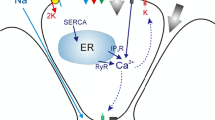
Abstract
We develop a mathematical model of a salivary gland acinar cell with the objective of investigating the role of two \(\mathrm{Cl}^-/\mathrm{HCO}_3^-\) exchangers from the solute carrier family 4 (Slc4), Ae2 (Slc4a2) and Ae4 (Slc4a9), in fluid secretion. Water transport in this type of cell is predominantly driven by \(\mathrm{Cl}^-\) movement. Here, a basolateral \(\mathrm{Na}^+/ \mathrm{K}^+\) adenosine triphosphatase pump (NaK-ATPase) and a \(\mathrm{Na}^+\)–\(\mathrm{K}^+\)–\(2 \mathrm{Cl}^-\) cotransporter (Nkcc1) are primarily responsible for concentrating the intracellular space with \(\mathrm{Cl}^-\) well above its equilibrium potential. Gustatory and olfactory stimuli induce the release of \(\mathrm{Ca}^{2+}\) ions from the internal stores of acinar cells, which triggers saliva secretion. \(\mathrm{Ca}^{2+}\)-dependent \(\mathrm{Cl}^-\) and \(\mathrm{K}^+\) channels promote ion secretion into the luminal space, thus creating an osmotic gradient that promotes water movement in the secretory direction. The current model for saliva secretion proposes that \(\mathrm{Cl}^-/ \mathrm{HCO}_3^-\) anion exchangers (Ae), coupled with a basolateral \(\mathrm{Na}^+/\hbox {proton}\) (\(\hbox {H}^+\)) (Nhe1) antiporter, regulate intracellular pH and act as a secondary \(\mathrm{Cl}^-\) uptake mechanism (Nauntofte in Am J Physiol Gastrointest Liver Physiol 263(6):G823–G837, 1992; Melvin et al. in Annu Rev Physiol 67:445–469, 2005. https://doi.org/10.1146/annurev.physiol.67.041703.084745). Recent studies demonstrated that Ae4 deficient mice exhibit an approximate \(30\%\) decrease in gland salivation (Peña-Münzenmayer et al. in J Biol Chem 290(17):10677–10688, 2015). Surprisingly, the same study revealed that absence of Ae2 does not impair salivation, as previously suggested. These results seem to indicate that the Ae4 may be responsible for the majority of the secondary \(\mathrm{Cl}^-\) uptake and thus a key mechanism for saliva secretion. Here, by using ‘in-silico’ Ae2 and Ae4 knockout simulations, we produced mathematical support for such controversial findings. Our results suggest that the exchanger’s cotransport of monovalent cations is likely to be important in establishing the osmotic gradient necessary for optimal transepithelial fluid movement.









Similar content being viewed by others
References
Arreola J, Begenisich T, Nehrke K, Nguyen HV, Park K, Richardson L, Yang B, Schutte BC, Lamb FS, Melvin JE (2002) Secretion and cell volume regulation by salivary acinar cells from mice lacking expression of the Clcn3 \(\text{ Cl }^-\) channel gene. J Physiol 545(1):207–216. https://doi.org/10.1113/jphysiol.2002.021980
Benjamin B, Johnson E (1997) A quantitative description of the Na–K–2Cl cotransporter and its conformity to experimental data. Am J Physiol Ren Physiol 273(3):F473–F482
Bruce J, Shuttleworth T, Giovannucci D, Yule D (2002) Phosphorylation of inositol 1, 4, 5-trisphosphate receptors in parotid acinar cells. A mechanism for the synergistic effects of cAMP on \(\text{ Ca }^{2+}\) signaling. J Biol Chem 277(2):1340–1348
Catalán MA, Nakamoto T, Melvin JE (2009) The salivary gland fluid secretion mechanism. J Med Invest 56(Supplement):192–196. https://doi.org/10.2152/jmi.56.192
Crampin EJ, Smith NP, Langham AE, Clayton RH, Orchard CH (2006) Acidosis in models of cardiac ventricular myocytes. Philos Trans R Soc Lond A Math Phys Eng Sci 364(1842):1171–1186. https://doi.org/10.1098/rsta.2006.1763
DeBiévre P, Valkiers S, Schaefer F, Peiser H, Seyfried P (1994) High-accuracy isotope abundance measurements for metrology. PTB Mitt 104(4):225–236
Delporte C, Steinfeld S (2006) Distribution and roles of aquaporins in salivary glands. Biochim Biophys Acta Biomembr 1758(8):1061–1070
Dupont G, Falcke M, Kirk V, Sneyd J (2016) Models of calcium signalling. Interdisciplinary applied mathematics, vol 43. Springer, Berlin
Evans RL, Park K, Turner RJ, Watson GE, Nguyen HV, Dennett MR, Hand AR, Flagella M, Shull GE (2000) Severe impairment of salivation in \(\text{ Na }^{+}/\text{ K }^{+}/2\text{ Cl }^{-}\) cotransporter (NKCC1)-deficient mice. J Biol Chem 275(35):26720–26726. https://doi.org/10.1074/jbc.M003753200
Falkenberg C, Jakobsson E (2010) A biophysical model for integration of electrical, osmotic, and ph regulation in the human bronchial epithelium. Biophys J 98(8):1476–1485. https://doi.org/10.1016/j.bpj.2009.11.045
Foskett JK, Melvin JE (1989) Activation of salivary secretion: coupling of cell volume and \([\text{ Ca }^{2+}]_i\) in single cells. Science 244(4912):1582–1585
Frizzell R, Hanrahan J (2012) Physiology of epithelial chloride and fluid secretion. Cold Spring Harb Perspect Med 2(6):a009563. https://doi.org/10.1101/cshperspect.a009563
Gin E, Crampin EJ, Brown DA, Shuttleworth TJ, Yule DI, Sneyd J (2007) A mathematical model of fluid secretion from a parotid acinar cell. J Theor Biol 248(1):64–80
Gordon R (1982) Essentials of human physiology, vol 55. Year Book Medical Publishers, Chicago
Grinstein S, Foskett JK (1990) Ionic mechanisms of cell volume regulation in leukocytes. Annu Rev Physiol 52(1):399–414. https://doi.org/10.1146/annurev.ph.52.030190.002151
Jayaraman S, Song Y, Vetrivel L, Shankar L, Verkman A (2001) Noninvasive in vivo fluorescence measurement of airway-surface liquid depth, salt concentration, and pH. J Clin Investig 107(3):317–324. https://doi.org/10.1172/JCI11154
Kunkel P, Mehrmann VL (2006) Differential-algebraic equations: analysis and numerical solution. European Mathematical Society, Zurich
Lau K, Case R (1988) Evidence for apical chloride channels in rabbit mandibular salivary glands. Pflügers Arch 411(6):670–675. https://doi.org/10.1007/BF00580864
Locker D (1995) Xerostomia in older adults: a longitudinal study. Gerodontology 12(1):18–25. https://doi.org/10.1111/j.1741-2358.1995.tb00125.x
Maclaren OJ, Sneyd J, Crampin EJ (2012) Efficiency of primary saliva secretion: an analysis of parameter dependence in dynamic single-cell and acinus models, with application to aquaporin knockout studies. J Membr Biol 245(1):29–50. https://doi.org/10.1007/s00232-011-9413-3
Mangos J, McSherry NR, Nousia-Arvanitakis S, Irwin K (1973) Secretion and transductal fluxes of ions in exocrine glands of the mouse. Am J Physiol 225(1):18–24
Martinez J, Cassity N (1983) Effect of transport inhibitors on secretion by perfused rat submandibular gland. Am J Physiol 245(5 Pt 1):G711–6. https://doi.org/10.1016/j.archoralbio.2010.06.012
Melvin JE, Yule D, Shuttleworth T, Begenisich T (2005) Regulation of fluid and electrolyte secretion in salivary gland acinar cells. Annu Rev Physiol 67:445–469. https://doi.org/10.1146/annurev.physiol.67.041703.084745
Mignogna M, Fedele S, Russo LL, Muzio LL, Wolff A (2005) Sjögren’s syndrome: the diagnostic potential of early oral manifestations preceding hyposalivation/xerostomia. J Oral Pathol Med 34(1):1–6. https://doi.org/10.1111/j.1600-0714.2004.00264.x
Moldover M, Trusler J, Edwards T, Mehl J, Davis R (1988) Measurement of the universal gas constant R using a spherical acoustic resonator. Phys Rev Lett 60(4):249
Nauntofte B (1992) Regulation of electrolyte and fluid secretion in salivary acinar cells. Am J Physiol Gastrointest Liver Physiol 263(6):G823–G837
Nguyen HV, Stuart-Tilley A, Alper SL, Melvin JE (2004) \(\text{ Cl }^-/\text{ HCO }_3^-\) exchange is acetazolamide sensitive and activated by a muscarinic receptor-induced \([\text{ Ca }^{2+}]_i\) increase in salivary acinar cells. Am J Physiol Gastrointest Liver Physiol 286(2):G312–G320. https://doi.org/10.1016/j.ceca.2014.01.005
Niedermeier W, Matthaeus C, Meyer C, Staar S, Müller RP, Schulze HJ (1998) Radiation-induced hyposalivation and its treatment with oral pilocarpine. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 86(5):541–549. https://doi.org/10.1016/S1079-2104(98)90343-2
Novak I, Young J (1986) Two independent anion transport systems in rabbit mandibular salivary glands. Pflügers Arch 407(6):649–656. https://doi.org/10.1007/BF00582647
Ogawa Y, Fernley R, Ito R, Ijuhin N (1998) Immunohistochemistry of carbonic anhydrase isozymes VI and II during development of the rat salivary glands. Histochem Cell Biol 110(1):81–88. https://doi.org/10.1007/s004180050268
Palk L, Sneyd J, Shuttleworth TJ, Yule DI, Crampin EJ (2010) A dynamic model of saliva secretion. J Theor Biol 266(4):625–640. https://doi.org/10.1016/j.jtbi.2010.06.027
Pedersen G, Petersen O (1973) Membrane potential measurement in parotid acinar cells. J Physiol 234(1):217. https://doi.org/10.1113/jphysiol.1973.sp010342
Peña-Münzenmayer G, Catalán MA, Kondo Y, Jaramillo Y, Liu F, Shull GE, Melvin JE (2015) Ae4 (Slc4a9) anion exchanger drives \(\text{ Cl }^-\)uptake-dependent fluid secretion by mouse submandibular gland acinar cells. J Biol Chem 290(17):10677–10688
Peña-Münzenmayer G, George AT, Shull GE, Melvin JE, Catalàn MA (2016) Ae4 (Slc4a9) is an electroneutral monovalent cation-dependent \(\text{ Cl }^-/\text{ HCO }_3^-\) exchanger. J Gen Physiol 147(5):423–436. https://doi.org/10.1085/jgp.201611571
Roussa E (2011) Channels and transporters in salivary glands. Cell Tissue Res 343(2):263–287. https://doi.org/10.1007/s00441-010-1089-y
Roussa E, Alper S, Thévenod F (2001) Immunolocalization of anion exchanger AE2, \(\text{ Na }^{+}/\text{ H }^+\) exchangers NHE1 and NHE4, and vacuolar type \(\text{ H }^+-\text{ ATP }\)ase in rat pancreas. J Histochem Cytochem 49(4):463–474. https://doi.org/10.1155/2013/840121
Sharp K, Crampin E, Sneyd J (2015) A spatial model of fluid recycling in the airways of the lung. J Theor Biol 382:198–215. https://doi.org/10.1016/j.jtbi.2015.06.050
Silva P, Stoff J, Field M, Fine L, Forrest J, Epstein F (1977) Mechanism of active chloride secretion by shark rectal gland: role of Na–K-ATPase in chloride transport. Am J Physiol Ren Physiol 233(4):F298–F306. https://doi.org/10.1038/ki.1996.224
Smith N, Crampin E (2004) Development of models of active ion transport for whole-cell modelling: cardiac sodium–potassium pump as a case study. Prog Biophys Mol Biol 85(2):387–405. https://doi.org/10.1016/j.pbiomolbio.2004.01.010
Soltoff S, McMillian M, Cantley L, Cragoe E, Talamo B (1989) Effects of muscarinic, alpha-adrenergic, and substance P agonists and ionomycin on ion transport mechanisms in the rat parotid acinar cell. the dependence of ion transport on intracellular calcium. J Gen Physiol 93(2):285–319. https://doi.org/10.1085/jgp.93.2.285
Takahata T, Hayashi M, Ishikawa T (2003) SK4/IK1-like channels mediate TEA-insensitive, \(\text{ Ca }^{2+}\)-activated \(\text{ K }^+\) currents in bovine parotid acinar cells. Am J Physiol Cell Physiol 284(1):C127–C144. https://doi.org/10.1152/ajpcell.00250.2002
Thaysen JH, Thorn NA, Schwartz IL (1954) Excretion of sodium, potassium, chloride and carbon dioxide in human parotid saliva. Am J Physiol 178(1):155–159
Wang H, Yan Y, Kintner D, Lytle C, Sun D (2003) GABA-mediated trophic effect on oligodendrocytes requires Na–K–2Cl cotransport activity. J Neurophysiol 90(2):1257–1265. https://doi.org/10.1523/JNEUROSCI.2569-04.2004
Young JA (1968) Microperfusion investigation of chloride fluxes across the epithelium of the main excretory duct of the rat submaxillary gland. Pflügers Arch 303(4):366–374
Young J, Schögel E (1966) Micropuncture investigation of sodium and potassium excretion in rat submaxillary saliva. Pflügers Arch 291(1):85–98. https://doi.org/10.1007/BF00362654
Yusuke I, Hiroshi NC, Yoshizaki M, Gen P (1973) Evidence for the osmotic flow across dog submaxillary gland epithelia as a cause of salivary secretion. Jpn J Physiol 23(6):635–644. https://doi.org/10.2170/jjphysiol.23.635
Acknowledgements
This study was supported by the Marsden Fund of the Royal Society of New Zealand (ES and JS), NIDCR Grant R01 DE019245-06A1 (ES and JS), the Intramural Research Program of the National Institute of Dental and Craniofacial Research, National Institutes of Health (JEM) and Grant FONDECYT # 11150454 (GPM).
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix
\(\hbox {Na}^+\)–\(\hbox {K}^+\)–\(2\hbox {Cl}^-\) Cotransporter (Nkcc1)
The Nkcc1 cotransporter expression is ubiquitous in nearly all cells and secretory epithelia (Wang et al. 2003). The Nkcc1-mediated-\(\hbox {Cl}^-\) uptake mechanism is a secondary active transport, i.e. the energy required for its activity comes indirectly from ATP hydrolysis. The Nkcc1 model we use was first constructed by Benjamin and Johnson (1997). The model assumes equilibrium ion binding, binding symmetry and identity of \(\hbox {Cl}^-\) binding sites. This results in a 10 state model. Palk et al. (2010) and Gin et al. (2007) simplified it to a two-state model that assumes simultaneous binding and unbinding of \(\hbox {Cl}^-\), \(\hbox {K}^+\) and \(\hbox {Na}^+\). The reaction occurs as follows:
The steady-state flux is given by
where \(\alpha _{\mathrm{Nkcc1}}\) is the density of the cotransporter.
Nkcc1 | Description | Value | Units |
|---|---|---|---|
\(\alpha _{\mathrm{Nkcc1}}^{\tiny {\dagger }}\) | Membrane density | 2.15 | \(\hbox {amol}/\upmu \hbox {m}^3\) |
\(a_1\) | Rate | 157.5 | \(\hbox {s}^{-1}\) |
\(a_2\) | Rate | \(2.0096\times 10^7\) | \(\hbox {mM}^{-4}\) \(\hbox {s}^{-1}\) |
\(a_3\) | Rate | 1.0306 | \(\hbox {s}^{-1}\) |
\(a_4\) | Rate | \(1.3852\times 10^6\) | \(\hbox {mM}^{-4}\ \hbox {s}^{-1}\) |
\(\hbox {Na}^+/\hbox {K}^+\) ATPase Pump (NaK)
The \(\hbox {Na}^+/\hbox {K}^+\) ATPase pump extrudes 3 \(\hbox {Na}^+\) ions while introducing 2 \(\hbox {K}^+\) ions against their electrochemical gradients at the expense of hydrolysing an ATP molecule per cycle. The net reaction for the pump cycle is
where \(\text {P}_i\) represents the intracellular concentration of phosphate (a result of ATP conversion to ADP). Smith and Crampin (2004) constructed a mathematical model of the NaK-ATPase pump intended to be used as a component in whole-cell myocyte modelling with the objective to predict pump function and whole myocyte behaviour when cellular metabolism is compromised. Palk et al. (2010) reduced the model to two states:
where I refers to an ‘Inside’ state and O to an ‘Outside’ state. The simplification assumes that external \(\hbox {Na}^+\) and internal \(\hbox {K}^+\) ions simultaneously bind and unbind and are supplied at a constant rate. In addition, the forward reaction rates are higher than the reverse and that the steady-state flux through the pump is given by
where \(\alpha _{\mathrm{NaK}}\) is the density of the pump.
NaK-ATPase | Description | Value | Units |
|---|---|---|---|
\(\alpha _{\mathrm{NaK}}^{\dagger }\) | Membrane density | 4.84 | \(\hbox {amol}/\upmu \hbox {m}^3\) |
r | Rate | \(1.305\times 10^6\) | \(\hbox {mM}^{-3}\ \hbox {s}^{-1}\) |
\(\alpha _1\) | Half saturation | 0.641 | \(\hbox {mM}^{-1}\) |
\(\hbox {Ca}^{2+}\)-Activated-\(\hbox {K}^+\) Channel (CaKC)
Takahata et al. (2003) characterised the biophysical and pharmacological properties of native TEA-insensitive \(\hbox {Ca}^{2+}\) activated currents in bovine parotid acinar cells. In their study, they developed a mathematical model that describes the basolateral CaKC current. The open probability of the channel is given by
where \(\eta _2=2.54\) is the Hill coefficient and \(K_{\mathrm{CaKC}}\) the dissociation constant (a function of the potential difference across the membrane). We used the value found by Palk et al. (2010) of \(0.182\ \upmu \hbox {M}\), as we require a small open probability at steady-state \(\hbox {Ca}^{2+}\) concentrations. The flux is defined as
with Nernst potential
where \(z^\text {K}=+1\), the ion’s valence.
\(\hbox {Ca}^{2+}\)-Activated-\(\hbox {Cl}^-\) Channel (CaCC)
Frizzell and Hanrahan (2012) demonstrated that the apical CaCC channels are activated at low \(\hbox {Ca}^{2+}\) concentrations by membrane depolarisation and when \(\hbox {Ca}^{2+}\) reaches micromolar concentrations. As a simplification, we used a model similar to that of Takahata et al. (2003). Previous mathematical models for the acinar cell have used the model of Arreola et al. (2002). We found that there is no qualitative difference between the models. The CaCC model predicts a large maximum single channel conductance. With a Hill coefficient of \(\eta _1=1.46\) and a dissociation constant of \(K_{\mathrm{CaCC}}=0.26 \,\upmu \hbox {M}\). The open probability is given by
In this way, the flux is defined as
with Nernst potential
where \(z^{Cl}=-1\), the ion’s valence.
SLC4A2 Anion Exchanger (Ae2)
We use a model created by Falkenberg and Jakobsson (2010). It relies on a concentration gradient, i.e. it uses the \(\hbox {HCO}_3^-\) gradient to pump \(\hbox {Cl}^-\) into the cell. The energy required for this exchange is derived from the NaK-ATPase activity (Roussa et al. 2001). Its flux depends on the ionic concentrations in the cytoplasm and interstitium, the number of binding sites and the half saturation constants. The model evaluates the product of two terms and the exchanger’s conductance which is proportional to the density of active membrane proteins. The contribution from the concentrations and binding site properties represented by the Michaelis–Menten terms. Additionally as a simplification, we include the difference of the transporter in reverse which renders the exchanger bidirectional.
Ae2 | Description | Value | Units |
|---|---|---|---|
\(G_{\mathrm{Ae2}}^{\tiny {\dagger }}\) | Ae2 activity | 0.01807 | \(\text {fmol}/\text {s}\) |
\(K_{\mathrm{Cl}}\) | Half saturation | 5.6 | mM |
\(K_\mathrm{B}\) | Half saturation | \(10^{4}\) | mM |
\(\hbox {Na}^+/\hbox {H}^+\) Exchanger (Nhe1)
Similarly to the Ae2 exchanger, our model for the Nhe1 exchanger is based on Falkenberg and Jakobsson (2010). Its flux is given by
Nhe1 | Description | Value | Units |
|---|---|---|---|
\(G_{\mathrm{Nhe1}}^{\tiny {\dagger }}\) | Nhe1 activity | 0.0305 | \(\text {fmol}/\text {s}\) |
\(K_\mathrm{H}\) | Half saturation | \(4.5\times 10^{-4}\) | mM |
\(K_{\mathrm{Na}}\) | Half saturation | 15 | mM |
SLC4A9 Anion Exchanger (Ae4)
The anion exchanger 4 transports interstitial \(\hbox {Cl}^-\) into the cell while extruding 2 \(\hbox {HCO}_3^-\) ions and \(\hbox {Na}^+\)-like monovalent cations (per cycle). In our model, the only monovalent cation, other than \(\hbox {Na}^+\), is \(\hbox {K}^+\). However, it has been suggested that \(\hbox {Cs}^+\), \(\hbox {Li}^+\) and \(\hbox {Rb}^+\) are also extruded through the Ae4 (Peña-Münzenmayer et al. 2016).
We modelled the Ae4 exchanger as a Markov state model for a single exchanger (Dupont et al. 2016). We assume 2 conformational states: (1) the exchanger working forwards and the backward reaction with the \(\hbox {HCO}_3^-\) binding site exposed in the interior (\(A_i\) and \(B_i\)) and (2) with the \(\hbox {HCO}_3^-\) binding site exposed on the exterior (\(A_e\) and \(B_e\)), respectively (Fig. 10). The lower-case notation \(\hbox {cl}\), \(\hbox {hco}_3^-\), \(\hbox {na}^+\) and \(\hbox {k}^+\) denotes the concentration of each ion, respectively. As a simplification, we have assumed simultaneous binding and unbinding of ions to the exchanger. The equations that describe the exchanger under this model are
where \(\beta _j=(\hbox {hco}_3^-)^2 (\hbox {na}^+ + \hbox {k}^+)\); for \(j=i,e\). Eq. (30), is a conservation equation. The steady-state flux is
where \(K_i=k_{-i}/k_i\), and \(i=1,\ldots ,4\). The values \(\gamma _j\), where \(j=1,\ldots ,7\), are a condensed form to write the combination of the different parameters that make the equation. After some careful algebraic manipulation, it can be shown that the expression above simplifies to:
where \(G_{Ae4}\) (with units of \(\text {fmol}\)) denotes the density of exchangers. Thus, \(J_{Ae4}\) has units of concentration/time. The parameters, \(\hbox {k}_+\) and \(\hbox {k}_-\), are the association and dissociation rates, respectively. These were found by solving the bicarbonate steady-state equation (Eq. 4).
Ae4 | Description | Value | Units |
|---|---|---|---|
\(G_{\mathrm{Ae4}}\) | Density of Ae4 | 0.66 | \(\hbox {amol}/\upmu \hbox {m}^{3}\) |
\(\hbox {k}_+\) | Rate | \(1.92\times 10^{-2}\) | \(\hbox {mM}^{-4}\ \hbox {s}^{-1}\) |
\(\hbox {k}_-\) | Rate | \(1.3 \times 10^{-5}\) | \(\hbox {mM}^{-4}\ \hbox {s}^{-1}\) |
\(\hbox {CO}_2\) Transport and \(\hbox {HCO}_3^-\) Buffering
The acinar cell’s membrane is permeable to \(\hbox {CO}_2\) which diffuses down its concentration gradient into the cytoplasm where it combines with water to form carbonic acid (\(\hbox {H}_2\hbox {CO}_3\)). Carbonic anhydrases catalyse the reaction and dissociate the acid quickly into \(\hbox {H}^+\) ions and \(\hbox {HCO}_3^-\) ions. For simplicity, we assume the reaction occurs as follows:
Using the law of mass action, and assuming chemical equilibrium, we have
Diffusion across both membranes is modelled as
\(P_{\mathrm{CO}_2}\) is the membrane permeability to \(\hbox {CO}_2\) (same for both membranes). From the reaction above, we can derive (using the law of mass action) a term for the production of \(\hbox {HCO}_3^-\) and \(\hbox {H}^+\) that is proportionally dependent on the influx of \(\hbox {CO}_2\),
\(\hbox {HCO}_3^-\) Buffer | Description | Value | Units |
|---|---|---|---|
\(k_{1}\) | Rate | 11 | \(\hbox {s}^{-1}\) |
\(k_{-1}\) | Rate | \(2.6\times 10^4\) | \(\hbox {s}^{-1}\) |
\(P_{\mathrm{CO}_2}\) | Membrane \(\hbox {CO}_2\) transport rate | \(1.97\times 10^{-13}\) | \(\hbox {s}^{-1}\) |
Electroneutrality
It is a constraint of the model that the interstitium, the cellular media and the acinar lumen must maintain electroneutrality. For instance, in the cellular compartment we keep track of 6 ionic species. However, in reality there are many more. The number of moles of large negatively charged molecules (with valence \(z_x \le -1\)) that are impermeable to the cellular membrane and thus trapped inside the cell is denoted \(x_i\). To find its value, we note
where \(\omega _i\) is the volume of the cell. We solve for \(x_i\),
Similarly, in the acinar lumen and the interstitium we must have:
Tight Junction
The tight junction currents are given by a linear current–voltage (I–V) relationship:
Here \(z^{\text {Na}}=+1\) and \(z^\text {K}=+1\), correspond to the valence of each ion species, respectively. \(V_{Na}^t\) and \(V_K^t\) are their respective Nernst potentials:
The potential at the tight junction, \(V_t\), is given as:
Other Parameters of the Model
See Table 2.
Rights and permissions
About this article
Cite this article
Vera-Sigüenza, E., Catalán, M.A., Peña-Münzenmayer, G. et al. A Mathematical Model Supports a Key Role for Ae4 (Slc4a9) in Salivary Gland Secretion. Bull Math Biol 80, 255–282 (2018). https://doi.org/10.1007/s11538-017-0370-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-017-0370-6