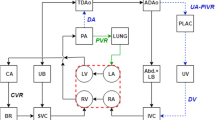
Abstract
Fetal acidemia during labor is associated with an increased risk of brain injury and lasting neurological deficits. This is in part due to the repetitive occlusions of the umbilical cord (UCO) induced by uterine contractions. Whereas fetal heart rate (FHR) monitoring is widely used clinically, it fails to detect fetal acidemia. Hence, new approaches are needed for early detection of fetal acidemia during labor. We built a mathematical model of the UCO effects on FHR, mean arterial blood pressure (MABP), oxygenation and metabolism. Mimicking fetal experiments, our in silico model reproduces salient features of experimentally observed fetal cardiovascular and metabolic behavior including FHR overshoot, gradual MABP decrease and mixed metabolic and respiratory acidemia during UCO. Combined with statistical analysis, our model provides valuable insight into the labor-like fetal distress and guidance for refining FHR monitoring algorithms to improve detection of fetal acidemia and cardiovascular decompensation.








Similar content being viewed by others
Abbreviations
- FHR:
-
Fetal heart rate
- MABP:
-
Mean arterial blood pressure
- UCO:
-
Umbilical cord occlusions
- BD:
-
Base deficit
- RMSSD:
-
Root-mean-square of the successive differences
- \(a_h\) :
-
Coefficient related to ventricular elastance during relaxation (mmHg/ml\(^2\))
- \(\alpha _b\) :
-
Maximum binding capacity of hemoglobin (ml \(\mathrm {O_2}\)/g Hb)
- b :
-
Parameter related to ventricular volume for zero diastolic pressure (ml)
- \(\beta _d\) :
-
Scaling factor on dissolved oxygen (1/mmHg)
- \(\beta _h(H)\) :
-
Time for the onset of ventricular relaxation (s)
- \(\eta \) :
-
Parameter controlling the steepness of the relation for heart pressure
- \(\theta \) :
-
Median value of time for peak pressure in heart model (1/s)
- \(\phi \) :
-
Median value of heart pressure in heart model (1/s)
- \(\psi \) :
-
Scaling factor in mean arterial pressure (1/s)
- \(\mathrm {[CO_2]}_i\) :
-
Carbon dioxide concentration in compartment \(i=\mathrm{nc,c,um}\) (ml \(\mathrm {CO_2}\)/ml blood)
- \(\mathrm {[CO_2]}_{a}\) :
-
Feeding Carbon dioxide concentration in fetus (ml \(\mathrm {CO_2}\)/ml blood)
- \(c_\mathrm{f}, c_\mathrm{m}\) :
-
Heart contractility for fetus/mother (mmHg/ml)
- \(c_{1,\mathrm m}, c_{1,\mathrm f}\) :
-
Scaling constant in the relation between Oxygen content and its partial pressure for mother and fetus (\(\hbox {mmHg}^3\))
- \(c_{2,\mathrm{m}}, c_{2,\mathrm{f}}\) :
-
Scaling constant in the relation between Oxygen content and its partial pressure for mother and fetus (\(\hbox {mmHg}^2\))
- \(C_i\) :
-
Compliance in compartment \(i=\mathrm{nc,c,um,ut}\) (ml/mmHg)
- \(d_\mathrm{f}, d_\mathrm{m}\) :
-
Parameter related to the volume-dependent and volume-independent components of the developed pressure (mmHg)
- \(\delta G_{T}\) :
-
Scaling factor for sympathetic gain (\(\hbox {s}^2\))
- D :
-
Mass transfer coefficient in Oxygen model (\(\hbox {ml }\mathrm {O_2}\hbox {/s/mmHg}\))
- \(D_{\mathrm{CO}_2}\) :
-
Mass transfer coefficient in Carbon dioxide model (\(\hbox {ml }\mathrm {CO_2} \hbox {/s/mmHg}\))
- \(\Delta C_v\) :
-
Effector in venous compliance (ml/mmHg)
- \(\Delta c\) :
-
Effector in heart contractility (mmHg/ml)
- \(f_{\mathrm{br}}, f_{\mathrm{cr}}\) :
-
Afferent firing rate via stimulation of baro/chemoreceptor (1/s)
- \(f_{i,o}\) :
-
Offset values that create a threshold for vagal or sympathetic response \(i=\mathrm{va},s\beta ,s\alpha \) (1/s)
- \(f_{i,\mathrm{min}}, f_{i,\mathrm{max}}\) :
-
Control constants in sigmoid functions \(i=s,\hbox {va},\hbox {hy},\hbox {br},\hbox {cr}\) (1/s)
- \(f_{i,n}\) :
-
A normal value for corresponding afferent or efferent firing rate \(i=\hbox {br}, \hbox {cr}, \hbox {va},s\beta ,s\alpha \) (1/s)
- \(f_\mathrm{s,0}, f_{\mathrm{s},\infty }\) :
-
Constants in sympathetic firing rate function (1/s)
- \(f_{\mathrm{s}\beta }, f_{\mathrm{s}\alpha }\) :
-
Efferent \(\alpha \)- and \(\beta \)- sympathetic firing rate (1/s)
- \(f_{\mathrm{s},1}\) :
-
Control parameter in sympathetic firing rate function (1/s)
- \(f_\mathrm{va}\) :
-
Efferent vagal firing rate (1/s)
- \(G_{i}\) :
-
Gain constants \(i=T_\mathrm{va}, T_s\) (\(\hbox { s}^2\))
- \(G_c, G_\mathrm{va}\) :
-
Constants in effectors (mmHg s/ml)
- \(\mathrm {[GL]}_a\) :
-
Arterial glucose concentration (mM)
- \(\mathrm {[GL]}_i\) :
-
Glucose concentration in compartment \(i=\hbox {nc}, \hbox {c}, \hbox {um}\) (mM)
- g(t):
-
Activation function in heart model
- H (FHR):
-
Heart rate (1/s)
- \(\mathrm {[H^+]}_a\) :
-
Arterial proton concentration (nM)
- \(\mathrm {[H^+]}_i\) :
-
Proton concentration in compartment \(i=\hbox {nc}, \hbox {c}, \hbox {um}\) (nM)
- \(Hb_\mathrm{f}, Hb_\mathrm{m}\) :
-
Hemoglobin concentration in fetus or mother (g Hb/ml blood)
- \(K_{f}\) :
-
Constant in Oxygen metabolic model (ml blood/s)
- \(K_1\) :
-
Kinetic constant (mmol/s)
- \(K_i\) :
-
Kinetic constant \(i=2,3,4,5,6\) (ml/s)
- \(K_7\) :
-
Kinetic constant (\(10^{-6}\,\hbox {ml/ s}\))
- \(K_{8}, K_9\) :
-
Kinetic constant (nmol/s)
- \(K_{10}\) :
-
Kinetic constant (\(10^{6}\,\hbox {ml/s}\))
- \(K_{\mathrm {CO_2}}\) :
-
Scaling constants (ml \(\mathrm {CO_2}\)/ml blood/mmHg)
- \(k_{\mathrm {CO_2}}\) :
-
Scaling constants ml (\(\mathrm {CO_2}/\hbox {ml blood}\))
- \(k_{c}\) :
-
Control constant
- \(k_\mathrm{hy}, k_{\mathrm{pr},s}\) :
-
Control constant (mmHg)
- \(k_{i}\) :
-
Constant \(i=R_\mathrm{nc},\hbox {br}, \hbox {cr}, \hbox {s}, \hbox {va}\) (1/s)
- \(\mathrm {[LA]}_{a}\) :
-
Arterial lactate concentration (mM)
- \(\mathrm {[LA]}_{i}\) :
-
Lactate concentration in compartment \(i=\hbox {nc}, \hbox {c}, \hbox {um}\) (mM)
- \(M_i\) :
-
Production rate of \(\mathrm {[CO_2]_i}\) with \(i=\hbox {nc}, \hbox {c}, \hbox {um}\) (ml/s)
- \(M_\mathrm{pH}\) :
-
Production rate due to \(\mathrm {[H^+]}\) accumulation (ml/s)
- \(N_p\) :
-
Normalization factor (s)
- \(\nu \) :
-
Parameter related to steepness of the time for peak pressure in heart model
- m, n :
-
Parameters related to the onset time of ventricular relaxation in heart model
- \(\mathrm {[O_{2}]}_\mathrm{th}\) :
-
Threshold value to control Oxygen metabolic uptake (\(\hbox {ml }\mathrm {O_2}/ \hbox {ml blood}\))
- \(\mathrm {[O_{2}]}_{a}, \mathrm {[O_{2}]}_{m,a}\) :
-
Feeding Oxygen concentration in fetus and mother (\(\hbox {ml }\mathrm {O_2}/\hbox {ml blood}\))
- \(\mathrm {[O_2]}_{a,n}\) :
-
A normal value to control the sigmoid function in systemic resistance (\(\hbox {ml }\mathrm {O_2}/\hbox {ml blood}\))
- \(\mathrm {[O_2]_i}\) :
-
Oxygen concentration in compartment \(i=\hbox {nc}, \hbox {c}, \hbox {um}, \hbox {ivs}\) (ml \(\mathrm {O_2}/\hbox {ml blood}\))
- \(\mathrm {O_{\mathrm{met},i}}\) :
-
Metabolic Oxygen uptake in compartment \(i=\hbox {nc, c}\) (ml \(\mathrm {O_2}/\hbox {s}\))
- \(p_\mathrm{min},p_\mathrm{max}\) :
-
Control constants in heart model (mmHg)
- \(p_i\) :
-
Blood pressure in compartment \(i=\hbox { nc, c, um, h, ut, ivs}\) (mmHg)
- \(\bar{p}_a\) (MABP):
-
Mean arterial blood pressure (mmHg)
- \(\mathrm {P}_{\mathrm {O_2},i}\) :
-
Oxygen partial pressure in compartment \(i=\hbox {nc, c, um, ivs}\) (mmHg)
- \(\mathrm {P}_{\mathrm {CO_2},i}\) :
-
Carbon dioxide partial pressure in compartment \(i=\hbox {nc, c, um}\) (mmHg)
- \(\mathrm {P}_{\mathrm {CO_2},\hbox {nc, n}}\) :
-
A normal value for Carbon dioxide partial pressure in systemic compartment (mmHg)
- \(\mathrm {P}_{\mathrm {O_{2}},c,0}\) :
-
Threshold value for cerebral Oxygen partial pressure to control the sigmoid function in firing rate function \(f_\mathrm{hy}\) (mmHg)
- \(\mathrm {[PY]_a}\) :
-
Arterial pyruvate concentration (mM)
- \(\mathrm {[PY]_i}\) :
-
Pyruvate concentration in compartment \(i=\hbox {nc, c, um}\) (mM)
- \(q_{i,a}\) :
-
Arterial flow rate into compartment \(i=\hbox {nc, c, um}\) (ml/s)
- \(q_{i,v}\) :
-
Venous flow rate out of compartment \(i=\hbox {nc, c, um}\) (ml/s)
- \(q_\mathrm{in}, q_\mathrm{out}\) :
-
Flow rate in/out of corresponding compartment by summing over flow rate from local branches (ml/s)
- \(R_i\) :
-
Resistance in compartment \(i=\mathrm{nc, c, um, ut, ivs, m}\) (mmHg s/ml)
- \(R_{c,a,0}, R_{cn,0}\) :
-
Baseline values for cerebral and systemic resistance (mmHg s/ml)
- \(R_{c, \mathrm{min}}, R_{c, \mathrm{max}}\) :
-
Constants in cerebral autoregulation model (mmHg s/ml)
- \(R_{\mathrm{nc, min}}, R_{\mathrm{nc, max}}\) :
-
Constants in systemic resistance model (mmHg s/ml)
- \(\Delta R_\mathrm{nc}\) :
-
Effector in systemic resistance (mmHg s/ml)
- \(S_{ij}\) :
-
Relative sensitivity matrix
- \(S_{\mathrm {[O_2]},d}\) :
-
Oxygen diffusion (ml \(\mathrm {O_2}/\hbox {s}\))
- \(S_{\mathrm {P[O_2]}}\) :
-
Scaling function to relate Oxygen content and its partial pressure
- \(s_\mathrm{mv}, s_\mathrm{av}\) :
-
Indicator function of mitral/aortic valve
- \(t_\mathrm{p}\) :
-
Peak time (s)
- \(t_\mathrm{min},t_\mathrm{max}\) :
-
Control constants in heart model (s)
- \(T_0\) :
-
Base line value for heart period (s)
- \(T_{v,0}, T_{\mathrm{s},0}\) :
-
Contributions from vagal and sympathetic activities on the base heart period (s)
- \(\tau _i\) :
-
Control constant \(i=\hbox {gs},\hbox {gv}\) (1/s)
- \(\tau _0\) :
-
Offset constant in heart period (s)
- \(\tau _\mathrm{pr,br},\tau _\mathrm{pr,cr}\) :
-
Control constant (mmHg)
- \(\tau _\mathrm{br}, \tau _\mathrm{cr}, \tau _s, \tau _{Rc}\) :
-
Time constant (s)
- \(\tau _{T,s}, \tau _{T,v}\) :
-
Time constant (s)
- \(\Delta T_s,\Delta T_\mathrm{va}\) :
-
Contribution to heart period from sympathetic/vagal activity (s)
- \(V_i\) :
-
Volume in compartment \(i=\hbox {nc, c, um, h, ut}\) (ml)
- \(V_\mathrm{total}\) :
-
Total volume of all compartments in the circulation system (ml)
- \(W_{\mathrm{br},i}, W_{\mathrm{cr},i}\) :
-
Weight factor \(i=s\alpha , s\beta \)
References
Agrawal S, Doucette F, Gratton R, Richardson B, Gagnon R (2003) Intrapartum computerized fetal heart rate parameters and metabolic acidosis at birth. Obstet Gynecol 102:731
Bennet L, Westgate J, Liu Y, Wassink G, Gunn A (2005) Fetal acidosis and hypotension during repeated umbilical cord occlusions are associated with enhanced chemoreflex responses in near-term fetal sheep. J Appl Physiol 99:1477
Bishai J, Blood A, Hunter C, Longo L, Power G (2003) Fetal lamb cerebral blood flow (CBF) and oxygen tensions during hypoxia: a comparison of laser Doppler and microsphere measurements of CBF. J Physiol 456:869
Breit M (2004) Sensitivity analysis of biological pathways, Master’s thesis, University for Health Sciences, Medical Informatics and Technology, Germany
Cabrera M, Saidel G, Kalhan S (1998) Role of O\(_2\) in regulation of lactate dynamics during hypoxia: mathematical model and analysis. Ann Biomed Eng 26:1
Cloutier M, Bolger F, Lowry J, Wellstead P (2009) An integrative dynamic model of brain energy metabolism using in vivo neurochemical measurements. J Comput Neurosci 3:391
Couto P, Meurs W, Bernardes J, de Sa JM, Goodwin J (2002) Mathematical model for educational simulation of the oxygen delivery to the fetus. Control Eng Pract 10:59
D’Angelo C, Papelier Y (2005) Mathematical modeling of the cardiovascular system and skeletal muscle interaction during excecise. ESAIM Proc 14:72
de Haan H, Gunn A, Gluckman P (1997) Fetal heart rate changes do not reflect cardiovascular deterioration during brief repeated umbilical cord occlusions in near-term fetal lambs. Am J Obstet Gynecol 176:8
Durosier L, Green G, Batkin I, Seely A, Ross M, Richardson B, Frasch M (2014) Sampling rate of heart rate variability impacts the ability to detect acidemia in ovine fetuses near-term. Front Pediatr 2:PMC4017161
Ellwein L, Tran H, Zapata C, Novak V, Olufsen M (2008) Sensitivity analysis and model assessment: mathematical models for arterial blood flow and blood pressure. Cardiovasc Eng 8:94
Frasch M, Muller T, Hoyer D, Weiss C, Schubert H, Schwab M (2009a) Nonlinear properties of vagal and sympathetic modulations of heart rate variability in ovine fetus near term. Am J Physiol Regul Integr Comp Physiol 296:R702
Frasch M, Mansano R, Gagnon R, Richardson B, Ross M (2009b) Measures of acidosis with repetitive umbilical cord occlusions leading to fetal asphyxia in the near-term ovine fetus. AJOG 200(200):e1
Frasch M, Muller T, Hoyer D, Weiss C, Schubert H, Schwab M (2009c) Heart rate variability analysis allows early asphyxia detection in ovine fetus. Reprod Sci 16:509
Frasch M, Keen A, Gagnon R, Ross M, Richardson B (2011) Monitoring fetal electrocortical activity during labour for predicting worsening acidemia: a prospective study in the ovine fetus near term. PLoS One 6:e22100
Frasch M, Frank B, Last M, Muller T (2012) Time scales of autonomic information flow in near-term fetal sheep. Front Physiol 3:378
Gold N, Frasch M, Herry C, Richardson B, Seely A, Wang X (2015) Online change-point detection for non stationary biological time series with Gaussian processes. In preparation
Gui R, Schüthe C, Bernhard S (2015) Mathematical modeling and sensitivity analysis of arterial anastomosis in arm arteries. ZIB Report
Hunter C, Blood A, Power G (2003) Cerebral metabolism during cord occlusion and hypoxia in the fetal sheep: a novel method of continuous measurement based on heat production. J Physiol 552:241
Jensen A, Garnier Y, Berger R (1999) Dynamics of fetal circulatory responses to hypoxia and asphyxia. Eur J Obstet Gynecol Reprod Biol 84:155
Khoo M, Kronauer R, Strohl K, Slutsky A (1982) Factors inducing periodic breathing in humans: a general model. J Appl Physiol 53:644
Khoo M, Gottschalk A, Pack A (1991) Sleep induced periodic breathing and apnea: a theoretical study. J Appl Physiol 70:2014
Koos B, Chau A, Qgunyemi D (1995) Adenosine mediates metabolic and cardiovascular responses to hypoxia in fetal sheep. J Physiol 488:761
Kubota T, Alexander J, Itaya R, Todaka K, Sugimachi M, Sunagawa K, Nose Y (1992a) Dynamic effects of carotid sinus baroreflex on ventriculoarterial coupling studied in anesthetized dogs. Circ Res 70:1044
Kubota T, Chishaki H, Yoshida T, Sunagawa K, Takeshita A, Nose Y (1992b) How to encode arterial pressure into carotid sinus nerve to invoke natural baroreflex. Am J Physiol 263:H307
Levy M, Zieske H (1969) Autonomic control of cardiac pacemaker activity and atrioventricular transmission. J Appl Physiol 27:465
Liang F, Liu H (2006) Simulation of hemodynamic responses to the Valsalva Maneuver: an integrative computational model of the cardiovascular system and the autonomic nervious system. J Physiol Sci 56:45
Liang F, Takagi S, Himeno R (2009) Multi-scale modeling of the human cardiovascular system with applications to aortic valvular and arterial stenoses. Med Biol Eng Comput 47:743
Liston R, Sawchuck D, Young D (2007) Fetal health surveillance: antepartum and intrapartum consensus guideline. J Obstet Gynecol Can 29:53
Low J, Panagiotopoulos C, Derrick E (1995) Newborn complications after intrapartum asphyxia with metabolic acidosis in the preterm fetus. Am J Obstet Gynecol 172:805
Magosso E, Ursino M (2001) A mathematical model of CO\(_2\) effect on cardiovascular regulation. Am J Physiol Heart Circ Physiol 281:H2036
Mallard E, Williams C, Johnston B, Gunning M, Davis S et al (1995) Repeated episodes of umbilical cord occlusion in fetal sheep lead to preferential damage to the striatum and sensitize the heart to further insults. Pediatr Res 37:707
Olufsen M, Tran H, Ottesen J (2004) Modeling cerebral blood flow control during posture change from sitting to standing. Cardiovasc Eng Int J 4:47
Olufsen M, Ottesen J, Tran H, Ellwein L, Lipsitz L, Novak V (2005) Blood pressure and blood flow variation during postural change from sitting to standing: model development and validation. J Appl Physiol 99:1523
Orlowski P, Chappell M, Park C, Grau V, Payne S (2011) Modeling of pH dynamics in brain cells after stroke. Interface Focus 1:408
Quilligan E, Vasicka A, Aznar R, Lipsitz P, Moore T, Bloor BM (1960) Partial pressure of oxygen in the intervillous space and the umbilical vessels. Am J Obstet Gynecol 79:1048
Ramsay J, Hooker G, Campbell D, Cao J (2007) Parameter estimation for differential equations: a generalized smoothing approach. J R Stat Soc B 69:741
Ross M, Jessie M, Amaya K, Matushewski B, Durosier L, Martin M, Richardson B (2013) Correlation of arterial fetal base deficit and lactate changes with severity of variable heart rate decelerations in the near-term ovine fetus. Am J Obstet Gynecol 208:e1
Shi Y, Patricia L, Rodney H (2011) Review of zero-D and 1-D models of blood flow in the cardiovascular system. Biomed Eng Online 10:1
Thakor A, Giussani D (2008) Effects of acute acidemia on the fetal cardiovascular defense to acute hypoxemia. Am J Physiol Regul Integr Comp Physiol 296:R90
Ursino M (1998) Interaction between carotid baroregulation and the pulsating heart: a mathematical model. Am J Physiol 275:H1733
Ursino M, Magosso E (2000) Acute cardiovascular response to isocapnic hypoxia. I. A mathematical model. Am J Physiol Heart Circ Physiol 279:H149
Ursino M, Magosso E (2003) Role of short-term cardiovascular regulation in heart period variability. a modeling study. Am J Physiol Heart Circ Physiol 284:H1479
van der Hout-van der Jagt M, Oei S, Bovendeerd P (2012) A mathematical model for simulation of early decelerations in cardiotocogram during labor. Med Eng Phys 34:579
van der Hout-van der Jagt M, Oei S, Bovendeerd P (2013) Simulation of reflex late decelerations in labor with a mathematical model. Early Hum Dev 89:7
Wang X, Durosier L, Ross M, Richardson B, Frasch M (2014) Online detection of fetal acidemia during labour by testing synchronization of EEG and heart rate: a prospective study in fetal sheep. PLoS One 9:PMC4182309
Westgate J, Bennet L, Gunn A (1999) Fetal heart rate variability changes during brief repeated umbilical cord occlusion in near term fetal sheep. J Obstet Gynecol 106:664
Westgate J, Bennet L, de Haan H, Gunn A (2001) Fetal heart rate overshoot during repeated umbilical cord occlusion in sheep. Obstet Gynecol 97:454
Westgate J, Wibbens B, Bennet L, Wassink G, Parer J, Gunn A (2007) The intrapartum deceleration in center stage: a physiologic approach to the interpretation of fetal heart rate changes in labor. Am J Obstet Gynecol 197(236):e1
Yan E, Baburamant A, Walker A, Walker D (2009) Changes in cerebral blood flow, cerebral metabolites, and breathing movements in the sheep fetus following asphyxia produced by occlusion of the umbilical cord. Am J Physiol Regul Integr Comp Physiol 297:R60
Acknowledgments
The authors are grateful to Dr. Josh Chang for some useful discussions. This work is partially supported by MITACS, NeuroDevNet, NSERC, CIHR and the Fields Institute.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, Q., Gold, N., Frasch, M.G. et al. Mathematical Model of Cardiovascular and Metabolic Responses to Umbilical Cord Occlusions in Fetal Sheep. Bull Math Biol 77, 2264–2293 (2015). https://doi.org/10.1007/s11538-015-0122-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-015-0122-4