Abstract
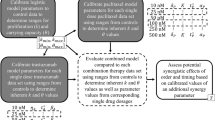
Triple-negative breast cancer (TNBC) is a heterogenous disease that is defined by its lack of targetable receptors, thus limiting treatment options and resulting in higher rates of metastasis and recurrence. Combination chemotherapy treatments, which inhibit tumor cell proliferation and regeneration, are a major component of standard-of-care treatment of TNBC. In this manuscript, we build a coupled ordinary differential equation model of TNBC with compartments that represent tumor proliferation, necrosis, apoptosis, and immune response to computationally describe the biological tumor affect to a combination of chemotherapies, doxorubicin (DRB) and paclitaxel (PTX). This model is parameterized using longitudinal [18F]-fluorothymidine positron emission tomography (FLT-PET) imaging data which allows for a noninvasive molecular imaging approach to quantify the tumor proliferation and tumor volume measurements for two murine models of TNBC. Animal models include a human cell line xenograft model, MDA-MB-231, and a syngeneic 4T1 mammary carcinoma model. The mathematical models are parameterized and the percent necrosis at the end time point is predicted and validated using histological hematoxylin and eosin (H&E) data. Global Sobol’ sensitivity analysis is conducted to further understand the role each parameter plays in the model’s goodness of fit to the data. In both the MDA-MB-231 and the 4T1 tumor models, the designed mathematical model can accurately describe both tumor volume changes and final necrosis volume. This can give insight into the ordering, dosing, and timing of DRB and PTX treatment. More importantly, this model can also give insight into future novel combinations of therapies and how the immune system plays a role in therapeutic response to TNBC, due to its calibration to two types of TNBC murine models.





Similar content being viewed by others
Data availability
The datasets analyzed throughout this study are available from the corresponding author upon reasonable request.
References
Aggarwal M, Hussaini M, De La Fuente L, Navarrete F, Cogan N (2018) A framework for model analysis across multiple experiment regimes: investigating effects of zinc on Xylella fastidiosa as a case study. J Theor Biol 14(457):88–100. https://doi.org/10.1016/j.jtbi.2018.08.028
Archer G, Saltelli A, Sobol’ I (1997) Sensitivity measures, ANOVA-like techniques and the use of bootstrap. J Statist Comput Simul. https://doi.org/10.1080/00949659708811825
Byrne H, Cox S, Kelly C (2004) Macrophage-tumour interactions: in vivo dynamics. Discrete Contin Dyn Syst - B. https://doi.org/10.3934/dcdsb.2004.4.81
Day J, Rubin J, Vodovotz Y, Chow CC, Reynolds A, Clermont G (2006) A reduced mathematical model of the acute inflammatory response II. Capturing scenarios of repeated endotoxin administration. J Theor Biol 242(1):237–256. https://doi.org/10.1016/j.jtbi.2006.02.015
Dent R, Trudeau M, Pritchard K, Hanna W, Kahn H, Sawka C, Narod S (2007) Triple-negative breast cancer: clinical features and patterns of recurrence. Clin Cancer Res 13(15):4429–4434. https://doi.org/10.1158/1078-0432
Eikenberry S (2009) A tumor cord model for doxorubicin delivery and dose optimization in solid tumors. Theor Biol Med Model 6:16. https://doi.org/10.1186/1742-4682-6-16
Ghobrial I, Whitzig T, Adjei A (2005) Targeting apoptosis pathways in cancer therapy. CA A Cancer J Clin 55(3):178–194. https://doi.org/10.3322/canjclin.55.3.178
Gomez J, Holmes N, Hansen A, Adhikarla V, Gutova M, Rockne R, Cho H (2022) Mathematical modeling of therapeutic neural stem cell migration in mouse brain with and without brain tumors. Math Biosci Eng. https://doi.org/10.3934/mbe.2022119
Hanahan D, Weinber R (2011) Hallmarks of cancer: the next generation. Cell. https://doi.org/10.1016/j.cell.2011.02.013
Hanahan D, Weinberg R (2000) The hallmarks of cancer. Cell 100(1):57–70. https://doi.org/10.1016/s0092-8674(00)81683-9
Jang S, Wientjes M, Au J (2001) Determinants of paclitaxel uptake, accumulation and retention in solid tumors. Invest New Drugs 19:113–123. https://doi.org/10.1023/A:1010662413174
Jarrett A, Hormuth D, Barnes S, Feng X, Huang W, Yankeelov T (2018) Incorporating drug delivery into an imaging-driven, mechanics-coupled reaction diffusion model for predicting the response of breast cancer to neoadjuvant chemotherapy: theory and preliminary clinical results. Phys Med Biol. https://doi.org/10.1088/1361-6560/aac040
Jarrett A, Bloom M, Godfrey W, Syed A, Ekrut D, Ehrlich L, Sorace A (2019) Mathematical modelling of trastuzumab-induced immune response in an in vivo murine model of HER2+ breast cancer. Math Med Biol J IMA. https://doi.org/10.1093/imammb/dqy014
Kamdee K, Saleewong T (2020) Mathematical modeling of triple negative breast cancer with doxorubicin. pp 435–438. https://doi.org/10.1109/ECTI-CON49241.2020.9158080
Kenny L, Coombes R, Vigushin D, Al-Nahhas A, Shousha S, Aboagye E (2007) Imaging early changes in proliferation at 1 week post chemotherapy: a pilot study in breast cancer patients with 3’-deoxy-3’-[18F]fluorothymidine positron emission tomography. Eur J Nucl Med Mol Imaging 34(9):1339–1347. https://doi.org/10.1007/s00259-007-0379-4
Klemm F, Joyce J (2015) Microenvironmental regulation of therapeutic response in cancer. Trends Cell Biol 25(4):1989–213. https://doi.org/10.1016/j.tcb.2014.11.006
Lampinen J (2002) A constraint handling approach for the differential evolution algorithm. Proceedings of the 2002 Congress on Evolutionary Computation, Vol. 2. IEEE, 2002, 1468–1473. https://doi.org/10.1109/CEC.2002.1004459
Liedtke C, Mazouni C, Hess KR, André F, Tordai A, Mejia JA, Fraser Symmans W, Gonzalez-Angulo AM, Hennessy B, Green M, Cristofanilli M, Hortobagyi GN, Pusztai L (2008) Response to neoadjuvant therapy and long-term survival in patients with triple-negative breast cancer. J Clin Oncol 26(8):1275–1281. https://doi.org/10.1200/JCO.2007.14.4147
Yun L, Li M, Massicano AVF, Song PN, Mansur A, Heinzman KA, Larimer BM, Lapi SE, Sorace AG (2021) [89Zr]-Pertuzumab PET imaging reveals paclitaxel treatment efficacy is positively correlated with HER2 expression in human breast cancer xenograft mouse models. Molecules 26(6):1568. https://doi.org/10.3390/molecules26061568
Ma G, Liu C, Lian W, Zhang Y, Yuan H, Zhang Y, Yang Z (2021) 18F-FLT PET/CT imaging for early monitoring response to CDK4/6 inhibitor therapy in triple negative breast cancer. Ann Nucl Med 35(5):600–607. https://doi.org/10.1007/s12149-021-01603-w
Macklin P, Lowengrub J (2007) Nonlinear simulation of the effect of microenvironment on tumor growth. J Theor Biol. https://doi.org/10.1016/j.jtbi.2006.12.004
Maughan K, Lutterbie M, Ham P (2010) Treatment of breast cancer. Am Fam Physician 81(11):1339–1346
McKenna M, Weis J, Barnes S, Tyson D, Miga M, Quaranta V, Yankeelov T (2017) A predictive mathematical modeling approach for the study of doxorubicin treatment in triple negative breast cancer. Sci Rep. https://doi.org/10.1038/s41598-017-05902-z
Nedeljkovic M, Damjanovic A (2019) Mechanisms of chemotherapy resistance in triple-negative breast cancer - how we can rise to the challenge. Cells 8(9):957
Network NC (2020) Metastatic breast cancer. Retrieved from National Comprehensive Cancer Network: https://www.nccn.org/patients/guidelines/content/PDF/stage_iv_breast-patient.pdf
Oreskes N, Shrader-Frechette K, Belitz K (1994) Verification, validation, and confirmation of numerical models in the earth sciences. Science 263:641–646. https://doi.org/10.1126/science.263.5147.641
Raccagni I, Belloli S, Valtorta S, Stefano A, Presotto L, Pascali C, Moresco R (2018) [18F]FDG and [18F]FLT PET for the evaluation of response to neo-adjuvant chemotherapy in a model of triple negative breast cancer. PLoS ONE 13(5):e0197754. https://doi.org/10.1371/journal.pone.0197754
Reynolds A, Rubin J, Clermont G, Day J, Vodovotz Y, Ermentrout G (2006) A reduced mathematical model of the acute inflammatory response: I. Derivation of model and analysis of anti-inflammation. J Theor Biol 242:220–236. https://doi.org/10.1016/j.jtbi.2006.02.016
Roe-Dale R, Isaacson D, Kupfershmid M (2011) A mathematical model of breast cancer treatment with CMF and doxorubicin. Bull Math Biol 73:585–608. https://doi.org/10.1007/s11538-010-9549-9
Saltelli A (2002) Making best use of model evaluations to compute sensitivity indices. Comput Phys Commun 145:280–297
Sanghera B, Wong W, Sonoda L, Beynon G, Makris A, Woolf D, Ardeshna K (2014) FLT PET-CT in evaluation of treatment response. Indian J Nucl Med 29(2):65–73. https://doi.org/10.4103/0972-3919.130274
Shields A, Grierson J, Dohmen B, Machulla H, Stayanoff J, Lawhorn-Crews J, Mangner T (1998) Imaging proliferation in vivo with [F-18]FLT and positron emission tomography. Nat Med 4:1334–1336. https://doi.org/10.1038/3337
Sobol’ I (2001) Global sensitivity indices for nonlinear mathematical models and their Monte Carlo estimates. Math Comput Simul 55:271–280
Weis J, Miga M, Arlinghaus L, Li X, Abramson V, Chakravarthy A, Yankeelov T (2015) Predicting the response of breast cancer to neoadjuvant therapy using a mechanically coupled reaction-diffusion model. Cancer Res. https://doi.org/10.1158/0008-5472.CAN-14-2945
Whisenant J, McIntyre J, Peterson T, Kang H, Sanchez V, Manning H, Yankeelov T (2015) Utility of 18FLT-PET to asses treatment response in trastuzumab-resistant and sensitive HER2-overexpressing human breast cancer xenografts. Mol Imag Biol 17(1):119–128. https://doi.org/10.1007/s11307-014-0770-z
Wu J, Dhingra R, Manoj G, Remais J (2012) Sensitivity analysis of infectious disease models: methods, advances and their application. J R Soc Interface. https://doi.org/10.1098/rsif.2012.1018
Yankeelov T, Atuegwu N, Hormuth D, Weis J, Barnes S, Miga M, Quaranta V (2013) Clinically relevant modeling of tumor growth and treatment response. Sci Transl Med. https://doi.org/10.1126/scitranslmed.3005686
Acknowledgements
We thank the American Cancer Society for funding through RSG-18-006-01-CCE. We also thank the UAB O’Neal Comprehensive Cancer Center’s Preclinical imaging shared facility, P30CAO13148-48.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Davenport, A.A., Lu, Y., Gallegos, C.A. et al. Mathematical Model of Triple-Negative Breast Cancer in Response to Combination Chemotherapies. Bull Math Biol 85, 7 (2023). https://doi.org/10.1007/s11538-022-01108-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11538-022-01108-1