Abstract
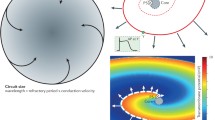
The aim of the study is to understand in depth the meaning of “reentry”, and to decipher if and how it can lead to malfunctions of the heart and possibly of the brain. A simple model is used to reveal the mechanism by which a single pulse of action potential rotating around a ring of excitable medium, the latter simulating a reentry circuit, can generate spirals (single and/or double) when the pulse can emerge from and develop outside the ring. Two mechanisms of spiral generation are demonstrated: (1) a mechanism in which a source of single spirals is created at the contact with the core soon after the pulse freeing action, their chirality being due to the sense of the preceding pulse rotation. Interestingly, these spirals, adhering to the core, become “double-spiral patterns” while leaving behind the seeds of the new single spirals. (2) A second possible mechanism, similar to the known “arms encountering methods”, in which a double spiral (a figure of eight) is repeatedly created on the other side of the core. Similar procedures are assumed to occur in the heart, leading to tachycardia and fibrillation and possibly in the brain leading to epilepsy. The exact processes of the hitherto assumed spiral generations by reentry were established. The novel deep understanding of the mechanisms involved in these processes can lead to new methods of treating heart fibrillation (e.g., by judicial ablation).





Similar content being viewed by others
References
Bentes C (2017) Epileptic manifestations in stroke patients treated with intravenous alteplase. Eur J Neurol 24:755–761. https://doi.org/10.1111/ene.13292
Berenfeld O, Jalife J (2016) Mechanisms of atrial fibrillation: rotors, ionic determinants, and excitation frequency. Heart Fail Clin 12:167–178. https://doi.org/10.1016/j.hfc.2015.08.014
Biton Y, Rabinovitch A, Aviram I, Braunstein D (2009) Two mechanisms of spiral-pair-source creation in excitable media. Phys Lett A 373:1762–1767
Biton Y, Rabinovitch A, Braunstein D, Friedman M, Aviram I (2010) Sharp corners as sources of spiral pairs. Phys Lett A 374:2248–2253
Brugada J, Boersma L, Allessie M, Navarro-Lopez F (1993) The complexity of mechanisms in ventricular tachycardia pacing and clinical electrophysiology. PACE 16:680–686
Clayton RH (2011) Models of cardiac tissue electrophysiology: progress, challenges and open questions. Prog Biophys Mol Biol 104:22–48. https://doi.org/10.1016/j.pbiomolbio.2010.05.008
Di Diego JM, Antzelevitch C (2014) Acute myocardial ischemia: cellular mechanisms underlying ST segment elevation. J Electrocardiol 47:486–490. https://doi.org/10.1016/j.jelectrocard.2014.02.005
El-Sherif N, Smith RA, Evans K (1981) Canine ventricular arrhythmias in the late myocardial infarction period. 8. Epicardial mapping of reentrant circuits. Circ Res 49:255–265
Friedman M, Ovsyshcher IE, Fleidervish I, Crystal E, Rabinovitch A (2004) Stimulation of unidirectional pulses in excitable systems. Phys Rev E Stat Nonlinear Soft Matter Phys 70:041903. https://doi.org/10.1103/PhysRevE.70.041903
Glass L, Mackey MC (1988) From clocks to chaos the rhythms of life. Princeton University Press, Princeton
Huang X, Troy WC, Yang Q, Ma H, Laing CR, Schiff SJ, Wu JY (2004) Spiral waves in disinhibited mammalian neocortex. J Neurosci 24:9897–9902. https://doi.org/10.1523/JNEUROSCI.2705-04
Josephson CB, Sandy S, Jette N, Sajobi TT, Marshall D, Wiebe S (2016) A systematic review of clinical decision rules for epilepsy. Epilepsy Behav 57:69–76. https://doi.org/10.1016/j.yebeh.2016.01.019
Kotecha D, Piccini JP (2015) Atrial fibrillation in heart failure: what should we do? Eur Heart J 36:3250–3257. https://doi.org/10.1093/eurheartj/ehv513
Leask A (2015) Getting to the heart of the matter: new insights into cardiac fibrosis. Circ Res 116:1269–1276. https://doi.org/10.1161/CIRCRESAHA.116.305381
Mills WR, Mal N, Forudi F, Popovic ZB, Penn MS, Laurita KR (2006) Optical mapping of late myocardial infarction in rats. Am J Physiol Heart Circ Physiol 290:H1298–1306. https://doi.org/10.1152/ajpheart.00437
Morris C, Lecar H (1981) Voltage oscillations in the barnacle giant muscle fiber. Biophys J 35:193–213. https://doi.org/10.1016/S0006-3495(81)84782-0
Nattel S (2016) How does fibrosis promote atrial fibrillation persistence: in silico findings, clinical observations, and experimental data. Cardiovasc Res 110:295–297. https://doi.org/10.1093/cvr/cvw092
Panfilov AV, Keener JP (1993) Effects of high frequency stimulation on cardiac tissue with an inexcitable obstacle. J Theor Biol 163:439–448. https://doi.org/10.1006/jtbi.1993.1129
Quail T, Shrier A, Glass L (2014) Spatial symmetry breaking determines spiral wave chirality. Phys Rev Lett 113:158101. https://doi.org/10.1103/PhysRevLett.,113.158101
Rabinovitch A, Biton Y, Gutman M, Aviram I (2009) Dynamics of a spiral pair source and its interaction with plane waves. Comput Biol Med 39:405–411. https://doi.org/10.1016/j.compbiomed.2009.01.015
Rabinovitch A, Biton Y, Braunstein D, Friedman M, Aviram I (2012) New generation methods of spiral-pairs and 3D patterns. In: Jobbgy E (ed) 5th European Congress of the International Federation for Medical and Biological Engineering. Springer, pp 371–373
Ribeiro TL, Copelli M (2008) Deterministic excitable media under Poisson drive: power law responses, spiral waves, and dynamic range. Phys Rev E Stat Nonlinear Soft Matter Phys 77:051911. https://doi.org/10.1103/PhysRevE.77.051911
Rinzel J, Ermentrout GB (1998) Methods in neuronal modeling: from ions to networks. MIT Press, Cambridge
Sato D, Shiferaw Y, Garfinkel A, Weiss JN, Qu Z, Karma A (2006) Spatially discordant alternans in cardiac tissue: role of calcium cycling. Circ Res 99:520–527. https://doi.org/10.1161/01.RES.0000240542.03986.e7
Schotten U, Dobrev D, Platonov PG, Kottkamp H, Hindricks G (2016) Current controversies in determining the main mechanisms of atrial fibrillation. J Intern Med 279:428–438. https://doi.org/10.1111/joim.12492
Seitz J (2017) AF ablation guided by spatiotemporal electrogram dispersion without pulmonary vein isolation: a wholly patient-tailored approach. J Am Coll Cardiol 69:303–321. https://doi.org/10.1016/j.jacc.2016.10.065
Sneyd J, Keener JP (2009) Mathematical physiology. Springer, New York
Vigmond E, Pashaei A, Amraoui S, Cochet H, Hassaguerre M (2016) Percolation as a mechanism to explain atrial fractionated electrograms and reentry in a fibrosis model based on imaging data. Heart Rhythm 13:1536–1543. https://doi.org/10.1016/j.hrthm.2016.03.019
Vinet A, Comtois P (2005) A multistability of reentrant rhythms in an ionic model of a two-dimensional annulus of cardiac tissue. Phys Rev E Stat Nonlinear Soft Matter Phys 72:051927. https://doi.org/10.1103/PhysRevE.72.051927
Viventi J (2011) Flexible, foldable, actively multiplexed, high-density electrode array for mapping brain activity in vivo. Nat Neurosci 14:1599–1605. https://doi.org/10.1038/nn.2973
Yu-Ye L, Bing J, Hua-Guang G, Shu-Cheng A (2012) Parameter diversity induced multiple spatial coherence resonances and spiral waves in neuronal network with and without noise. Commun Theor Phys 57:817
Zahid S (2016) Patient-derived models link re-entrant driver localization in atrial fibrillation to fibrosis spatial pattern. Cardiovasc Res 110:443–454. https://doi.org/10.1093/cvr/cvw073
Zarubin D, Zhuchkova E, Schreiber S (2012) Effects of cooperative ion–channel interactions on the dynamics of excitable membranes. Phys Rev E Stat Nonlinear Soft Matter Phys 85:061904. https://doi.org/10.1103/PhysRevE.85.061904
Acknowledgements
The authors would like to thank Prof. R. Thieberger and Mrs. S. Rabinovitch for helpful discussions.
Author information
Authors and Affiliations
Corresponding author
Appendix: The Morris Lecar Constants for the Heart Syncytium or for Coupled Neurons
Appendix: The Morris Lecar Constants for the Heart Syncytium or for Coupled Neurons
See Table 1.
Rights and permissions
About this article
Cite this article
Rabinovitch, A., Aviram, I., Biton, Y. et al. Reentry as an Origin for Rotors. Bull Math Biol 80, 3023–3037 (2018). https://doi.org/10.1007/s11538-018-0506-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-018-0506-3