Abstract
Deterministic approximations to stochastic Susceptible–Infectious–Susceptible models typically predict a stable endemic steady-state when above threshold. This can be hard to relate to the underlying stochastic dynamics, which has no endemic steady-state but can exhibit approximately stable behaviour. Here, we relate the approximate models to the stochastic dynamics via the definition of the quasi-stationary distribution (QSD), which captures this approximately stable behaviour. We develop a system of ordinary differential equations that approximate the number of infected individuals in the QSD for arbitrary contact networks and parameter values. When the epidemic level is high, these QSD approximations coincide with the existing approximation methods. However, as we approach the epidemic threshold, the models deviate, with these models following the QSD and the existing methods approaching the all susceptible state. Through consistently approximating the QSD, the proposed methods provide a more robust link to the stochastic models.



Similar content being viewed by others
Data and materials
Matlab code for solving the models will be published online with the manuscript. Python code will be added to the Epidemics on Networks package.
References
Allen LJS, Burgin AM (2000) Comparison of deterministic and stochastic SIS and SIR models in discrete time. Math Biosci 163(1):1–33
Andersson H, Britton T (2000) Stochastic epidemics in dynamic populations: quasi-stationarity and extinction. J Math Biol 41(6):559–580
Artalejo JR, Economou A, Lopez-Herrero MJ (2010) The maximum number of infected individuals in SIS epidemic models: computational techniques and quasi-stationary distributions. J Comput Appl Math 233(10):2563–2574
Artalejo JR, Economou A, Lopez-Herrero MJ (2013) Stochastic epidemic models with random environment: quasi-stationarity, extinction and final size. J Math Biol 67(4):799–831
Boccaletti S, Latora V, Moreno Y, Chavez M, Hwang DU (2006) Complex networks: structure and dynamics. Phys Rep 424(4):175–308
Dambrine S, Moreau M (1981) Note on the stochastic theory of a self-catalytic chemical reaction. I. Phys A Stat Mech Appl 106(3):559–573
Dambrine S, Moreau M (1981) Note on the stochastic theory of a self-catalytic chemical reaction. II. Phys A 106(3):574–588
Darroch JN, Seneta E (1967) On quasi-stationary distributions in absorbing continuous-time finite markov chains. J Appl Probab 4(1):192–196
Dickman R, Vidigal R (2002) Quasi-stationary distributions for stochastic processes with an absorbing state. J Phys A Math Gen 35(5):1147
Eames KTD, Keeling MJ (2002) Modeling dynamic and network heterogeneities in the spread of sexually transmitted diseases. Proc Natl Acad Sci 99(20):13330–13335
Ferreira SC, Castellano C, Pastor-Satorras R (2012) Epidemic thresholds of the susceptible-infected-susceptible model on networks: a comparison of numerical and theoretical results. Phys Rev E 86(4):041125
Frasca M, Sharkey KJ (2016) Discrete-time moment closure models for epidemic spreading in populations of interacting individuals. J Theor Biol 399:13–21
Hadjichrysanthou C, Sharkey KJ (2015) Epidemic control analysis: designing targeted intervention strategies against epidemics propagated on contact networks. J Theor Biol 365:84–95
Hagenaars TJ, Donnelly CA, Ferguson NM (2004) Spatial heterogeneity and the persistence of infectious diseases. J Theor Biol 229(3):349–359
Harris TE (1974) Contact interactions on a lattice. Ann Probab 969–988
Holling CS (1973) Resilience and stability of ecological systems. Annu Rev Ecol Syst 4(1):1–23
Keeling MJ (1999) The effects of local spatial structure on epidemiological invasions. Proc R Soc B Biol Sci 266(1421):859–867
Keeling MJ, Eames KTD (2005) Networks and epidemic models. J R Soc Interface 2(4):295–307
Kephart JO, White SR, Chess DM (1993) Computers and epidemiology. IEEE Spectr 30(5):20–26
Kiss IZ, Miller JC, Simon PL (2017) Mathematics of epidemics on networks. Springer, Cham
Klein DR (1968) The introduction, increase, and crash of reindeer on St. Matthew island. J Wildlife Manag 350–367
Kryscio RJ, Lefevre C (2004) On the extinction of the SIS stochastic logistic epidemic, pp 213–228. Statistical Methods in Computer Security
Lajmanovich A, Yorke JA (1976) A deterministic model for gonorrhea in a nonhomogeneous population. Math Biosci 28(3–4):221–236
Liggett TM (2012) Interacting particle systems, vol 276. Springer, New York
Mata AS, Ferreira SC (2013) Pair quenched mean-field theory for the susceptible-infected-susceptible model on complex networks. Europhys Lett 103(4):48003
Mech LD (1966) The wolves of isle royale
Murray WH (1988) The application of epidemiology to computer viruses. Comput Sec 7(2):139–145
Nåsell I (1996) The quasi-stationary distribution of the closed endemic SIS model. Adv Appl Probab 28(3):895–932
Nåsell I (1999) On the quasi-stationary distribution of the stochastic logistic epidemic. Math Biosci 156(1–2):21–40
Nåsell I (1999) On the time to extinction in recurrent epidemics. J R Stat Soc Ser B (Stat Methodol) 61(2):309–330
Oppenheim I, Shuler KE, Weiss GH (1977) Stochastic theory of nonlinear rate processes with multiple stationary states. Phys A 88(2):191–214
Overton CE, Broom M, Hadjichrysanthou C, Sharkey KJ (2019) Methods for approximating stochastic evolutionary dynamics on graphs. J Theor Biol 468:45–59
Pakes AG (1987) Limit theorems for the population size of a birth and death process allowing catastrophes. J Math Biol 25(3):307–325
Parshani R, Carmi S, Havlin S (2010) Epidemic threshold for the susceptible-infectious-susceptible model on random networks. Phys Rev Lett 104(25):258701
Parsons RW, Pollett PK (1987) Quasistationary distributions for autocatalytic reactions. J Stat Phys 46(1–2):249–254
Pastor-Satorras R, Vespignani A (2001) Epidemic spreading in scale-free networks. Phys Rev Lett 86(14):3200
Pastor-Satorras R, Castellano C, Van Mieghem P, Vespignani A (2015) Epidemic processes in complex networks. Rev Mod Phys 87(3):925
Pollett PK, Kumar S (1987) On the long-term behaviour of a population that is subject to large-scale mortality or emigration. In: Proceedings of the 8th National Conference of the Australian Society for Operations Research, vol 196, p 207
Pollett PK (1988) On the problem of evaluating quasistationary distributions for open reaction schemes. J Stat Phys 53(5–6):1207–1215
Pollett PK (1995) The determination of quasistationary distributions directly from the transition rates of an absorbing Markov chain. Math Comput Model 22(10–12):279–287
Rock K, Brand S, Moir J, Keeling MJ (2014) Dynamics of infectious diseases. Rep Prog Phys 77(2):026602
Rogers T (2011) Maximum-entropy moment-closure for stochastic systems on networks. J Stat Mech Theory Exp 2011(05):P05007
Scheffer VB (1951) The rise and fall of a reindeer herd. Sci Monthly 73(6):356–362
Sharkey KJ (2011) Deterministic epidemic models on contact networks: correlations and unbiological terms. Theor Popul Biol 79(4):115–129
Sharkey KJ, Kiss IZ, Wilkinson RR, Simon PL (2015) Exact equations for SIR epidemics on tree graphs. Bull Math Biol 77(4):614–645
Van Mieghem P (2011) The N-intertwined SIS epidemic network model. Computing 93(2–4):147–169
Van Mieghem P, Omic J, Kooij R (2009) Virus spread in networks. IEEE/ACM Trans Netw 17(1):1–14
Wang Y, Chakrabarti D, Wang C, Faloutsos C (2003) Epidemic spreading in real networks: an eigenvalue viewpoint. In: Proceedings of the 22nd International Symposium on Reliable Distributed Systems, 2003, pp 25–34. IEEE
Wierman JC, Marchette DJ (2004) Modeling computer virus prevalence with a susceptible-infected-susceptible model with reintroduction. Comput Stat Data Anal 45(1):3–23
Wilkinson RR, Sharkey KJ (2013) An exact relationship between invasion probability and endemic prevalence for Markovian SIS dynamics on networks. PLoS ONE 8(7):e69028
Zachary WW (1977) An information flow model for conflict and fission in small groups. J Anthropol Res 33(4):452–473
Acknowledgements
CO and KS acknowledge support from EPSRC grant (EP/N014499/1). The authors would like to thank Ian Smith for use of the ARC Condor high throughput computing system at the University of Liverpool http://condor.liv.ac.uk/, which significantly sped up simulation of the stochastic models.
Author information
Authors and Affiliations
Contributions
CO, KS and RW created the project, performed the analysis and wrote the manuscript. JM created the project and performed the analysis. AL created the project.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Node-Level Individual-Based QSD Model
1.1 Derivation of Node-Level Conditional Distribution Equation
The rate of change in the probability that node i is infected in the QSD is given by the sum of the rates of change in the full system state probabilities for which node i is infected. That is, we have
where the terms are defined in Sect. 3. The numerator of the first term on the second line corresponds to the rate of change in the probability that node i is infected, which is given by \(\langle \dot{I_i} \rangle \) in Eq. (B3) in Appendix B. The summation in the second term corresponds to the probability that node i is infected, \(\langle I_i \rangle \). Therefore, we can write
Here, \((QP)_1\) is the rate at which the system enters the absorbing state. The system can only reach the absorbing state from a state with a single infected individual, in node j for example, which transitions to the all susceptible state at rate \(\gamma _j\). Therefore, \((QP)_1=\sum \limits _{j}\gamma _j\langle I_j S \rangle \), where we use \(\langle I_j S \rangle \) to denote the probability that node j is infected and all other nodes are susceptible. Using this along with Eq. (B3), we obtain
1.2 Proof that the Individual-Based Node-Level QSD Model is Invariant on \([0,1]^N\)
Proof
To prove that the model in Eq. (10) is invariant, we use the method from Lajmanovich and Yorke (1976). Along the boundaries to the set we are interested in, we either have \(\langle Y_i \rangle =0\) and \(\langle X_i \rangle =1\) or \(\langle Y_i \rangle =1\) and \(\langle X_i \rangle =0\). To show the system is invariant, we need to show that along these boundaries the trajectories do not point away from this set.
First consider \(\langle Y_i \rangle =0\). At this boundary, we have
If \(\langle Y_j \rangle \in [0,1]\), this cannot be negative, and therefore at \(\langle Y_i \rangle =0\) the trajectory in the i direction cannot leave the set \([0,1]^N\). Now, consider \(\langle Y_i\rangle =1\). We have
The product in this equation is in [0, 1] if \(\langle X_k \rangle \in [0,1]\) for all k. Therefore, this equation can never be positive, so along this boundary the trajectory cannot leave the set [0, 1]. Therefore, this model is invariant on \([0,1]^N\). \(\square \)
1.3 Proof of Theorem 1
Proof
Consider the node-level individual-based model (Eq. (10)) on a k-regular network with homogeneous transmission and recovery. If we start with a fully infected population, \(\langle Y_i \rangle \) will be equal for all i at every time point. Therefore, we can denote \(\langle I_i \rangle = a\) for all \(i \in \mathcal {V}\). We can write the rate of change in the node probabilities as
In the steady state, \(\dot{a}=0\). If we rule out \(a=0\), since Eq. (A2) is undefined for \(a=0\), then we obtain
We are therefore interested in solutions to \(f(a)=0\) with \(a \in [0,1]\), where
To see if a solution exists within this interval, we check the signs at the end points.
At \(a=1\)
the function is negative.
As a goes to zero
Therefore, as long as the transmission rate \(\tau \) is greater than zero there exists a solution to \(f(a)=0\) in the open interval (0, 1), since f(a) is non-singular on (0, 1).
We now need to show that our approximation to the expected number of infected individuals in the QSD is bounded below by one. This proof holds for all networks provided a solution exists satisfying \(\langle Y_i \rangle \in (0,1)\) for all i, which we have proven for k-regular networks. Consider the node-level individual-based model; i.e.
To approximate the QSD, we calculate \(\langle Y_i^*\rangle /(1-\prod \limits _k \langle X_k^* \rangle )\), where \(\langle Y^*\rangle \) and \(\langle X^*\rangle \) are steady-state solutions to (A3).
Let S be the sum of N independent Bernoulli random variables with success probabilities given by the vector \(\langle Y_i^* \rangle \) for \(i \in \{1,2,...,N\}\), which is a feasible solution of Eq. (A3). It is straightforward then that \(\mathbb {E}[S]=\sum _i\langle Y_i^*\rangle \), and we can write
So when we approximate the expected number infected in the QSD as
this cannot be less than 1. Therefore, provided a nonzero solution exists to Eq. (A3), the approximation to the expected number of infected individuals in the QSD is not less than 1. \(\square \)
Standard Approximate Models
Due to the prohibitive computational cost of solving the master equation (Eq. (1)), approximation methods are useful. In this section, we give an overview of the heterogeneous mean-field and pair-approximation methods, which can be interpreted as approximating the expected behaviour of the stochastic model. For detailed derivations and analysis of these models, see (Kiss et al. 2017).
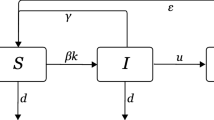
Under the heterogeneous mean-field model, we assume that: all individuals with the same degree can be treated identically, the status of neighbouring individuals is independent, \(\gamma _i=\gamma \) for all \(i \in \mathcal {V}\), and \(T_{ij}=\tau \) for all \(i,j \in \mathcal {V}\) with \(T_{ij}>0\) or \(T_{ji}>0\) (the network is assumed undirected for simplicity). The rate of change in the expected number of susceptible and infected individuals, stratified by the degree of the individual, is then approximated by Kiss et al. (2017)
where \([S_k]\) is the expected number of susceptible individuals of degree k at time t, \(|C_k|\) is the number of degree k nodes, \(|C_{k,l}|\) is the number of pairs involving a degree k node and a degree l node, and \(\mathcal {M}\) is the set of unique degrees on the network. Above, and throughout, we use ‘dot’ notation for derivatives with respect to time. Whilst the assumption of neighbouring individuals being independent is unrealistic, the resulting model has low computational cost, and hence, it is popular to study.
Instead of assuming statistical independence between individuals, models have been derived by writing down exact equations for the expected number of individuals and pairs:
where \([A_kB_l]\) is the expected number of pairs at time t, between degree k and l individuals in states A and B, respectively, and \([A_kB_lC_h]\) is the expected number of triples at time t, between degree k, l and h individuals, in states A, B and C, respectively.
Solving this system exactly involves deriving a full hierarchy of equations describing triples and quads and so on Eames and Keeling (2002), and therefore, we wish to approximate this system by closing the hierarchy early. This can be done by expressing triples as some function of pairs and individuals. To approximate the triples, we analyse the number of edges starting from a susceptible node, following (Eames and Keeling 2002; Kiss et al. 2017). The total number of SA edges (for \(A\in \{S,I\}\)) from a degree k node to a degree l node are \([S_kA_l]\). Since we have \([S_k]\) susceptible degree k nodes, we have approximately \([S_kA_l]/(k[S_k])\) edges leading from a given susceptible degree k node to a given degree l node in state A. Therefore, for a chosen susceptible degree k node the probability that two neighbours, with degree l and m, are in states A and B is given by \([A_lS_k][S_kB_m]/k^2[S_k]^2\). We have \(k(k-1)\) choices of the two neighbours, and \([S_k]\) choices of the susceptible node, and therefore, we can approximate the expected number of triples \([A_lS_kB_m]\) as
This approximation makes the homogeneity assumption that the neighbours of susceptible degree k nodes are interchangeable and the states of pairs are independent. Using this expression, the system of Eq. (B1) is closed at the level of pair terms, which allows the system to be solved with reasonably low computational cost.
These two models act at the population level, since they describe how the expected number of individuals with certain traits change. Following the motivation behind these models, node-level models have been developed that describe how the probability of individual nodes being infected change with time. Such models have been referred to as individual-based models (Sharkey 2011; Sharkey et al. 2015), node-level models (Overton et al. 2019), propagation models (Kiss et al. 2017) or quenched-mean field (Ferreira et al. 2012; Mata and Ferreira 2013). The advantage of such models over the population-level models is that we do not need to make any homogeneity assumptions about the underlying populations, and therefore, properties such as clustering, directed edges and degree heterogeneity are naturally captured. The downside, however, is that the computational cost scales with at least the number of nodes.
Under Markovian network-based SIS, the dynamics of individual nodes are given by Sharkey (2011)
where \(\langle A_i \rangle \) represents the probability \(P(\varSigma _i(t)=A)\) with \(A \in \{S,I\}\), and \(\langle A_i B_j \rangle \) represents the probability \(P(\varSigma _i(t)=A, \varSigma _j(t)=B)\) with \(A,B \in \{S,I\}\).
This equation exactly describes the rate of change for individual nodes in terms of pairs. Pairs of nodes are exactly described by
where \(\langle A_i B_j C_k \rangle \) represents the probability \(P(\varSigma _i(t)=A, \varSigma _j(t)=B, \varSigma _k(t)=C)\) with \(A,B,C \in \{S,I\}\). To solve this requires a hierarchy of equations up to full system size. Following similar logic to the population-level equations, this system can be approximated by making assumptions of statistical independence. Assuming that the states of individuals are independent, \(\langle S_iI_j\rangle \approx \langle S_i \rangle \langle I_j \rangle \), we can close the hierarchy at the level of individuals. Alternatively, we can assume independence at the level of pairs. The natural assumption of statistical independence to apply to pairs is that, given three nodes in a line, if the state of the central node is known, then the state of the outer two nodes is independent. For all triples in the system above, the central node in the configuration is always the centre node of a line between the two outer nodes. Therefore, if we consider the triple \(\langle A_i B_j C_k \rangle \), this can be approximated as a function of lower order terms by using conditional probabilities and assuming statistical independence. By the definition of conditional probabilities, we obtain
Assuming that the states of nodes i and k are independent given the state of node j, this becomes
which closes the hierarchy at the level of pairs. Other methods to approximate triples in terms of pairs and individuals have been proposed (Keeling 1999; Rogers 2011; Sharkey 2011); however, we do not consider them in this paper.
The population-level methods described above can be derived rigorously from the node-level methods (Sharkey 2011). In the exact case, we have
and
where \(A,B \in \{S,I\}\) and \(k_i\) is the degree of node i. Using this, the rate of change for the population-level terms can be derived. From this, we can also approximate the node-level quantities as
and
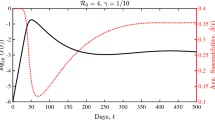
The models described here exhibit an epidemic threshold, above which the pathogen persists and below which the pathogen dies out (illustrated in Fig. 4 for the node-level pair-based model). For the population-level models and individual-based node-level model, above these thresholds a unique, globally stable steady-state exists (Keeling 1999; Keeling and Eames 2005; Kiss et al. 2017; Lajmanovich and Yorke 1976; Van Mieghem 2011). For the node-level pair-based model, the disease-free solution has been shown to become unstable as the transmission rate increases (Mata and Ferreira 2013), at which point we have shown that an endemic steady-state solution exists (Appendix C). Numerically, this endemic equilibrium appears to be unique and globally attracting, similar to the endemic solutions in the other models.
Comparing the standard pair-based model (Eqs. (B3) and (B4) with the closure from Eq. (B5)) with closures with the output of stochastic simulations on Zachary’s karate club network. We plot the expected number of infected individuals against time for each of the methods. As the figures move from left to right, the transmission rate increases. In the right-most figure, steady-like behaviour is observed in the stochastic model, since the expected time to extinction is very long
When comparing these models to the underlying stochastic process (e.g. Fig. 4), below the epidemic threshold the models accurately capture the expected number of infected individuals in the stochastic process. However, as the transmission rate increases (or recovery rate decreases), we pass the epidemic threshold and observe an endemic equilibrium that does not correspond to the stochastic process. Eventually, when the parameters are sufficiently above the epidemic threshold, the endemic steady-state solutions of these models can approximate the behaviour of the stochastic model for a long time, since the time to extinction of the pathogen is very long. Here, the stochastic process behaves similarly to the quasi-stationary distribution of the model; i.e. the expected long-term behaviour if extinction has not occurred.
Proof of Existence of an Endemic Steady-State for the Standard Pair-Based Model
Proof
In Lajmanovich and Yorke (1976), a theorem is proven regarding the existence of stable endemic solutions for ordinary differential equation epidemic models. Here, we demonstrate that the standard pair-based SIS model (Eqs. (B3) and (B4) with the closure from Eq. (B5) Mata and Ferreira 2013) satisfies the requirements for this proof and therefore, has a stable endemic steady-state.
Consider an ODE of the form
If the following statements hold, then there exists a threshold above which an endemic steady-state exists.
-
1.
A compact convex set C on the domain of N is positively invariant, with \(y=0 \in C\).
-
2.
\(\lim \limits _{y \rightarrow 0}||N(y)||/||y||=0\)
-
3.
There exists \(r>0\) and a real eigenvector w or \(A^T\) such that \((w \cdot y)\ge r||y|| \ \ \forall y \in C\)
-
4.
\((w\cdot N(y)) \le 0 \ \ \forall y \in C\)
-
5.
\(y=0\) is the largest positively invariant set contained in \(H=\{y \in C |(w\cdot N(y))=0\}\)
The first step is to write the pair-based model in the form (C1). The pair-based model is given by
where \(\langle S_i \rangle = 1- \langle I_i \rangle \), \(\langle I_iI_j \rangle = \langle I_j \rangle - \langle S_iI_j \rangle \) and \(\langle S_iS_j \rangle = \langle S_i \rangle - \langle S_iI_j \rangle \).
This can be rewritten as
Defining \(y_i=\langle I_i \rangle \) for \(1\le i\le N\) and \(y_i=\langle S_1I_{i-N}\rangle \) for \(N+1 \le i \le 2N\), \(y_i=\langle S_2I_{i-2N} \rangle \) for \(2N+1 \le i \le 3N\), and so on, we can write the pair-based model in the form of Eq. (C1). Compiling the linear terms into the matrix A, we see that A is only negative on the diagonal. The remaining nonlinear terms define the function N(y), which only assigns negative values to each input. Now, it is required to check if the properties hold.
Property (1.) holds because the system is invariant on the set \(C=\{0 \le \langle I_i \rangle \le 1; 0 \le \langle S_iI_j \rangle \le 1\}\). Property (2.) holds because as \(y \rightarrow 0\) the denominator of all terms, \(1-\langle I_i \rangle \), goes to one, and the numerator is of the form \(y_iy_j\), which goes to zero faster than \(y_i\) and \(y_j\). Property (3.) holds because A is irreducible since all the equations are coupled. Since A is only negative on the diagonal, by the Perron–Frobenius theorem, \(A^T\) must have an eigenvector w such that \(w_i>0\) for all i. Property (4.) holds because the function N(y) is negative, so \((w \cdot N(y))\le 0\), since \(w_i>0\) for all i. We now need to test property (5.).
Property (5.) If \( y \in H\), then \((w \cdot N(y))=0\). This implies that
and
for all pairs (i, j). If we assume that \(y \in H\) and \(y \ne 0\), then \(y_h \ne 0\) for some h. If we assume that \(y_h = \langle S_iI_j\rangle \ne 0\), then we must have \(\langle S_iI_k\rangle =0\), for all \(k \in \mathcal {N}_i\). Also, we require \(\langle S_jI_k\rangle =0\) for some k or \(\langle I_iS_j\rangle =0\). We now need to investigate whether such a state can be invariant.
Define \(S=\{i:y_i =0\}\) and \(S'=\{i:y_i \ne 0\}\), both of which are non-empty since \(y \ne 0\) and \(\langle S_iI_j\rangle =0\) for some pair (i.j) by the above argument. Since A is irreducible, there must exist a pair \(k \in S\) and \(h \in S'\) such that \(dy_k/dt\) depends on \(y_h\).
First assume that \(y_h = \langle S_iI_j\rangle \) and \(y_k=\langle I_i \rangle \). We have
If this state is invariant, then \(dy/dt=0\), which implies that \(dy_k/dt=0\) for all k. This can only be the case if \(\langle S_kI_j \rangle =0\) for all j. However, we have assumed that \(\langle S_iI_j \rangle \ne 0\), so this is not the case and \(dy_k/dt \ne 0\).
Now, assume \(y_k=\langle S_jI_i\rangle \), which gives
Since \(\langle I_jS_i \rangle /\langle S_i \rangle \le 1\), the sum of the last two terms cannot be negative. Therefore, if \(dy_k/dt=0\), we have \(\langle I_i \rangle =0\). However, as has been shown by assuming \(\langle I_i \rangle =0\), this case is not possible. Therefore, \(dy_k/dt \ne 0\). Therefore, if \(\langle S_iI_j \rangle \ne 0\) for some pair (i, j) and \(y \in H\), then this state cannot be invariant.
Now, assume that \(y_h = \langle I_i \rangle \in S'\) for some i, and consider \(y_k = \langle S_jI_i \rangle \in S\). Since \(\langle S_xI_y\rangle =0\) for all (x, y), we have
Since \(\langle I_i \rangle \in S'\), \(dy_k/dt \ne 0\). Therefore, there are no invariant sets in H such that \(y \ne 0\), and \(y=0\) is the largest positively invariant set in H.
This shows that properties 1-5 are satisfied for this model. Therefore, there exists a stable endemic steady-state above the epidemic threshold of the standard pair-based SIS model.
Population-Level Individual-Based QSD Model
The node-level equations give detailed insight into the dynamics of individual nodes in the QSD; however, the number of equations scales with N. To build approximations with a reduced number of equations, population-level models can be constructed for undirected networks. The rate of change in the expected number of infected individuals with a given degree, under the conditional distribution, is found by taking the sum over the probability that each node with this degree is infected
The numerator in the first term on the right-hand side is the rate of change that an individual is infected. Taking the sum over all nodes with the same degree, this gives the rate of change in the expected number of infected individuals with that degree, which is given by Eq. (B1). Taking the sum of \(\langle I_i \rangle \) over all nodes with the same degree gives the expected number of infected nodes with that degree. Therefore, assuming
where \([A_k]\) is the expected number of individuals with degree k in state A and \(\bar{T}_{kl}\) is the rate of transmission from a degree l to a degree k node, we obtain
where \([A_kB_l]\) is the expected number of pairs between individuals of degree k and degree l, in states A and B, respectively, and \(k_i\) is the degree of node i. Above, and throughout, all expected numbers are with respect to the standard probability measure P. Assuming that the states of individuals are independent, (D1) becomes
where \(|C_k|\) is the number of degree k nodes in the network and \(|C_{k,l}|\) is the number of pairs between degree k and degree l nodes. This equation is not closed, since the final term and the denominators depend on node-level quantities. However, from (B6) the node-level quantities can be approximated by assuming \(\langle S_j \rangle = [S_k]/|C_k|\), where k is the degree of node j. Therefore,
and
where k is the degree of node j. Multiplying Eq. (D2) by the number of degree k nodes, \(|C_k|\), we obtain the probability of a single degree k node being infected, which we denote \(\tilde{P}(I_k=1)\). Therefore, we obtain
To find a steady state, we need to find vectors \(\langle X \rangle ^*\) and \(\langle Y \rangle ^*\) satisfying
from which we can approximate the expected number of infected degree k individuals in the QSD by computing \( [Y_k]^*/(1-\prod _{l} (\frac{[X_l]^*}{|C_l|})^{|C_l|})\). We require \([Y_k]^* \in [0,|C_k|], [X_k]^*=|C_k|-[Y_k]^*\) for all i. Such a solution can be found by defining
and specifying that \([Y_k(0)]\in [0,|C_k|]\) for all k and calculating the steady-state. Any solution will be a valid solution, since Eq. (D3) is bounded such that \([Y_k]^* \in [0,|C_k|]\) for all k (this can be shown using a method similar to Appendix A.2).
Node-Level Pair-Based QSD Model
If we do not assume independence at the level of individuals, we need to find equations describing pair probabilities in the conditional distribution. We have
where \(\langle A_i \rangle \) is shorthand for the marginal probability \(P(\varSigma _i(t)=A)\) with \(A \in \{S,I\}\), \(\langle A_i B_j \rangle \) is shorthand for \(P(\varSigma _i(t)=A, \varSigma _j(t)=B)\) with \(A,B \in \{S,I\}\), \(\langle A_i B_j C_k\rangle \) is shorthand for \(P(\varSigma _i(t)=A, \varSigma _j(t)=B,\varSigma _k(t)=C)\) with \(A,B,C \in \{S,I\}\), and \(\langle I_j S \rangle \) is shorthand for \(P(\varSigma _j=I,\varSigma _k=S \text{ for } \text{ all } k \ne j)\). We can simplify this system by assuming statistical independence at the level of pairs.
As described in Appendix B, we approximate the triples in terms of pairs and individuals by assuming
Under this assumption, Eq. (E1) becomes
Note that \(\langle S_i\rangle = 1-\langle I_i \rangle \), \(\langle I_iI_j\rangle = \langle I_j\rangle - \langle S_iI_j\rangle \) and \(\langle S_iS_j\rangle = \langle S_i\rangle -\langle S_iI_j\rangle \). Both \(\langle I_jS \rangle \) and the ground state probability, \(P_1\), are full system size, and therefore, following (Frasca and Sharkey 2016; Sharkey et al. 2015), a natural pair approximation for these is
and
In the QSD, both the pair level and individual level conditional probabilities are in a steady-state, so both equations in Eq. (E1) are equal to zero. Therefore, to find the approximation to the QSD under the pair level independence assumption, we need to find vectors \(\langle X^*\rangle \), \(\langle Y^* \rangle \), and matrices \(\langle XX^* \rangle \),\(\langle XY^* \rangle \), and \(\langle YY^* \rangle \) satisfying,
which, once solved, can be used to find the probability that i is infected in the QSD by computing \(\langle Y_i \rangle ^*/(1-\langle \sigma _1\rangle ^*)\). However, we require solutions \(\langle Y_i \rangle ^*\) and \(\langle X_iY_j \rangle ^* \in [0,1]\) which satisfy \(\langle X_i \rangle ^*=1-\langle Y_i \rangle ^*\) for all i and \(\langle X_iX_j \rangle = \langle X_i \rangle -\langle X_iY_j \rangle \), and \(\langle Y_iY_j\rangle =\langle Y_j\rangle - \langle X_iY_j \rangle \) for all i, j in order to be valid solutions to our original problem.
By calculating the steady-state of the system,
where
and
we can approximate the probability that i is infected in the QSD by computing \(\lim _{t \rightarrow \infty } \langle Y_i(t) \rangle ^*/(1-\langle \sigma _0(t) \rangle ^*)\).
Population-Level Pair-Based QSD Model
To obtain a population-level pair-based model, we sum over nodes with the same degree (and pairs of nodes with same pair of degrees); i.e.
where \([A_kB_lC_h]\) is the expected number of triples between degree k, degree l and degree h individuals in states A, B and C, respectively.
As described in Appendix B, we can express the triple terms as
We can set equations (F1) to zero and use the approximation (F2) to find equations describing the QSD.
A solution to the resulting system can be found by finding an steady-state of
where \(\tilde{P}(Y_l=1)=|C_l|\langle Y_iX \rangle \) for some i with \(k_i=l\). Here,
which requires node-level terms. We can approximate this by population-level quantities using
and
based on the discussion in Appendix B. This gives
To approximate the ground state, recall that in the previous section we have shown that a natural approximation to the ground state probability under the assumption of pair level independence is
Using equations (F4) and (F5), we can approximate this in terms of population level quantities, which yields
By substituting Eqs. (F7) and (F6) into Eq. (F3), we obtain a closed system of equations.
Rights and permissions
About this article
Cite this article
Overton, C.E., Wilkinson, R.R., Loyinmi, A. et al. Approximating Quasi-Stationary Behaviour in Network-Based SIS Dynamics. Bull Math Biol 84, 4 (2022). https://doi.org/10.1007/s11538-021-00964-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11538-021-00964-7