Abstract
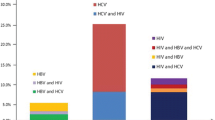
Injection drug users (IDUs) are at risk for infection with hepatitis B virus (HBV), hepatitis C virus (HCV), and human immunodeficiency virus (HIV). Information on time trends in prevalence of these viruses among IDUs and in behaviors influencing their transmission can help define the status of these epidemics and of public health efforts to control them. We conducted a secondary data analysis combining cross-sectional data from IDUs aged 18–30 years enrolled in four Seattle-area studies from 1994 to 2004. Participants in all four studies were tested for antibody to HIV (anti-HIV), hepatitis B core antigen (anti-HBc), and HCV (anti-HCV), and completed behavioral risk assessments. Logistic regression was used to investigate trends in prevalence over time after controlling for sociodemographic, drug use, and sexual behavior variables. Between 1994 and 2004, anti-HBc prevalence declined from 43 to 15% (p < 0.001), anti-HCV prevalence fell from 68 to 32% (p < 0.001) and anti-HIV prevalence remained constant at 2–3%. Declines in anti-HBc and anti-HCV prevalence were observed within the individual studies, although not all these declines were statistically significant. The declines in anti-HBc and anti-HCV prevalence remained significant after control for confounding. Although we did not observe coincident declines in injection equipment sharing practices, there were increases in self-reported needle-exchange use, condom use, and hepatitis B vaccination. We conclude that there has been a substantial and sustained reduction in prevalence rates for HBV and HCV infection among young Seattle IDUs, while HIV rates have remained low and stable.








Similar content being viewed by others
References
Centers for Disease Control and Prevention. HIV/AIDS surveillance Report, 2005. Vol. 17. 2005. Atlanta, GA. U.S. Department of Health and Human Services. Available at: http://www.cdc.gov/hiv/topics/surveillance/resources/reports.
MacDonald M, Crofts N, Kaldor J. Transmission of hepatitis C virus: rates, routes, and cofactors. Epidemiol Rev. 1996;18:137–148.
Nelson KE, Thomas DL. Viral Hepatitis. In: Nelson KE, Williams CM, Graham NMG, eds. Infectious Disease Epidemiology: Theory and Practice. Gaithersburg, MD: Aspen Publishing; 2001:567–609.
Centers for Disease Control and Prevention. Disease burden from hepatitis A, B, and C in the United States. Available at: http://www.cdc.gov/ncidod/diseases/hepatitis/resource/PDFs/disease_burden.pdf. Accessed on February 5, 2007.
Goldstein ST, Alter MJ, Williams IT, et al. Incidence and risk factors for acute hepatitis B in the United States, 1982–1998: implications for vaccination programs. J Infect Dis. 2002;185:713–719.
Centers for Disease Control and Prevention. Hepatitis Surveillance Report No. 61. Atlanta, GA: U.S. Department of Health and Human Services; 2006.
Centers for Disease Control and Prevention. Unpublished data. 2006.
Des Jarlais DC, Diaz T, Perlis T, et al. Variability in the incidence of human immunodeficiency virus, hepatitis B virus, and hepatitis C virus infection among young injecting drug users in New York City. Am J Epidemiol. 2003;157:467–471.
Garfein RS, Vlahov D, Galai N, Doherty MC, Nelson KE. Viral infections in short-term injection drug users: the prevalence of the hepatitis C, hepatitis B, human immunodeficiency, and human T-lymphotropic viruses. Am J Public Health. 1996;86:655–661.
Hagan H, McGough JP, Thiede H, Weiss NS, Hopkins S, Alexander ER. Syringe exchange and risk of infection with hepatitis B and C viruses. Am J Epidemiol. 1999;149:203–213.
Kuo I, Sherman SG, Thomas DL, Strathdee SA. Hepatitis B virus infection and vaccination among young injection and non-injection drug users: missed opportunities to prevent infection. Drug Alcohol Depend. 2004;73:69–78.
Thomas DL, Vlahov D, Solomon L, et al. Correlates of hepatitis C virus infections among injection drug users. Medicine (Baltimore). 1995;74:212–220.
Lorvick J, Kral AH, Seal K, Gee L, Edlin BR. Prevalence and duration of hepatitis C among injection drug users in San Francisco, Calif. Am J Public Health. 2001;91:46–47.
Garfein RS, Doherty MC, Monterroso ER, Thomas DL, Nelson KE, Vlahov D. Prevalence and incidence of hepatitis C virus infection among young adult injection drug users. J Acquir Immune Defic Syndr Hum Retrovirol. 1998;18(Suppl 1):S11–S19.
Hahn JA, Page-Shafer K, Lum PJ, et al. Hepatitis C virus seroconversion among young injection drug users: relationships and risks. J Infect Dis. 2002;186:1558–1564.
Thorpe LE, Ouellet LJ, Hershow R, et al. Risk of hepatitis C virus infection among young adult injection drug users who share injection equipment. Am J Epidemiol. 2002;155:645–653.
Des Jarlais DC, Perlis T, Arasteh K, et al. Reductions in hepatitis C virus and HIV infections among injecting drug users in New York City, 1990–2001. AIDS. 2005;19(Suppl 3):S20–S25.
Hagan H, Thiede H, Des J. Hepatitis C virus infection among injection drug users: survival analysis of time to seroconversion. Epidemiology. 2004;15:543–549.
Friedman SR, Tempalski B, Cooper H, et al. Estimating numbers of injecting drug users in metropolitan areas for structural analyses of community vulnerability and for assessing relative degrees of service provision for injecting drug users. J Urban Health. 2004;81:377–400.
Thiede H, Romero M, Bordelon K, Hagan H, Murrill CS. Using a jail-based survey to monitor HIV and risk behaviors among Seattle area injection drug users. J Urban Health. 2001;78:264–278.
Alter MJ, Kuhnert WL, Finelli L. Guidelines for laboratory testing and result reporting of antibody to hepatitis C virus. Centers for Disease Control and Prevention. MMWR Recomm Rep. 2003;52:1–13, 15.
Breslow NE, Day NE. Statistical Methods in Cancer Research. Lyon: IARC; 1980.
SPSS, version 14. Chicago, IL; 2004.
Villano SA, Vlahov D, Nelson KE, Lyles CM, Cohn S, Thomas DL. Incidence and risk factors for hepatitis C among injection drug users in Baltimore, Maryland. J Clin Microbiol. 1997;35:3274–3277.
Levine OS, Vlahov D, Brookmeyer R, Cohn S, Nelson KE. Differences in the incidence of hepatitis B and human immunodeficiency virus infections among injecting drug users. J Infect Dis. 1996;173:579–583.
Goldberg D, Burns S, Taylor A, Cameron S, Hargreaves D, Hutchinson S. Trends in HCV prevalence among injecting drug users in Glasgow and Edinburgh during the era of needle/syringe exchange. Scand J Infect Dis. 2001;33:457–461.
Crofts N, Aitken CK. Incidence of bloodborne virus infection and risk behaviours in a cohort of injecting drug users in Victoria, 1990–1995. Med J Aust. 1997;167:17–20.
Crofts N, Nigro L, Oman K, Stevenson E, Sherman J. Methadone maintenance and hepatitis C virus infection among injecting drug users. Addiction. 1997;92:999–1005.
Hagan H, Thiede H, Weiss NS, Hopkins SG, Duchin JS, Alexander ER. Sharing of drug preparation equipment as a risk factor for hepatitis C. Am J Public Health. 2001;91:42–46.
Nelson KE, Galai N, Safaeian M, Strathdee SA, Celentano DD, Vlahov D. Temporal trends in the incidence of human immunodeficiency virus infection and risk behavior among injection drug users in Baltimore, Maryland, 1988–1998. Am J Epidemiol. 2002;156:641–653.
Des Jarlais DC, Perlis T, Friedman SR, et al. Behavioral risk reduction in a declining HIV epidemic: injection drug users in New York City, 1990–1997. Am J Public Health. 2000;90:1112–1116.
Des Jarlais DC, Perlis T, Arasteh K, et al. “Informed altruism” and “partner restriction” in the reduction of HIV infection in injecting drug users entering detoxification treatment in New York City, 1990–2001. J Acquir Immune Defic Syndr. 2004;35:158–166.
Centers for Disease Control and Prevention. HBV vaccination among high-risk adolescents and adults—San Diego, Califorinia, 1998–2001. MMWR Morb Mortal Wkly Rep. 2002;51:618–621.
Heimer R, Clair S, Grau LE, Bluthenthal RN, Marshall PA, Singer M. Hepatitis-associated knowledge is low and risks are high among HIV-aware injection drug users in three US cities. Addiction. 2002;97:1277–1287.
Lum PJ, Ochoa KC, Hahn JA, Page SK, Evans JL, Moss AR. Hepatitis B virus immunization among young injection drug users in San Francisco, Calif: the UFO Study. Am J Public Health. 2003;93:919–923.
Seal KH, Ochoa KC, Hahn JA, Tulsky JP, Edlin BR, Moss AR. Risk of hepatitis B infection among young injection drug users in San Francisco: opportunities for intervention. West J Med. 2000;172:16–20.
Centers for Disease Control and Prevention. Hepatitis B virus: a comprehensive strategy for eliminating transmission in the United States through universal childhood vaccination. Recommendations of the Immunization Practices Advisory Committee (ACIP). MMWR Recomm Rep. 1991;40(RR–13):1–19.
Centers for Disease Control and Prevention. Update: Recommendations to prevent HBV virus transmission—United States. MMWR Morb Mortal Wkly Rep. 1995;44:574–575.
Centers for Disease Control and Prevention. Notice to readers update: recommendations to prevent hepatitis B transmission—United States. MMWR Morb Mortal Wkly Rep. 1999;48:33–34.
Metzger DS, Woody GE, McLellan AT, et al. Human immunodeficiency virus seroconversion among intravenous drug users in- and out-of-treatment: an 18-month prospective follow-up. J Acquir Immune Defic Syndr. 1993;6:1049–1056.
Rhodes T, Donoghoe M, Hunter G, Soteri A, Stimson GV. Sexual behaviour of drug injectors in London: implications for HIV transmission and HIV prevention. Addiction. 1994;89:1085–1096.
Tyndall MW, Patrick D, Spittal P, Li K, O’Shaughnessy MV, Schechter MT. Risky sexual behaviours among injection drugs users with high HIV prevalence: implications for STD control. Sex Transm Infect. 2002;78(Suppl 1):i170–i175.
Watkins KE, Metzger D, Woody G, McLellan AT. Determinants of condom use among intravenous drug users. AIDS. 1993;7:719–723.
Des Jarlais DC, Paone D, Milliken J, et al. Audio-computer interviewing to measure risk behaviour for HIV among injecting drug users: a quasi-randomised trial. Lancet. 1999;353:1657–1661.
Acknowledgements
The funding for this work came from the following sources: RAVEN—National Institute on Drug Abuse (1RO1DA08023), Centers for Disease Control and Prevention (U62/CCU006260); RAVEN II—National Institute on Drug Abuse (DA08023, DA 11447 & DA15026); Kiwi—Centers for Disease Control and Prevention (U62/CCU006260); DUIT/CIDUS III—funding was through a cooperative agreement with the Centers for Disease Control and Prevention.
Disclaimer
The findings and conclusion in this report are those of the authors and do not necessarily reflect the views of the Centers for Disease Control and Prevention.
Author information
Authors and Affiliations
Corresponding author
Additional information
Burt and Thiede are with Public Health—Seattle and King County, Seattle, WA, USA; Hagan is with the National Development and Research Institutes, New York, NY, USA; Garfein and Sabin were with the Division of HIV/AIDS Prevention, Centers for Disease Control and Prevention, Atlanta, GA, USA; Garfein is currently with the School of Medicine, University of California at San Diego, San Diego, CA, USA; Sabin is currently with the Global AIDS Program, CDC, Atlanta, GA, USA; Weinbaum is with the Division of Viral Hepatitis, Centers for Disease Control and Prevention, Atlanta, GA, USA.
Rights and permissions
About this article
Cite this article
Burt, R.D., Hagan, H., Garfein, R.S. et al. Trends in Hepatitis B Virus, Hepatitis C Virus, and Human Immunodeficiency Virus Prevalence, Risk Behaviors, and Preventive Measures among Seattle Injection Drug Users Aged 18–30 Years, 1994–2004. J Urban Health 84, 436–454 (2007). https://doi.org/10.1007/s11524-007-9178-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11524-007-9178-2