Abstract
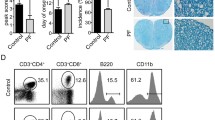
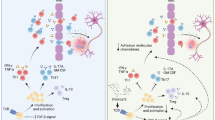
Multiple sclerosis (MS) is the prototypical inflammatory demyelinating disease of the central nervous system (CNS), and MS results in physical and cognitive impairments, such as fatigue, pain, depression and bladder dysfunction. Though many therapies for MS have been developed, the safety profile and effectiveness of these therapies still need to be defined. Thus, new therapies for MS must be explored. Celastrol, a quinonemethide triterpene, is a pharmacologically active compound present in Thunder God Vine root extracts used to treat inflammatory and autoimmune diseases. Molecular studies have identified several molecular targets, which are mostly centered on the inhibition of IKK-NF-κB signaling. The animal model of experimental autoimmune encephalomyelitis (EAE) has been widely used in MS studies; thus, we tried to explore the role of celastrol in EAE development in this study. We demonstrated that the intraperitoneal injection of celastrol significantly attenuated EAE disease. Th17 cell responses in the peripheral lymph nodes in EAE mice were also inhibited by celastrol. We determined that celastroldownregulated cytokine production in bone-marrow derived dendritic cells (BMDCs). Accordingly, T cells that were co-cultured with either BMDCs pre-treated with celastrolor splenic DCs and then collected on day 7 after EAE immunizationshowed that Th17 cell polarization is suppressed in the above two situations. Moreover, celastrol was required for tissue-infiltrating DCs to sustain Th17 responses in the central nervous system (CNS). Taken together, the results of our study demonstrate that celastrol ameliorates EAE development by suppressing pathogenic Th17 responses; this finding offers a better understanding of the role of celastrol in EAE development as well as new proposals for clinical interventions.






Similar content being viewed by others
References
Abdin AA, Hasby EA (2014) Modulatory effect of celastrol on Th1/Th2 cytokines profile, TLR2 and CD3+ T-lymphocyte expression in a relapsing-remitting model of multiple sclerosis in rats. Eur J Pharmacol. doi:10.1016/j.ejphar.2014.09.001
Allison AC, Cacabelos R, Lombardi VR, Alvarez XA, Vigo C (2001) Celastrol, a potent antioxidant and anti-inflammatory drug, as a possible treatment for Alzheimer’s disease. Prog Neuropsychopharmacol Biol Psychiatry 25:1341–1357
Bailey SL, Schreiner B, McMahon EJ, Miller SD (2007) CNS myeloid DCs presenting endogenous myelin peptides ‘preferentially’ polarize CD4+ T(H)-17 cells in relapsing EAE. Nat Immunol 8:172–180. doi:10.1038/ni1430
Bettelli E et al (2006) Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature 441:235–238. doi:10.1038/nature04753
Carrillo-Salinas FJ et al (2014) A cannabigerol derivative suppresses immune responses and protects mice from experimental autoimmune encephalomyelitis. PLoS One 9:e94733. doi:10.1371/journal.pone.0094733
Carroll WM (2010) Oral therapy for multiple sclerosis–sea change or incremental step? N Engl J Med 362:456–458. doi:10.1056/NEJMe0912019
Choi JW et al (2011) FTY720 (fingolimod) efficacy in an animal model of multiple sclerosis requires astrocyte sphingosine 1-phosphate receptor 1 (S1P1) modulation. Proc Natl Acad Sci U S A 108:751–756. doi:10.1073/pnas.1014154108
Chun J, Hartung HP (2010) Mechanism of action of oral fingolimod (FTY720) in multiple sclerosis. Clin Neuropharmacol 33:91–101. doi:10.1097/WNF.0b013e3181cbf825
Chung Y et al (2009) Critical regulation of early Th17 cell differentiation by interleukin-1 signaling. Immunity 30:576–587. doi:10.1016/j.immuni.2009.02.007
Comi G et al (2010) Phase II study of oral fingolimod (FTY720) in multiple sclerosis: 3-year results. Mult Scler 16:197–207. doi:10.1177/1352458509357065
Compston A, Coles A (2008) Multiple sclerosis. Lancet 372:1502–1517. doi:10.1016/s0140-6736(08)61620-7
Constantinescu CS, Farooqi N, O’Brien K, Gran B (2011) Experimental autoimmune encephalomyelitis (EAE) as a model for multiple sclerosis (MS). Br J Pharmacol 164:1079–1106. doi:10.1111/j.1476-5381.2011.01302.x
Cua DJ et al (2003) Interleukin-23 rather than interleukin-12 is the critical cytokine for autoimmune inflammation of the brain. Nature 421:744–748. doi:10.1038/nature01355
Diebold SS (2008) Determination of T-cell fate by dendritic cells. Immunol Cell Biol 86:389–397. doi:10.1038/icb.2008.26
Dong C (2008) TH17 cells in development: an updated view of their molecular identity and genetic programming. Nat Rev Immunol 8:337–348. doi:10.1038/nri2295
Edwards LJ, Robins RA, Constantinescu CS (2010) Th17/Th1 phenotype in demyelinating disease. Cytokine 50:19–23. doi:10.1016/j.cyto.2009.12.003
Engelmann C, Weih F, Haenold R (2014) Role of nuclear factor kappa B in central nervous system regeneration. Neural Regen Res 9:707–711. doi:10.4103/1673-5374.131572
Eroglu B et al (2014) Therapeutic inducers of the HSP70/HSP110 protect mice against traumatic brain injury. J Neurochem 130:626–641. doi:10.1111/jnc.12781
Faust K, Gehrke S, Yang Y, Yang L, Beal MF, Lu B (2009) Neuroprotective effects of compounds with antioxidant and anti-inflammatory properties in a Drosophila model of Parkinson’s disease. BMC Neurosci 10:109. doi:10.1186/1471-2202-10-109
Gilles L et al (2009) MAL/SRF complex is involved in platelet formation and megakaryocyte migration by regulating MYL9 (MLC2) and MMP9. Blood 114:4221–4232. doi:10.1182/blood-2009-03-209932
Goodin DS et al (2002) Disease modifying therapies in multiple sclerosis: report of the Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology and the MS Council for Clinical Practice Guidelines. Neurology 58:169–178
Goverman J (2009) Autoimmune T cell responses in the central nervous system. Nat Rev Immunol 9:393–407. doi:10.1038/nri2550
Greter M et al (2005) Dendritic cells permit immune invasion of the CNS in an animal model of multiple sclerosis. Nat Med 11:328–334. doi:10.1038/nm1197
Harari D et al (2014) Enhanced In Vivo Efficacy of a Type I Interferon Superagonist with Extended Plasma Half-life in a Mouse Model of Multiple Sclerosis. J Biol Chem. doi:10.1074/jbc.M114.602474
Huang G, Wang Y, Vogel P, Kanneganti TD, Otsu K, Chi H (2012) Signaling via the kinase p38alpha programs dendritic cells to drive TH17 differentiation and autoimmune inflammation. Nat Immunol 13:152–161. doi:10.1038/ni.2207
Iwasaki A, Medzhitov R (2010) Regulation of adaptive immunity by the innate immune system. Science 327:291–295. doi:10.1126/science.1183021
Kannaiyan R, Shanmugam MK, Sethi G (2011) Molecular targets of celastrol derived from Thunder of God Vine: potential role in the treatment of inflammatory disorders and cancer. Cancer Lett 303:9–20. doi:10.1016/j.canlet.2010.10.025
Kebir H et al (2007) Human TH17 lymphocytes promote blood–brain barrier disruption and central nervous system inflammation. Nat Med 13:1173–1175. doi:10.1038/nm1651
Kiaei M, Kipiani K, Petri S, Chen J, Calingasan NY, Beal MF (2005) Celastrol blocks neuronal cell death and extends life in transgenic mouse model of amyotrophic lateral sclerosis. Neurodegener Dis 2:246–254. doi:10.1159/000090364
Kim DH, Shin EK, Kim YH, Lee BW, Jun JG, Park JH, Kim JK (2009a) Suppression of inflammatory responses by celastrol, a quinone methide triterpenoid isolated from Celastrus regelii. Eur J Clin Invest 39:819–827. doi:10.1111/j.1365-2362.2009.02186.x
Kim DY, Park JW, Jeoung D, Ro JY (2009b) Celastrol suppresses allergen-induced airway inflammation in a mouse allergic asthma model. Eur J Pharmacol 612:98–105. doi:10.1016/j.ejphar.2009.03.078S0014-2999(09)00311-2
Kim JE et al (2013) Celastrol, an NF-kappaB inhibitor, improves insulin resistance and attenuates renal injury in db/db mice. PLoS One 8:e62068. doi:10.1371/journal.pone.0062068
Komiyama Y et al (2006) IL-17 plays an important role in the development of experimental autoimmune encephalomyelitis. J Immunol 177:566–573
Krakowski M, Owens T (1996) Interferon-gamma confers resistance to experimental allergic encephalomyelitis. Eur J Immunol 26:1641–1646. doi:10.1002/eji.1830260735
Kryczek I, Wei S, Vatan L, Escara-Wilke J, Szeliga W, Keller ET, Zou W (2007) Cutting edge: opposite effects of IL-1 and IL-2 on the regulation of IL-17+ T cell pool IL-1 subverts IL-2-mediated suppression. J Immunol 179:1423–1426
Langrish CL et al (2005) IL-23 drives a pathogenic T cell population that induces autoimmune inflammation. J Exp Med 201:233–240. doi:10.1084/jem.20041257
Lee JH et al (2006) Inhibition of NF-kappa B activation through targeting I kappa B kinase by celastrol, a quinone methide triterpenoid. Biochem Pharmacol 72:1311–1321. doi:10.1016/j.bcp.2006.08.014
Li H, Zhang YY, Tan HW, Jia YF, Li D (2008) Therapeutic effect of tripterine on adjuvant arthritis in rats. J Ethnopharmacol 118:479–484. doi:10.1016/j.jep.2008.05.028
Lock C et al (2002) Gene-microarray analysis of multiple sclerosis lesions yields new targets validated in autoimmune encephalomyelitis. Nat Med 8:500–508. doi:10.1038/nm0502-500
McMahon EJ, Bailey SL, Castenada CV, Waldner H, Miller SD (2005) Epitope spreading initiates in the CNS in two mouse models of multiple sclerosis. Nat Med 11:335–339. doi:10.1038/nm1202
McPherson RC, Anderton SM (2013) Adaptive immune responses in CNS autoimmune disease: mechanisms and therapeutic opportunities. J Neuroimmune Pharmacol 8:774–790. doi:10.1007/s11481-013-9453-9
Mills KH (2008) Induction, function and regulation of IL-17-producing T cells. Eur J Immunol 38:2636–2649. doi:10.1002/eji.200838535
Murphy CA et al (2003) Divergent pro- and antiinflammatory roles for IL-23 and IL-12 in joint autoimmune inflammation. J Exp Med 198:1951–1957. doi:10.1084/jem.20030896
Nanjundaiah SM, Venkatesha SH, Yu H, Tong L, Stains JP, Moudgil KD (2012) Celastrus and its bioactive celastrol protect against bone damage in autoimmune arthritis by modulating osteoimmune cross-talk. J Biol Chem 287:22216–22226. doi:10.1074/jbc.M112.356816
Park H et al (2005) A distinct lineage of CD4 T cells regulates tissue inflammation by producing interleukin 17. Nat Immunol 6:1133–1141. doi:10.1038/ni1261
Peng B, Xu L, Cao F, Wei T, Yang C, Uzan G, Zhang D (2010) HSP90 inhibitor, celastrol, arrests human monocytic leukemia cell U937 at G0/G1 in thiol-containing agents reversible way. Mol Cancer 9:79. doi:10.1186/1476-4598-9-79
Peterson JW, Bo L, Mork S, Chang A, Trapp BD (2001) Transected neurites, apoptotic neurons, and reduced inflammation in cortical multiple sclerosis lesions. Ann Neurol 50:389–400
Popescu BF, Lucchinetti CF (2012) Pathology of demyelinating diseases. Annu Rev Pathol 7:185–217. doi:10.1146/annurev-pathol-011811-132443
Popescu BF, Pirko I, Lucchinetti CF (2013) Pathology of multiple sclerosis: where do we stand? Continuum (Minneapolis, Minn) 19:901–921. doi:10.1212/01.con.0000433291.23091.65
Sethi G, Ahn KS, Pandey MK, Aggarwal BB (2007) Celastrol, a novel triterpene, potentiates TNF-induced apoptosis and suppresses invasion of tumor cells by inhibiting NF-kappaB-regulated gene products and TAK1-mediated NF-kappaB activation. Blood 109:2727–2735. doi:10.1182/blood-2006-10-050807
Veldhoen M, Hocking RJ, Atkins CJ, Locksley RM, Stockinger B (2006) TGFbeta in the context of an inflammatory cytokine milieu supports de novo differentiation of IL-17-producing T cells. Immunity 24:179–189. doi:10.1016/j.immuni.2006.01.001
Venkatesha SH, Astry B, Nanjundaiah SM, Yu H, Moudgil KD (2012) Suppression of autoimmune arthritis by Celastrus-derived Celastrol through modulation of pro-inflammatory chemokines. Bioorg Med Chem 20:5229–5234. doi:10.1016/j.bmc.2012.06.050
Vermeire K, Heremans H, Vandeputte M, Huang S, Billiau A, Matthys P (1997) Accelerated collagen-induced arthritis in IFN-gamma receptor-deficient mice. J Immunol 158:5507–5513
Wan Y et al (2010) Cognitive decline following major surgery is associated with gliosis, beta-amyloid accumulation, and tau phosphorylation in old mice. Crit Care Med 38:2190–2198. doi:10.1097/CCM.0b013e3181f17bcb
Wang X, Cattaneo F, Ryno L, Hulleman J, Reixach N, Buxbaum JN (2014) The systemic amyloid precursor transthyretin (TTR) behaves as a neuronal stress protein regulated by HSF1 in SH-SY5Y human neuroblastoma cells and APP23 Alzheimer’s disease model mice. J Neurosci: Off J Soc Neurosci 34:7253–7265. doi:10.1523/jneurosci. 4936-13.2014
Wu F, Han M, Wilson JX (2009) Tripterine prevents endothelial barrier dysfunction by inhibiting endogenous peroxynitrite formation. Br J Pharmacol 157:1014–1023. doi:10.1111/j.1476-5381.2009.00292.x
Yednock TA, Cannon C, Fritz LC, Sanchez-Madrid F, Steinman L, Karin N (1992) Prevention of experimental autoimmune encephalomyelitis by antibodies against alpha 4 beta 1 integrin. Nature 356:63–66. doi:10.1038/356063a0
Zepp J, Wu L, Li X (2011) IL-17 receptor signaling and T helper 17-mediated autoimmune demyelinating disease. Trends Immunol 32:232–239. doi:10.1016/j.it.2011.02.007
Zhang DH et al (2006) Tripterine inhibits the expression of adhesion molecules in activated endothelial cells. J Leukoc Biol 80:309–319. doi:10.1189/jlb.1005611
Acknowledgments
This work is supported by supported by the National Natural Science Foundation of China (No. 81400793 for Y. W.and No. 81102349 for B. P.); the Shanghai Pudong District Science and Technology Innovation Project (No. PKJ2013-Y03for Y. W.); Shanghai Gongli Hospital Youth Project (No. 2013GLQN05 forL. C.); Key Discipline Construction Project of Pudong Health Bureau of Shanghai (PWZx2014-03 for L-m. X.); Shanghai Pudong Youth Talent Project in Medicine (No. PWRq2013-11for F-f. C.);the Project of Excellent Academic Leader in Medicine of Shanghai (No. XBR2011054 for D-h. Z) and the Key Discipline Construct Project of Pudong Healthy Bureau of Shanghai (No. PWZx2014-03). We thank Mr. Steven Schwab for tidying up the English expressions and grammar.
Conflict of Interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, Y., Cao, L., Xu, Lm. et al. Celastrol Ameliorates EAE Induction by Suppressing Pathogenic T Cell Responses in the Peripheral and Central Nervous Systems. J Neuroimmune Pharmacol 10, 506–516 (2015). https://doi.org/10.1007/s11481-015-9598-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11481-015-9598-9