Abstract
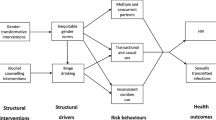
Alcohol consumption and abuse is widespread in sub-Saharan Africa where most HIV infections occur and has been associated with risky sexual behaviors. It may therefore be one of the most common, potentially modifiable HIV risk factors in this region. A deterministic system of ordinary differential equations incorporating heterogeneity and biased sexual preferences is formulated to assess the effects of alcohol consumption on the transmission dynamics of the disease in heterosexual settings. Extensive qualitative analysis of the model is carried out and epidemic threshold such as the alcohol-induced reproductive number \(({\mathcal{R}}_{A})\), and equilibria are derived and their stabilities examined. The disease-free equilibrium is found to be globally attracting whenever the reproductive number is less than unity. In the model, heterosexuality is the source of transmissions, and therefore, targeting a reduction of the basic reproductive number \(({\mathcal{R}}_{0})\) should be primary objective for any intervention programme. We show that the preference to form partnerships amongst the heterogeneous groups influences the severity of disease and its evolution, and consequently the rate of partnership formation between females and alcohol consumers and their relative infectiousness over nondrinkers has a huge positive correlation with the alcohol-induced reproductive number and hence the epidemic. The proportion or absolute number of drinkers is shown to have minimal influence on the disease dynamics, and in a community with alcohol consumers, it is more prudent to reduce their risk sexual behavior rather than to fight the spread of alcohol consumption. Thus, intervention measures targeted at reducing heterogeneous group interactions and behavior change are the key to disease control in these settings.







Similar content being viewed by others
References
Anderson, R. M., & May, R. M. (1991). Infectious diseases of humans. London: Oxford University Press.
Arriola, L., & Hyman, J. (2005). Lecture notes, forward and adjoint sensitivity analysis: with applications in dynamical systems, linear algebra and optimisation, Mathematical and Theoretical Biology Institute, Summer.
Baggaley, R. F., White, R. G., & Boily, M. C. (2008). Systematic review of orogenital HIV-1 transmission probabilities. Int. J. Epidemiol., 37(6), 1255–1265.
Bailey, R. C., Neema, S., & Othieno, R. (1999). Sexual behaviors and other HIV risk factors in circumcised and uncircumcised men in Uganda. J. Acquir. Immune Defic. Syndr., 22, 294–301.
Blythe, P. S., & Castillo-Chavez, C. (1989). Like with like preference and sexual mixing models. Math. Biosci., 96, 221–238.
Boily, M. C., Baggaley, R. F., Wang, L., Masse, B., White, R. G., Hayes, R. J., & Alary, M. (2009). Heterosexual risk of HIV-1 infection per sexual act: systematic review and meta-analysis of observational studies. Lancet Infect. Dis., 9, 118–129.
Breierova, L., vChoudhari, M., & Forrester, J. W. (2001). An introduction to sensitivity analysis, Massachusetts Institute of Technology.
Busenserg, S., & Chavez, C. C. (1991). A general solution of the problem of mixing of subpopulations and its application to risk, and age structured epidemic models of the spread of AIDS. IMA J. Math. Appl. Med. Biol., 8, 1–29.
Carr, J. (1981). Applications centre manifold theory. New York: Springer.
Castillo-Chavez, C., & Song, B. (2004). Dynamical models of tuberculosis and their applications. Math. Biosci. Eng., 1(2), 361–404.
Castillo-Chavez, C., & Yakubu, A. (2003). Mathematical models of isolation and quarantine. Am. Med. Assoc., 290(21), 2876–2877.
Castillo-Chavez, C., Feng, Z., & Huang, W. (2002). On the computation of \({\mathcal{R}}_{0}\) and its role on global stability. math.la.asu.edu/chavez/2002/JB276.pdf, accessed 20–09–2010.
Chronicle (2012). Men part of solution in reducing HIV infections. http://www.chronicle.co.zw (accessed 07-02-2012).
Clift, S., Anemona, A., Watson-Jones, D., Kanga, L., Ndeki, L., & Changalucha, J. (2003). Variations of HIV and STI prevalences assault among south African adolescents. Child Abuse Negl., 28, 683–696.
Cook, R. L., & Clark, D. (2005). Is there an association between alcohol consumption and sexually transmitted diseases? A systematic review of sexually transmitted diseases. Sex. Transm. Dis., 32, 156–164.
Del Valle, S. Y., Hyman, J. M., Hethcote, C., & Eubank, S. G. (2007). Mixing patterns between age groups in social networks. Soc. Netw., 29, 539–554.
Diekmann, O., Heesterbeek, J. A. P., & Metz, J. A. P. (1990). On the definition and computation of the basic reproductive ratio \({\mathcal{R}}_{0}\) in models for infectious diseases in heterogeneous populations. J. Math. Biol., 28, 365–382.
Dunkle, K. L., Jewkes, R. K., Brown, H. C., Gray, G. E., McIntryre, J. A., & Harlow, S. D. (2004). Transactional sex among women in Soweto, South Africa: prevalence, risk factors and association with HIV infection. Soc. Sci. Med., 59, 1581–1592.
Fan, M., Chowa, L., & Feng, Z. (2011). Dynamics of a multi-group epidemiological model with group-targeted vaccination strategies. J. Theor. Biol., 291, 56–64.
Fritz, K. E., Woelk, G. B., Bassett, M. T., McFarland, W. C., Routh, J. A., & Tobaiwa, O. (2002). The association between alcohol use, sexual risk behavior, and HIV infection among men attending beer halls in Harare, Zimbabwe. AIDS Behav., 6, 221–228.
Gorman, D. M., Mezic, J., Mezic, I., & Gruenewald, P. (2006). Agent-based modelling of drinking behavior: a preliminary model and potential applications to theory and practice. Am. J. Publ. Health, 96(11), 2055–2060.
Heck, A. (2009). Modelling intake and clearance of alcohol in humans; University of Amsterdam, AMSTEL Institute, http://staff.science.uva.nl/~heck/Research/art/ICTMT8_2.pdf (accessed 21 December 2009).
Hethcote, H. W. (2000). The mathematics of infectious diseases. SIAM Rev., 42(4), 599–653.
Hethcote, H. W., & Van Ark, W. J. (1987). Epidemiological models for heterogeneous populations: proportional mixing, parameter estimation and immunisation programs. Math. Biosci., 84, 85–118.
Hyman, J. M., & Stanley, J. L. (1997). Disease transmission with biased partnership selection. Appl. Numer. Math., 24, 379–392.
Jacquez, A. J., Simon, C. P., Koopman, J., Arbor, A., Sattenspiel, L., & Perry, T. (1988). Modeling and analyzing HIV transmission: the effect of contact patterns. Math. Biosci., 92, 119–199.
Jewkes, R., Levin, J., & Penn-Kekana, L. (2002). Risk factors for domestic violence: findings from a South African cross-sectional study. Soc. Sci. Med., 55, 1603–1617.
Jones, D. L., Ross, D., Weiss, S. M., Bhat, G., & Chitalu, N. (2005). Influence of partner participation on sexual risk behavior reduction among HIV-positive Zambian women. J. Urban Health, 82, 92–100.
Kapiga, S. H., Sam, N. E., Shao, J. F., Masenga, E. J., Renjifo, B., & Kiwelu, I. E. (2003). Herpes simplex virus type 2 infections among female bar and hotel workers in northern Tanzania: prevalence and risk factors. Sex. Transm. Dis., 30, 187–192.
King, G., Flisher, A. J., Noubary, F., Reece, R., Marais, A., & Lombard, C. (2004). Substance abuse and behavioral correlates of sexual assault among South African adolescents. Child Abuse Negl., 28, 683–696.
Koenig, M. A., Lutalo, T., Zhao, F., Nalugoda, F., Wabwire-Mangen, F., & Kiwanuka, N. (2003). Domestic violence in rural Uganda: evidence from a community-based study. Bull. World Health Organ., 81, 53–60.
Koopman, J. S., Simon, C. P., Jacquez, J. A., & Park, T. S. (1989). Selective contact within structured mixing with an application to HIV transmission risk from oral and anal sex. In C. Castillo-Chavez (Ed.), Lecture notes in biomathematics: Vol. 83. Mathematical and statistical approaches to AIDS epidemiology (pp. 316–348). New York: Springer.
Lajmanovich, A., & Yorke, J. A. (1980). A deterministic model of gonorrea in a non-homogeneous population. Math. Biosci., 52, 227–240.
Lakshmikantham, V., Leela, S., & Martynyuk, A. A. (1989). Stability analysis of nonlinear systems (pp. 1–315). New York: Dekker.
Lennox, R., Steele, P., Zarkin, G. A., & Bray, J. W. (1998). The differential effects of alcohol consumption and dependence on adverse alcohol-related consequences: implications for the workforce. Drug Alcohol Depend., 50, 211–220.
Lewis, J. J. C., Garnett, G. P., Mhlanga, S., Nyamukapa, C. A., Donnelly, C. A., & Gregson, S. (2005). Beer halls as a focus for HIV prevention activities in rural Zimbabwe. Sex. Transm. Dis., 32, 364–369.
Mataure, P., McFarland, W., Fritz, K., Woelk, G., Ray, S., & Rutherford, G. (2002). Alcohol use and high-risk sexual behavior among adolescents and young adults in Harare, Zimbabwe. AIDS Behav., 6(3), 211–219.
Mbulaiteyea, S. M., Ruberantwaria, A., Nakiyingia, J. S., Carpenter, L. M., Kamalia, A., & Whitwortha, J. (2000). Alcohol and HIV: a study among sexually active adults in rural southwest Uganda. Int. J. Epidemiol., 32, 228–229.
Morojele, N. K., Kachieng’a, M. A., Mokoko, E., Nkoko, M. A., Parry, C. D. H., & Nkowane, A. M. (2006). Alcohol use and sexual behavior among risky drinkers and bar and shebeen patrons in Gauteng province, South Africa. Soc. Sci. Med., 62, 217–227.
Mubayi, A., Greenwood, P. E., Castillo-Chavez, C., Gruenewald, P. J., & Gorman, D. M. (2010). The impact of relative residence times on the distribution of heavy drinkers in highly distinct environments. Socio-Econ. Plan. Sci., 44, 45–46.
Mukandavire, Z. (2006). A theoretical assessment of HIV/AIDS intervention strategies with some application to Zimbabwe. Ph.D. thesis, National University of Science and Technology, Bulawayo, Zimbabwe.
Mukandavire, Z., & Garira, W. (2006). HIV/AIDS model for assessing the effects of prophylactic sterilizing vaccines, condoms and treatment with amelioration. J. Biol. Syst., 14(3), 323–355.
Mukandavire, Z., & Garira, W. (2007). Sex-structured HIV/AIDS model to analyse the effects of condom use with application to Zimbabwe. J. Math. Biol., 54(5), 669–699.
Mukandavire, Z., Chiyaka, C., Magombedze, G., Musuka, G., & Malunguza, N. (2009a). Assessing the effects of homosexuals and bisexuals on the intrinsic dynamics of HIV/AIDS in African heterosexual settings. Math. Comput. Model., 49(9–10), 1869–1882.
Mukandavire, Z., Malunguza, N., Tchuenche, J. M., & Chiyaka, C. (2009b). HIV/AIDS model assessing the effects of gender-inequality affecting women in African heterosexual settings. Int. J. Biomath., 3(1), 43–67.
Myer, L., Matthews, C., & Little, F. (2002). Condom use and sexual behaviors among individuals procuring free male condoms in south Africa: a prospective study. Sex. Transm. Dis., 29, 239–241.
Nold, A. (1980). Heterogeneity in disease-transmission modelling. Math. Biosci., 5, 227–240.
Phiri, G. (2002). Alcohol abuse soars. The Weekend Tribune, 16–17 November. http://www.wtribune.co.zw/no16-no17/columns/003.htm (accessed 21 July 2011).
Phorano, O. D., Nthomang, K., & Ntseane, D. (2005). Alcohol abuse, gender-based violence and HIV/AIDS in Botswana: establishing the link based on empirical evidence. Sahara: J. Soc. Aspects HIV/AIDS, 2, 188–202.
Podder, C. N., Sharomi, O., Gumel, A. B., & Strawbridge, E. (2011). Mathematical analysis of a model for assessing the impact of anti retroviral therapy voluntary testing and condom use in curtailing the spread of HIV. Differ. Equ. Dyn. Syst., 19(4), 283–302.
Seth, C., Kalichman, L. C., Simbayi, M. K., Demetria, C., & Sean, J. (2006). Alcohol use and sexual risks for HIV/AIDS in sub-Saharan Africa: systematic review of empirical findings. Soc. Prev. Res., 8(2), 141–151.
Simbayi, L. C., Mwaba, K., & Kalichman, S. (2006). Perceptions of STI clinic attenders about HIV/AIDS and alcohol as a risk factor with regard to HIV infection in South Africa: implications for HIV prevention. Soc. Behav. Pers., 34(5), 535–544.
Tennenbaum, S., Kassem, T. G., Roudenko, S., & Castillo-Chavez, C. (2006). The role of transactional sex in spreading HIV in Nigeria. American Mathematical Society., 410, 367–389.
Thomas, G., & Lungu, E. M. (2009). The influence of heavy alcohol consumption on HIV infection and progression. J. Biol. Syst., 17(4), 685–712.
UNAIDS (2000) Report on the global HIV/AIDS epidemic. http://data.unaids.org/pub/Report/2000 (accessed 07–02–2012).
UNAIDS (2004) Regional report on HIV/AIDS. http://www.undp.org (accessed 25–11–2011).
UNAIDS (2006a). AIDS epidemic update. http://www.unaids.org (accessed 25–11–2011).
UNAIDS (2006b) Report on the Global HIV/AIDS Epidemic, Joint United Nations Programme on HIV/AIDS. http://www.unaids.org, UNAIDS (accessed 25–11–2011).
Van den Driessche, P., & Watmough, J. (2002). Reproductive numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Math. Biosci., 180, 29–48.
Wechsberg, W. M., Luseno, W. K., & Lam, W. K. (2005). Violence against substance-abusing South African sex workers: intersection with culture and HIV risk. AIDS Care, 17, S55–S64.
Weir, S. S., Pailman, C., Mahlalela, X., Coetzee, N., Meidany, F., & Boerma, J. T. (2003). From people to places: focusing AIDS prevention efforts where it matters most. AIDS, 17, 895–903.
Weiser, S. D., Leiter, K., Heisler, M., McFarland, W., & Percy-de Korte, F. (2006). A population-based study on alcohol and high-risk sexual behaviors in Botswana. PLoS Med., 3(10), 1940–1948.
Zhou, X., & Cui, J. (2011). Modeling and stability analysis for a cholera model with vaccination. Math. Methods Appl. Sci., 34, 1711–1724.
Acknowledgements
N.J.M. acknowledges with thanks the financial support of the National University of Science and Technology and the Research Council of Zimbabwe.
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix A
We now prove result in Lemma 4.
Proof
We make the following change of variables in order to use the Center Manifold theory (Carr 1981) as described in Castillo-Chavez and Song (2004) (Theorem 4.1) to establish the local asymptotic stability of the endemic equilibrium \(S_{f}=x_{1}, I_{f}=x_{2}, A_{f}=x_{3}, S_{m_{1}}=x_{4}, S_{m_{2}}=x_{5}, I_{m_{1}}=x_{6}, I_{m_{2}}=x_{7},\ \mbox{and}\ A_{m}=x_{8}\), so that \(N=\sum^{8}_{n=1}x_{n}\). We now use the vector notation X=(x 1,x 2,x 3,x 4,x 5,x 6,x 7,x 8)T. Then, model system (8) can be written in the form \(\frac{dX}{dt}=F=(f_{1}, f_{2}, f_{3}, f_{4}, f_{5}, f_{6}, f_{7}, f_{8})^{T}\) such that
The Jacobian matrix of transformed system (18) at the disease-free equilibrium is given by

where \(C_{1}=\frac{\pi c_{f}\beta_{m}\rho_{fm_{1}}}{(1-\pi)}\ \mbox{and}\ C_{2}=\frac{(1- \pi)(\alpha+\mu)c_{m}\beta_{f}\rho_{fm_{1}}}{\pi(\alpha+\delta+\mu)}\), and the K i ’s are as defined before. We deduce from the Jacobian that

It was shown in (16) that model system (8) is highly sensitive to changes in \({\mathcal{R}}_{A}\) and therefore its components. Taking β m as a bifurcation point, considering the case \({\mathcal{R}}_{A}=1\), and solving for \(\beta^{*}_{m}\) gives

The linearized system of the transformed equations (18) with \(\beta^{*}_{m}=\beta_{m}\) has a simple zero eigenvalue. Hence, the Center Manifold Theory (Carr 1981) can be used to analyze the dynamics of system (18) near β ∗=β m . It can be shown that the Jacobian of (18) at \(\beta^{*}_{m}=\beta_{m}\) has a right eigenvector associated with the zero eigenvalue given by u=[u 1,u 2,u 3,u 4,u 5,u 6,u 7,u 8]T, where
The left eigenvector of J(ξ 0) associated with the zero eigenvalue at β ∗=β m is given by v=[v 1,v 2,v 3,v 4,v 5,v 6,v 7,v 8], where
For system (18), the associated nonzero partial derivatives of F at the disease-free equilibrium are given by
The bifurcation coefficient a is calculated as follows:

where
□
Appendix B

where \(C_{1}=\vartheta\rho_{fm_{2}}((\alpha+\mu)(\alpha+\gamma+\mu)\rho_{fm_{1}}+(\alpha+\gamma+\delta+\mu)^{2}\rho_{fm_{2}})\) and \(C_{2}=\rho_{fm_{1}}((\alpha+\gamma+\mu)(2\alpha+\gamma+2\delta+\mu)\rho_{fm_{1}}+\delta^{2}\vartheta\rho_{fm_{2}})\).
Rights and permissions
About this article
Cite this article
Malunguza, N.J., Hove-Musekwa, S.D., Musuka, G. et al. Investigating Alcohol Consumption as a Risk Factor for HIV Transmission in Heterosexual Settings in Sub-Saharan African Communities. Bull Math Biol 74, 2094–2124 (2012). https://doi.org/10.1007/s11538-012-9747-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-012-9747-8