Abstract
Evidence is presented to show that self-sustained oscillations of purely hemodynamical origin are possible in some arcade-type microvascular networks supplied with steady boundary conditions, but that in others the oscillations disappear with sufficient reduction of the time step Δt, showing them to be numerical artefacts. In an attempt to elucidate the mechanisms involved in the onset of fluctuations, we proceed to perform a linear stability analysis for the convective model of Kiani et al. (Microvasc. Res. 45:219–232, 1993; Am. J. Physiol. 266(35):H1822–H1828, 1994), and show that this leads via a system of delay differential equations to a nonlinear eigenvalue problem. This result generalises the characteristic equation obtained by Carr et al. (Ann. Biomed. Eng. 33:764–771, 2005) and Geddes et al. (SIAM J. Appl. Dyn. Syst. 6(4):694–727, 2007) who solved a special case in a two node network. An implicit numerical method is proposed for the computation of blood flows in networks using the convective model.
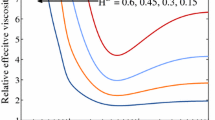
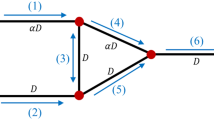
In a moderate size subnetwork of one of the networks chosen by Kiani et al. (Am. J. Physiol. 266(35):H1822–H1828, 1994), the topology, vessel lengths, and diameters of which were based on microvascular networks in the rat mesentery, we compare results generated using the original explicit numerical method of Kiani et al. (Am. J. Physiol. 266(35):H1822–H1828, 1994) with those from our implicit scheme. From the linear stability theory, a critical value D RBC,crit of a red blood cell diameter parameter D RBC in the plasma skimming model of Fenton et al. (Pflügers Arch. 403:396–401, 1985b) is identified for the onset of oscillations about steady state and both the explicit and implicit methods are used to calculate the inflow hematocrit solutions in all vessels of the subnetwork at the critical parameter value, subject to perturbed initial conditions. The results of the implicit method are demonstrated to be in excellent and superior agreement with the predictions of the linear analysis in this case. For values of D RBC slightly larger than D RBC,crit the bifurcating periodic solutions calculated using either the explicit or implicit schemes are characteristic of those of a supercritical Hopf bifurcation and the graphs of D RBC vs. oscillation amplitude would seem to converge as Δt→0.













Similar content being viewed by others
References
Broyden, C. G. (1965). A class of methods for solving nonlinear simultaneous equations. Math. Comput., 19(92), 577–593.
Buzsáki, G., & Draguhn, A. (2004). Neuronal oscillations in cortical networks. Science, 304, 1926–1929.
Carr, R. T., & Lacoin, M. (2000). Nonlinear dynamics of microvascular flow blood flow. Ann. Biomed. Eng., 28, 641–652.
Carr, R. T., Geddes, J. B., & Wu, F. (2005). Oscillations in a simple microvascular network. Ann. Biomed. Eng., 33, 764–771.
Cokelet, G. R. (1997). A commentary on the “in vivo viscosity law”. Biorheology, 34(4–5), 363–367. Discussion 369–373.
Davis, J. M., & Pozrikidis, C. (2011). Numerical simulation of unsteady blood flow through capillary networks. Bull. Math. Biol., 73, 1857–1880.
Dawant, B., Levin, M., & Popel, A. S. (1986). Effect of dispersion of vessel diameters and lengths in stochastic networks. I. Modeling of microcirculatory flow. Microvasc. Res., 31, 203–222.
Dellimore, J. W., Dunlop, M. J., & Canham, P. B. (1983). Ratio of cells and plasma in blood flowing past branches in small plastic channels. Am. J. Physiol., 244, H635–H643.
Eisenstat, S. C., Schultz, M. H., & Sherman, A. H. (1982). Algorithms and data structures for sparse symmetric Gaussian elimination. SIAM J. Sci. Stat. Comput., 2(2), 225–237.
Ellis, C. G., Wrigley, S. M., & Groom, A. C. (1994). Heterogeneity of red blood cell perfusion in capillary networks supplied by a single arteriole in resting skeletal muscle. Circ. Res., 75, 357–368.
Fabry, M. E., Kaul, D. K., Carmen, R., Baez, S., Rieder, R., & Nagel, R. L. (1981). Some aspects of the pathophysiology of homozygous Hb CC erythrocytes. J. Clin. Invest., 67(5), 1284–1291.
Fagrell, B., Intaglietta, M., & Östergren, J. (1980). Relative hematocrit in human skin capillaries and its relationship to capillary flow velocity. Microvasc. Res., 20, 327–335.
Fåhraeus, R. (1929). The suspension stability of the blood. Physiol. Rev., 9, 241–274.
Fåhraeus, R., & Lindqvist, T. (1931). The viscosity of blood in narrow capillary tubes. Am. J. Physiol., 96, 562–568.
Fenton, B. M., Carr, R. T., & Cokelet, G. R. (1985a). Nonuniform red cell distribution in 20 to 100 μm bifurcations. Microvasc. Res., 29, 103–126.
Fenton, B. M., Wilson, D. W., & Cokelet, G. R. (1985b). Analysis of the effects of measured white blood cell entrance times on hemodynamics in a computer model of a microvascular bed. Pflügers Arch., 403, 396–401.
Forouzan, O., Yang, X., Sosa, J. M., Burns, J. M., & Shevkoplyas, S. S. (2012). Spontaneous oscillations of capillary blood flow in artificial microvascular networks. Microvasc. Res., 84, 123–132.
Fung, Y.-C. (1973). Stochastic flow in capillary blood vessels. Microvasc. Res., 5(1), 34–48.
Furman, M. B., & Olbricht, W. L. (1985). Unsteady cell distributions in capillary networks. Biotechnol. Prog., 1(1), 26–32.
Geddes, J. B., Carr, R. T., Karst, N. J., & Wu, F. (2007). The onset of oscillations in microvascular blood flow. SIAM J. Appl. Dyn. Syst., 6(4), 694–727.
Geddes, J. B., Carr, R. T., Wu, F., Lao, Y., & Maher, M. (2010). Blood flow in microvascular networks: a study in nonlinear biology. Chaos, 20, 045123.
Glass, L. (2001). Synchronization and rhythmic processes in physiology. Nature, 410, 277–284.
Griffith, T. M. (1996). Temporal chaos in the microcirculation. Cardiovasc. Res., 31, 342–358.
Hestenes, M. R., & Stiefel, E. (1952). Methods of conjugate gradients for solving linear systems. J. Res. Natl. Bur. Stand., 49(6), 410–436.
Hopf, E. (1942). Abzweigung einer periodischen Lösung von einer stationären Lösung eines Differentialsystems. In Berichten der Mathematisch-Physischen Klasse der Sächsischen Akademie der Wissenschaften zu Leipzig (Vol. XCIV, pp. 1–22).
Jarlebring, E., & Voss, H. (2005). Rational Krylov for nonlinear eigenproblems, an iterative projection method. Appl. Math.-Czech., 50(6), 543–554.
Jarlebring, E., Michiels, W., & Meerbergen, K. (2012). A linear eigenvalue algorithm for the nonlinear eigenvalue problem. Numer. Math., 122(1), 169–195. doi:10.1007/s00211-012-0453-0.
Kiani, M. F., Cokelet, G. R., & Sarelius, I. H. (1993). Effect of diameter variability along a microvessel segment on pressure drop. Microvasc. Res., 45, 219–232.
Kiani, M. F., Pries, A. R., Hsu, L. L., Sarelius, I. H., & Cokelet, G. R. (1994). Fluctuations in microvascular blood flow parameters caused by hemodynamic mechanisms. Am. J. Physiol., Heart Circ. Physiol., 266(35), H1822–H1828.
Klitzman, B., & Johnson, P. C. (1982). Capillary network geometry and red cell distribution in hamster cremaster muscle. Am. J. Physiol., Heart Circ. Physiol., 242(11), H211–H219.
Kublanovskaja, V. N. (1969). The application of Newton’s method to the determination of the eigenvalues of λ-matrices. Dokl. Akad. Nauk SSSR, 188, 1004–1005. Soviet Math. Dokl. 10, 1240–1241.
Kublanovskaya, V. N. (1970). On an approach to the solution of the generalized latent value problem for λ-matrices. SIAM J. Numer. Anal., 7(4), 532–537.
Levin, M., Dawant, B., & Popel, A. S. (1986). Effect of dispersion of vessel diameters and lengths in stochastic networks. II. Modeling of microvascular hematocrit distribution. Microvasc. Res., 31, 223–234.
Lew, H. S. (1972). Role of flow bifurcation in creating an oscillatory flow in capillary blood vessels. J. Biomech., 5(2), 231–238.
Liao, X.-H., Qian, Y., Mi, Y.-Y., Xia, Q.-Z., Huang, X.-Q., & Hu, G. (2011). Oscillation sources and wave propagation paths in complex networks consisting of excitable nodes. Front. Phys., 6(1), 124–132.
Lorthois, S., & Cassot, F. (2010). Fractal analysis of vascular networks: insights from morphogenesis. J. Theor. Biol., 262, 614–633.
Moler, C. B., & Stewart, G. W. (1973). An algorithm for generalized matrix eigenvalue problems. SIAM J. Numer. Anal., 10, 241–256.
Obrist, D., Weber, B., Buck, A., & Jenny, P. (2010). Red blood cell distribution in simplified capillary networks. Philos. Trans. R. Soc. A, 368, 2897–2918.
Papenfuss, H.-D., & Gross, J. F. (1981). Microhemodynamics of capillary networks. Biorheology, 18, 673–692.
Pop, S. R., Richardson, G., Waters, S. L., & Jensen, O. E. (2007). Shock formation and non-linear dispersion in a microvascular capillary network. Math. Med. Biol., 24(4), 379–400.
Popel, A. S., & Johnson, P. C. (2005). Microcirculation and hemorheology. Annu. Rev. Fluid Mech., 37, 43–69.
Pozrikidis, C. (2009). Numerical simulation of blood flow through microvascular capillary networks. Bull. Math. Biol., 71, 1520–1541.
Pries, A. R., Secomb, T. W., Gaehtgens, P., & Gross, J. F. (1990). Blood flow in microvascular networks. Experiments and simulation. Circ. Res., 67(4), 826–834.
Pries, A. R., Neuhaus, D., & Gaehtgens, P. (1992). Blood viscosity in tube flow: dependence on diameter and hematocrit. Am. J. Physiol., Heart Circ. Physiol., 96(32), H1770–H1778.
Pries, A. R., Secomb, T. W., Geßner, T., Sperandio, M. B., Gross, J. F., & Gaehtgens, P. (1994). Resistance to blood flow in microvessels in vivo. Circ. Res., 75, 904–915.
Pries, A. R., Secomb, T. W., & Gaehtgens, P. (1996). Biophysical aspects of blood flow in the microvasculature. Cardiovasc. Res., 32, 654–667.
Ruhe, A. (1973). Algorithms for the nonlinear eigenvalue problem. SIAM J. Numer. Anal., 10(4), 674–689.
Ruhe, A. (1996). Computing nonlinear eigenvalues with spectral transformation Arnoldi. ZAMM—Z. Angew. Math. Mech., 76 S2, 17–20.
Saad, Y., & Schultz, M. H. (1986). GMRES: a generalized minimal residual algorithm for solving nonsymmetric linear systems. SIAM J. Sci. Stat. Comput., 7, 856–869.
Schmid-Schönbein, G. W., Skalak, R., Usami, S., & Chien, S. (1980). Cell distribution in capillary networks. Microvasc. Res., 19, 18–44.
Selverston, A. I., & Moulins, M. (1985). Oscillatory neural networks. Annu. Rev. Physiol., 47, 29–48.
Sleijpen, G. L. G., Booten, G. L., Fokkema, D. R., & van der Vorst, H. A. (1996). Jacobi Davidson type methods for generalized eigenproblems and polynomial eigenproblems. BIT, 36, 595–633.
Vogels, T. P., Rajan, K., & Abbott, L. F. (2005). Neural network dynamics. Annu. Rev. Neurosci., 28, 357–376.
Voronoi, G. (1908). Nouvelles applications des paramètres continus à la théorie des formes quadratiques. J. Reine Angew. Math., 133, 97–178.
Voss, H. (2003). A Jacobi–Davidson method for nonlinear eigenproblems (Report 66). Arbeitsbereich Mathematik, TU Hamburg-Harburg. Proc. ICCS04, Krakow, Poland, pp. 34–41.
Voss, H. (2004). An Arnoldi method for nonlinear eigenvalue problems. BIT, 44(2), 387–401.
Acknowledgements
Sylvie Lorthois is thanked for having suggested a study of this problem to the second author a couple of years ago and for proposing Voronoi diagrams as a way of generating large complex arcade-type network topologies. The authors wish to thank Russell T. Carr, Mohammad Kiani, and Axel Pries for supplying them with helpful technical information about the computations performed in some of their articles. This work was partially supported by a Discovery Grant from the Natural Sciences and Engineering Research Council of Canada.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tawfik, Y., Owens, R.G. A Mathematical and Numerical Investigation of the Hemodynamical Origins of Oscillations in Microvascular Networks. Bull Math Biol 75, 676–707 (2013). https://doi.org/10.1007/s11538-013-9825-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-013-9825-6