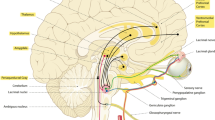
Abstract
Primarily regarded as immune proteins, chemokines are emerging as a family of molecules serving neuromodulatory functions in the developing and adult brain. Among them, CXCL12 is constitutively and widely expressed in the CNS, where it was shown to act on cellular, synaptic, network, and behavioral levels. Its receptor, CXCR4, is abundant in the amygdala, a brain structure involved in pathophysiology of anxiety disorders. Dysregulation of CXCL12/CXCR4 signaling has been implicated in anxiety-related behaviors. Here we demonstrate that exogenous CXCL12 at 2 nM but not at 5 nM increased neuronal excitability in the lateral division of the rat central amygdala (CeL) which was evident in the Late-Firing but not Regular-Spiking neurons. These effects were blocked by AMD3100, a CXCR4 antagonist. Moreover, CXCL12 increased the excitability of the neurons of the basolateral amygdala (BLA) that is known to project to the CeL. However, CXCL12 increased neither the spontaneous excitatory nor spontaneous inhibitory synaptic transmission in the CeL. In summary, the data reveal specific activation of Late-Firing CeL cells along with BLA neurons by CXCL12 and suggest that this chemokine may alter information processing by the amygdala that likely contributes to anxiety and fear conditioning.
Graphical Abstract




Similar content being viewed by others
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Code Availability
The code used for analysis and visualization in the current study was written in R and is available from the corresponding author upon reasonable request.
References
Adler MW, Rogers TJ (2005) Are chemokines the third major system in the brain? J Leukoc Biol 78:1204–1209. https://doi.org/10.1189/jlb.0405222
Bajo M, Varodayan FP, Madamba SG et al (2015) IL-1 interacts with ethanol effects on GABAergic transmission in the mouse central amygdala. Front Pharmacol 6:49. https://doi.org/10.3389/fphar.2015.00049
Banisadr G, Fontanges P, Haour F et al (2002) Neuroanatomical distribution of CXCR4 in adult rat brain and its localization in cholinergic and dopaminergic neurons. Eur J Neurosci 16:1661–1671. https://doi.org/10.1046/j.1460-9568.2002.02237.x
Callewaere C, Banisadr G, Desarménien MG et al (2006) The chemokine SDF-1/CXCL12 modulates the firing pattern of vasopressin neurons and counteracts induced vasopressin release through CXCR4. Proc Natl Acad Sci USA 103:8221–8226. https://doi.org/10.1073/pnas.0602620103
Cassell MD, Gray TS (1989) Morphology of peptide-immunoreactive neurons in the rat central nucleus of the amygdala. J Comp Neurol 281:320–333. https://doi.org/10.1002/cne.902810212
Ciocchi S, Herry C, Grenier F et al (2010) Encoding of conditioned fear in central amygdala inhibitory circuits. Nature 468:277–282. https://doi.org/10.1038/nature09559
Dantzer R (2001) Cytokine-induced sickness behavior: mechanisms and implications. Ann N Y Acad Sci 933:222–234. https://doi.org/10.1111/j.1749-6632.2001.tb05827.x
Dantzer R, Kelley KW (2007) Twenty years of research on cytokine-induced sickness behavior. Brain Behav Immun 21:153–160. https://doi.org/10.1016/j.bbi.2006.09.006
Duvarci S, Pare D (2014) Amygdala microcircuits controlling learned fear. Neuron 82:966–980. https://doi.org/10.1016/j.neuron.2014.04.042
Ehrlich I, Humeau Y, Grenier F et al (2009) Amygdala inhibitory circuits and the control of fear memory. Neuron 62:757–771. https://doi.org/10.1016/j.neuron.2009.05.026
Faber ESL, Sah P (2002) Physiological role of calcium-activated potassium currents in the rat lateral amygdala. J Neurosci 22:1618–1628
Freeman K, Staehle MM, Vadigepalli R et al (2013) Coordinated dynamic gene expression changes in the central nucleus of the amygdala during alcohol withdrawal. Alcohol Clin Exp Res 37 Suppl 1E88–100. https://doi.org/10.1111/j.1530-0277.2012.01910.x
Fumagalli A, Heuninck J, Pizzoccaro A et al (2020) The atypical chemokine receptor 3 interacts with connexin 43 inhibiting astrocytic gap junctional intercellular communication. Nat Commun 11:4855. https://doi.org/10.1038/s41467-020-18634-y
Gilpin NW, Herman MA, Roberto M (2015) The central amygdala as an integrative hub for anxiety and alcohol use disorders. Biol Psychiatry 77:859–869. https://doi.org/10.1016/j.biopsych.2014.09.008
Group PIN, Ascoli GA, Alonso-Nanclares L et al (2008) Petilla terminology: nomenclature of features of GABAergic interneurons of the cerebral cortex. Nat Rev Neurosci 9:557–568. https://doi.org/10.1038/nrn2402
Guyon A, Banisadr G, Rovère C et al (2005) Complex effects of stromal cell-derived factor-1 alpha on melanin-concentrating hormone neuron excitability. Eur J Neurosci 21:701–710. https://doi.org/10.1111/j.1460-9568.2005.03890.x
Guyon A, Skrzydelsi D, Rovère C et al (2006) Stromal cell-derived factor-1alpha modulation of the excitability of rat substantia nigra dopaminergic neurones: presynaptic mechanisms. J Neurochem 96:1540–1550. https://doi.org/10.1111/j.1471-4159.2006.03659.x
Guyon A, Skrzydelski D, Rovère C et al (2008) Stromal-cell-derived factor 1alpha /CXCL12 modulates high-threshold calcium currents in rat substantia nigra. Eur J Neurosci 28:862–870. https://doi.org/10.1111/j.1460-9568.2008.06367.x
Harper KM, Knapp DJ, Todd CA et al (2020) Phenotyping CCL2 containing central amygdala neurons controlling alcohol withdrawal-induced anxiety. Front Cell Neurosci 14:580583. https://doi.org/10.3389/fncel.2020.580583
Haubensak W, Kunwar PS, Cai H et al (2010) Genetic dissection of an amygdala microcircuit that gates conditioned fear. Nature 468:270–276. https://doi.org/10.1038/nature09553
Heinisch S, Kirby LG (2010) SDF-1alpha/CXCL12 enhances GABA and glutamate synaptic activity at serotonin neurons in the rat dorsal raphe nucleus. Neuropharmacology 58:501–514. https://doi.org/10.1016/j.neuropharm.2009.08.022
Heinisch S, Palma J, Kirby LG (2011) Interactions between chemokine and mu-opioid receptors: anatomical findings and electrophysiological studies in the rat periaqueductal grey. Brain Behav Immun 25:360–372. https://doi.org/10.1016/j.bbi.2010.10.020
Herry C, Ferraguti F, Singewald N et al (2010) Neuronal circuits of fear extinction. Eur J Neurosci 31:599–612. https://doi.org/10.1111/j.1460-9568.2010.07101.x
Hou W-H, Kuo N, Fang G-W et al (2016) Wiring specificity and synaptic diversity in the mouse lateral central amygdala. J Neurosci 36:4549–4563. https://doi.org/10.1523/{JNEUROSCI}.3309-15.2016
Hunt S, Sun Y, Kucukdereli H et al (2017) Intrinsic circuits in the lateral central amygdala. Eneuro 4. https://doi.org/10.1523/{ENEURO}.0367-16.2017
Janak PH, Tye KM (2015) From circuits to behaviour in the amygdala. Nature 517:284–292. https://doi.org/10.1038/nature14188
Kalin NH, Shelton SE, Engeland CG et al (2006) Stress decreases, while central nucleus amygdala lesions increase, IL-8 and MIP-1alpha gene expression during tissue healing in non-human primates. Brain Behav Immun 20:564–568. https://doi.org/10.1016/j.bbi.2006.01.003
Kasiyanov A, Fujii N, Tamamura H, Xiong H (2008) Modulation of network-driven, GABA-mediated giant depolarizing potentials by SDF-1alpha in the developing hippocampus. Dev Neurosci 30:285–292. https://doi.org/10.1159/000112520
Krettek JE, Price JL (1978) A description of the amygdaloid complex in the rat and cat with observations on intra-amygdaloid axonal connections. J Comp Neurol 178:255–280. https://doi.org/10.1002/cne.901780205
LeDoux JE (2000) Emotion circuits in the brain. Annu Rev Neurosci 23:155–184. https://doi.org/10.1146/annurev.neuro.23.1.155
Levoye A, Balabanian K, Baleux F et al (2009) CXCR7 heterodimerizes with CXCR4 and regulates CXCL12-mediated g protein signaling. Blood 113:6085–6093. https://doi.org/10.1182/blood-2008-12-196618
Li H, Penzo MA, Taniguchi H et al (2013) Experience-dependent modification of a central amygdala fear circuit. Nat Neurosci 16:332–339. https://doi.org/10.1038/nn.3322
Li Z-Q, Yan Z-Y, Lan F-J et al (2018) Suppression of NLRP3 inflammasome attenuates stress-induced depression-like behavior in NLGN3-deficient mice. Biochem Biophys Res Commun 501:933–940. https://doi.org/10.1016/j.bbrc.2018.05.085
Limatola C, Ciotti MT, Mercanti D et al (2000) The chemokine growth-related gene product beta protects rat cerebellar granule cells from apoptotic cell death through alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionate receptors. Proc Natl Acad Sci USA 97:6197–6201. https://doi.org/10.1073/pnas.090105997
Lopez de Armentia M, Sah P (2004) Firing properties and connectivity of neurons in the rat lateral central nucleus of the amygdala. J Neurophysiol 92:1285–1294. https://doi.org/10.1152/jn.00211.2004
Maes M, Song C, Lin A et al (1998) The effects of psychological stress on humans: increased production of pro-inflammatory cytokines and a Th1-like response in stress-induced anxiety. Cytokine 10:313–318. https://doi.org/10.1006/cyto.1997.0290
Mahan AL, Ressler KJ (2012) Fear conditioning, synaptic plasticity and the amygdala: implications for posttraumatic stress disorder. Trends Neurosci 35:24–35. https://doi.org/10.1016/j.tins.2011.06.007
Martina M, Royer S, Paré D (1999) Physiological properties of central medial and central lateral amygdala neurons. J Neurophysiol 82:1843–1854. https://doi.org/10.1152/jn.1999.82.4.1843
Mattson MP (2008) Hormesis defined. Ageing Res Rev 7:1–7. https://doi.org/10.1016/j.arr.2007.08.007
Meyrath M, Szpakowska M, Zeiner J et al (2020) The atypical chemokine receptor ACKR3/CXCR7 is a broad-spectrum scavenger for opioid peptides. Nat Commun 11:3033. https://doi.org/10.1038/s41467-020-16664-0
Nishiyori A, Minami M, Ohtani Y et al (1998) Localization of fractalkine and CX3CR1 mRNAs in rat brain: does fractalkine play a role in signaling from neuron to microglia? FEBS Lett 429:167–172. https://doi.org/10.1016/s0014-5793(98)00583-3
Patel RR, Khom S, Steinman MQ et al (2019) IL-1expression is increased and regulates GABA transmission following chronic ethanol in mouse central amygdala. Brain Behav Immun 75:208–219. https://doi.org/10.1016/j.bbi.2018.10.009
Penzo MA, Robert V, Tucciarone J et al (2015) The paraventricular thalamus controls a central amygdala fear circuit. Nature 519:455–459. https://doi.org/10.1038/nature13978
Pitkänen A, Savander V, LeDoux JE (1997) Organization of intra-amygdaloid circuitries in the rat: an emerging framework for understanding functions of the amygdala. Trends Neurosci 20:517–523. https://doi.org/10.1016/s0166-2236(97)01125-9
Poon K, Barson JR, Ho HT, Leibowitz SF (2016) Relationship of the chemokine, CXCL12, to effects of dietary fat on feeding-related behaviors and hypothalamic neuropeptide systems. Front Behav Neurosci 10:51. https://doi.org/10.3389/fnbeh.2016.00051
Puchert M, Pelkner F, Stein G et al (2017) Astrocytic expression of the CXCL12 receptor, CXCR7/ACKR3 is a hallmark of the diseased, but not developing CNS. Mol Cell Neurosci 85:105–118. https://doi.org/10.1016/j.mcn.2017.09.001
R Core Team (2021) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Ragozzino D, Giovannelli A, Mileo AM et al (1998) Modulation of the neurotransmitter release in rat cerebellar neurons by GRO beta. NeuroReport 9:3601–3606. https://doi.org/10.1097/00001756-199811160-00011
Réaux-Le Goazigo A, Van Steenwinckel J, Rostène W, Mélik Parsadaniantz S (2013) Current status of chemokines in the adult CNS. Prog Neurobiol 104:67–92. https://doi.org/10.1016/j.pneurobio.2013.02.001
Roozendaal B, McEwen BS, Chattarji S (2009) Stress, memory and the amygdala. Nat Rev Neurosci 10:423–433. https://doi.org/10.1038/nrn2651
Rostène W, Guyon A, Kular L et al (2011) Chemokines and chemokine receptors: new actors in neuroendocrine regulations. Front Neuroendocrinol 32:10–24. https://doi.org/10.1016/j.yfrne.2010.07.001
Sah P, Lopez De Armentia M (2003) Excitatory synaptic transmission in the lateral and central amygdala. Ann N Y Acad Sci 985:67–77. https://doi.org/10.1111/j.1749-6632.2003.tb07072.x
Sah P, Faber ESL, Lopez De Armentia M, Power J (2003) The amygdaloid complex: anatomy and physiology. Physiol Rev 83:803–834. https://doi.org/10.1152/physrev.00002.2003
Sanchez-Alavez M, Nguyen W, Mori S, Alcoholism et al (2019) Clin Experimental Res 43:2547–2558. https://doi.org/10.1111/acer.14209
Schönemeier B, Kolodziej A, Schulz S et al (2008) Regional and cellular localization of the CXCl12/SDF-1 chemokine receptor CXCR7 in the developing and adult rat brain. J Comp Neurol 510:207–220. https://doi.org/10.1002/cne.21780
Sheridan GK, Wdowicz A, Pickering M et al (2014) CX3CL1 is up-regulated in the rat hippocampus during memory-associated synaptic plasticity. Front Cell Neurosci 8:233. https://doi.org/10.3389/fncel.2014.00233
Shi L, Bi Q, Li W et al (2016) CXCL12 impairs the acquisition and extinction of auditory fear conditioning in rats via crosstalk with GABAergic system. Pharmacol Biochem Behav 148:21–27. https://doi.org/10.1016/j.pbb.2016.05.008
Skelly DT, Hennessy E, Dansereau M-A, Cunningham C (2013) A systematic analysis of the peripheral and CNS effects of systemic LPS, IL-1, [corrected] TNF-and IL-6 challenges in C57BL/6 mice. PLoS ONE 8:e69123. https://doi.org/10.1371/journal.pone.0069123
Sosulina L, Meis S, Seifert G et al (2006) Classification of projection neurons and interneurons in the rat lateral amygdala based upon cluster analysis. Mol Cell Neurosci 33:57–67. https://doi.org/10.1016/j.mcn.2006.06.005
Sowa JE, Tokarski K (2021) Cellular, synaptic, and network effects of chemokines in the central nervous system and their implications to behavior. Pharmacol Rep. https://doi.org/10.1007/s43440-021-00323-2
Stuart MJ, Singhal G, Baune BT (2015) Systematic review of the neurobiological relevance of chemokines to psychiatric disorders. Front Cell Neurosci 9:357. https://doi.org/10.3389/fncel.2015.00357
Stumm RK, Rummel J, Junker V et al (2002) A dual role for the SDF-1/CXCR4 chemokine receptor system in adult brain: Isoform-selective regulation of SDF-1 expression modulates CXCR4-dependent neuronal plasticity and cerebral leukocyte recruitment after focal ischemia. J Neurosci 22:5865–5878. https://doi.org/10.1523/JNEUROSCI.22-14-05865.2002
Stumm RK, Zhou C, Ara T et al (2003) CXCR4 regulates interneuron migration in the developing neocortex. J Neurosci 23:5123–5130
Tarozzo G, Bortolazzi S, Crochemore C et al (2003) Fractalkine protein localization and gene expression in mouse brain. J Neurosci Res 73:81–88. https://doi.org/10.1002/jnr.10645
Trettel F, Di Castro MA, Limatola C (2020) Chemokines: key molecules that orchestrate communication among neurons, microglia and astrocytes to preserve brain function. Neuroscience 439:230–240. https://doi.org/10.1016/j.neuroscience.2019.07.035
Tye KM, Prakash R, Kim S-Y et al (2011) Amygdala circuitry mediating reversible and bidirectional control of anxiety. Nature 471:358–362. https://doi.org/10.1038/nature09820
Washburn MS, Moises HC (1992) Inhibitory responses of rat basolateral amygdaloid neurons recorded in vitro. Neuroscience 50:811–830. https://doi.org/10.1016/0306-4522(92)90206-h
Wickham H (2016) ggplot2: elegant graphics for data analysis. Springer-, New York
Wickham H, François R, Henry L, Müller K (2021) Dplyr: A grammar of data manipulation
Williams JL, Holman DW, Klein RS (2014) Chemokines in the balance: maintenance of homeostasis and protection at CNS barriers. Front Cell Neurosci 8:154. https://doi.org/10.3389/fncel.2014.00154
Winchester G, Liu S, Steele OG et al (2020) Eventer. Software for the detection of spontaneous synaptic events measured by electrophysiology or imaging. https://doi.org/10.5281/zenodo.3991677. Version 1.0.1. Zenodo. URL
Yang L, Wang M, Guo YY et al (2016) Systemic inflammation induces anxiety disorder through CXCL12/CXCR4 pathway. Brain Behav Immun 56:352–362. https://doi.org/10.1016/j.bbi.2016.03.001
Funding
This work was supported by the National Science Center grant number 2016/21/N/NZ4/03621 and 2019/32/T/NZ4/00592.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study’s conception and design. Joanna Ewa Sowa performed the experiments, analyzed the data, and wrote the first draft of the manuscript. Grzegorz Hess commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical Approval
All protocols complied with the European Union 2010 Animal Welfare Act and national law and were approved by the Local Ethical Committee for Animal Experiments at the Institute of Pharmacology, Polish Academy of Sciences in Krakow.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sowa, J.E., Tokarski, K. & Hess, G. Activation of the CXCR4 Receptor by Chemokine CXCL12 Increases the Excitability of Neurons in the Rat Central Amygdala. J Neuroimmune Pharmacol 19, 9 (2024). https://doi.org/10.1007/s11481-024-10112-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11481-024-10112-2