3):375–380
Zhu J, Ji L, Chen Y, Li H, Huang M, Dai Z, Wang J, Xiang D, Fu G, Lei Z, Chu X (2023) Organoids and organs-on-chips: insighths into predicting the efficacy of systemic treatment in colorectal cancer. Cell Death Discov 9:72
Ziraldo C, Mi Q, An G, Vodovotz Y (2013) Computational modeling of inflammtion in wound healing. Adv Wound Care (New Rochelle) 2(9):527–537
Acknowledgements
RE and OA acknowledge funding from a French Agence Nationale de Recherche (ANR) Grant Number ANR-21-CE45-0025-01; SPAB and SU acknowledge funding from a Luxembourg National Research Fund (FNR) Grant Number INTER/ANR/21/16399490; G.R. acknowledges funding from an ANR Grant Number ANR-21-CE45-0025-03; FC’s work is partially supported by the I-Site BFC project NAANoD, the EIPHI Graduate School (contract ANR-17-EURE-0002) and the ANR project S-KELOID (ANR-21-CE45-0025-04). FC is also grateful to the Center for Mathematical Modelling Grant FB20005.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Eftimie, R., Rolin, G., Adebayo, O.E. et al. Modelling Keloids Dynamics: A Brief Review and New Mathematical Perspectives. Bull Math Biol 85, 117 (2023). https://doi.org/10.1007/s11538-023-01222-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11538-023-01222-8