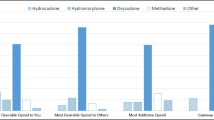
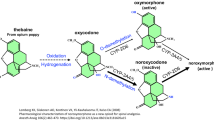
Abstract
Oxycodone is an important therapeutic agent in the treatment of pain. However, its excessive use in medical practice, as well as escalating rates of illegal sharing and high potential for abuse, has generated a public health crisis in North America. The objective of this review is to discuss the prevalence, clinical utility, pharmacology, pharmacokinetics/pharmacodynamics, and addictive properties of oxycodone. Oxycodone has an abuse liability that is similar to other opioids, and in some cases, even greater. The introduction to oxycodone use can come from peers at school, during the treatment of pain, or from the black market. Implementing approaches that increase training of doctors in conjunction with more information for patients, as well as employing harm reduction strategies, diversion resistant products, and drug monitoring programs, may help to reduce the risk of oxycodone misuse and abuse.
Similar content being viewed by others
References
American Psychiatric Association. (2013). Opioid use disorder. In Diagnostic and statistical manual of mental disorders (5th ed.). Washington, DC: American Psychiatric Publishing.
Babalonis, S., Lofwall, M. R., Nuzzo, P. A., Siegel, A. J., & Walsh, S. L. (2013). Abuse liability and reinforcing efficacy of oral tramadol in humans. Drug and Alcohol Dependence, 129, 116–124.
Basbaum, A.I., & Jessell, T.M. (2013). Pain. In E.R. Kandel, et al. (Eds.), Principles of neural science (5th ed.). New York: McGraw Hill.
Beardsley, P. M., Aceto, M. D., Cook, C. D., Bowman, E. R., Newman, J. L., & Harris, L. S. (2004). Discriminative stimulus, reinforcing, physical dependence, and antinociceptive effects of oxycodone in mice, rats, and rhesus monkeys. Experimental and Clinical Psychopharmacology, 12(3), 163–172.
Bell, J. (2010). The global diversion of pharmaceutical drugs. Addiction, 105(9), 1531–1537.
Berridge, K. C., & Robinson, T. E. (1998). What is the role of dopamine in reward: hedonic impact, reward learning, or incentive salience? Brain Research. Brain Research Reviews, 28(3), 309–369.
Betourne, A., Familiades, J., Lacassagne, L., Halley, H., Cazales, M., Ducommun, B., Lassalle, J. M., Zajac, J. M., & Frances, B. (2008). Decreased motivational properties of morphine in mouse models of cancerous- or inflammatory-chronic pain: implication of supraspinal neuropeptide FF(2) receptors. Neuroscience, 157, 12–21.
Butler, S. F., Cassidy, T. A., Chilcoat, H., Black, R. A., Landau, C., Budman, S. H., & Coplan, P. M. (2013). Abuse rates and routes of administration of reformulated extended-release oxycodone: initial findings from a sentinel surveillance sample of individuals assessed for substance abuse treatment. The Journal of Pain, 14(4), 351–358.
Campbell, A. T., Kwiatkowski, D., Boughner, E., & Leri, F. (2012). Effect of yohimbine stress on reacquisition of oxycodone seeking in rats. Psychopharmacology, 222(2), 247–255.
Canadian Centre on Substance Abuse (CCSA). (2015). Prescription opioids. Ottawa: Canadian Centre on Substance Abuse.
Carmichael, J. P., & Lee, M. A. (2010). Symptoms of opioid withdrawal syndrome afterswitch from oxycodone to alfentanil. Journal of Pain and Symptom Management, 40(6), e4–e6.
Casey, B. J., Jones, R. M., & Hare, T. A. (2008). The adolescent brain. Annals of the New York Academy of Sciences, 1124, 111–126.
Centers for Disease Control and Prevention (CDC). (2014). Opioid painkiller prescribing. Retrieved from https://www.cdc.gov/vitalsigns/opioid-prescribing/.
Chakravarthy, B., Shah, S., & Lotfipour, S. (2011). Prescription drug monitoring programs and other interventions to combat prescription opioid abuse. Western Journal of Emergency Medicine, 13(5), 422–425.
Ciccarone, D. (2009). Heroin in brown, black and white: Structural factors and medical consequences in the US heroin market. International Journal of Drug Policy, 20(3), 277–282.
Ciccarone, D., Unick, G. J., & Kraus, A. (2009). Impact of South American heroin on the US heroin market 1993–2004. International Journal of Drug Policy, 20(5), 392–401.
Cicero, T. J., & Ellis, M. S. (2015). Abuse-deterrent formulations and the prescription opioid abuse epidemic in the United States. JAMA Psychiatry, 72(5), 424.
Cicero, T. J., Kurtz, S. P., Surratt, H. L., Ibanez, G. E., Ellis, M. S., Levi-Minzi, M. A., & Inciardi, J. A. (2011). Multiple determinants of specific modes of prescription opioid diversion. Journal of Drug Issues, 41(2), 283–304.
Cicero, T. J., Ellis, M. S., & Surratt, H. L. (2012). Effect of abuse-deterrent formulation of OxyContin. The New England Journal of Medicine, 367, 187–189.
Cicero, T. J., Ellis, M. S., Surratt, H. L., & Kurtz, S. P. (2014). The changing face of heroin use in the United States. JAMA Psychiatry, 71(7), 821.
Colucci, S. V., Perrino, P. J., Shram, M., Bartlett, C., Wang, Y., & Harris, S. C. (2014). Abuse potential of intravenous oxycodone/naloxone solution in nondependent recreational drug users. Clinical Drug Investigation, 34(6), 421–429.
Comer, S. D., Sullivan, M. A., Whittington, R. A., Vosburg, S. K., & Kowalczyk, W. J. (2008). Abuse liability of prescription opioids compared to heroin in morphine-maintained heroin abusers. Neuropsychopharmacology, 33(5), 1179–1191.
Comer, S. D., Sullivan, M. A., Vosburg, S. K., Kowalczyk, W. J., & Houser, J. (2010). Oxycodone: Laboratory study of the relationship between pain and abuse liability. Drug and AlocoholDependence, 109, 130–138.
Comer, S. D., Metz, V. E., Cooper, Z. D., Kowalczyk, W. J., Jones, J. D., Sullivan, M. A., et al. (2013). Comparison of a drug versus money and drug versus drug self-administration choice procedure with oxycodone and morphine in opioid addicts. Behavioural Pharmacology, 24, 504–516.
Coplan, P. M., Kale, H., Sandstrom, L., Landau, C., & Chilcoat, H. D. (2013). Changes in oxycodone and heroin exposures in the National Poison Data System after introduction of extended-release oxycodone with abuse-deterrent characteristics. Pharmacoepidemiology and Drug Safety, 22(12), 1274–1282.
Dart, R. C., Surratt, H. L., Cicero, T. J., Parrino, M. W., Severtson, S. G., Bartelson, B. B., & Green, J. L. (2015). Trends in opioid analgesic abuse and mortality in the United States. New England Journal of Medicine, 372(16), 1572–1574.
Dasgupta, N., Freifeld, C., Brownstein, J. S., Menone, C. M., Surratt, H. L., Poppish, L., et al. (2013). Crowdsourcing black market prices for prescription opioids. Journal of Medical Internet Research, 15(8), e178.
Davis, W. R., & Johnson, B. D. (2008). Prescription opioid use, misuse, and diversion among street drug users in New York City. Drug and Alcohol Dependence, 92(1–3), 267–276.
Davis, C. P., Franklin, L. M., Johnson, G. S., & Schrott, L. M. (2010). Prenatal oxycodone exposure impairs spatial learning and/or memory in rats. Behavioural Brain Research, 212(1), 27–34.
Dhalla, I. A., Mamdani, M. M., Sivilotti, M. L., Kopp, A., Qureshi, O., & Juurlink, D. N. (2009). Prescribing of opioid analgesics and related mortality before and after the introduction of long-acting oxycodone. CMAJ, 181(12), 891–896.
Drug Enforcement Administration. (2014). Oxycodone. Sprinfield, Virginia: Drug Enforcement Administration.
Edlund, M. J., Steffick, D., Hudson, T., Harris, K. M., & Sullivan, M. (2007). Risk factors for clinically recognized opioid abuse and dependence among veterans using opioids for chronic non-cancer pain. Pain, 129, 355–362.
Emery, M. A., Bates, M. L. S., Wellman, P. J., & Eitan, S. (2015). Differential effects of oxycodone, hydrocodone, and morphine on the responses of D2/D3 dopamine receptors. Behavioural Brain Research, 284, 37–41.
Emery, M. A., Bates, S., Wellman, P. J., & Eitan, S. (2016). Differential effects of oxycodone, hydrocodone, and morphine on activation levels of signaling molecules. Pain Medicine, 17(5), 908–914.
Enga, R. M., Jackson, A., Damaj, M. I., & Beardsley, P. M. (2016). Oxycodone physical dependence and its oral self-administration in C57BL/6J mice. European Journal of Pharmacology, 789, 75–80.
Everitt, B. J., & Robbins, T. W. (2005). Neural systems of reinforcement for drug addiction: from actions to habits to compulsion. Nature Neuroscience, 8(11), 1481–1489.
Frank, D., Mateu-Gelabert, P., Guarino, H., Bennett, A., Wendel, T., Jessell, L., & Teper, A. (2015). High risk and little knowledge: overdose experiences and knowledge among young adult nonmedical prescription opioid users. International Journal of Drug Policy, 26(1), 84–91.
Gerak, L. R., Galici, R., & France, C. P. (2009). Self-administration of heroin and cocaine in morphine-dependent and morphine-withdrawn rhesus monkeys. Psychopharmacology, 204, 403–411.
Gibson, A. E., & Degenhardt, L. J. (2007). Mortality related to pharmacotherapies for opioid dependence: a comparative analysis of coronial records. Drug and Alcohol Review, 26(4), 405–410.
Gomes, T., & Juurlink, D. (2016). Opioid use and overdose: what we’ve learned in Ontario. Healthcare Quarterly, 18(4), 8–11.
Goodman & Gilman’s. (2001). The Pharmacological Basis of Therapeutics. In J. G. Hardman, L. E. Limbird, & A. G. Gilman (Eds.), (10th ed.). New York: McGraw-Hill.
Goodman & Gilman’s. (2011). The Pharmacological Basis of Therapeutics. In L. L. Brunton, J. S. Lazo, & K. L. Parker (Eds.), (11th ed.). New York: McGraw-Hill.
Gossop, M., Green, L., Phillips, G., & Bradley, B. (1989). Lapse, relapse and survival among opiate addicts after treatment. A prospective follow-up study. The British Journal of Psychiatry, 154, 348–353.
Gossop, M., Stewart, D., Browne, N., & Marsden, J. (2002). Factors associated with abstinence, lapse or relapse to heroin use after residential treatment: protective effect of coping responses. Addiction, 97(10), 1259–1267.
Gould, T. J. (2010). Addiction and cognition. Addict SciClinPract, 5(2), 4–14.
Grau, L. E., Dasgupta, N., Harvey, A. P., Irwin, K., Givens, A., Kinzly, M. L., & Heimer, R. (2007). Illicit use of opioids: Is OxyContin® a “gateway drug”? The American Journal on Addictions, 16(3), 166–173.
Grella, S. L., Levy, A., Campbell, A., Djazayeri, S., Allen, C. P., Goddard, B., & Leri, F. (2011). Oxycodone dose-dependently imparts conditioned reinforcing properties to discrete sensory stimuli in rats. Pharmacological Research, 64(4), 364–370.
Gwira, J. A., Wiedeman, C., Dunn, J. R., et al. (2014). High-risk use by patients prescribed opioids for pain and its role in overdose deaths. JAMA Internal Medicine, 174(5), 796–801.
Hagemeier, N. E., Gray, J. A., & Pack, R. P. (2013). Prescription drug abuse: a comparison of prescriber and pharmacist perspective. Substance Use & Misuse, 48(9), 761–768.
Harris, S. C., Perrino, P. J., Smith, I., Shram, M. J., Colucci, S. V., Bartlett, C., & Sellers, E. M. (2013). Abuse potential, pharmacokinetics, pharmacodynamics, and safety of intranasallyadministered crushed oxycodone HCl abuse-deterrent controlled-release tablets in recreational opioid users. The Journal of Clinical Pharmacology, 54(4), 468–477.
Health Canada. (2017). New measures to inform Canadians of the risks of prescription opioids out for consultation. Retrieved from https://www.canada.ca/en/health-canada/news/2017/06/new_measures_to_informcanadiansoftherisksofprescriptionopioidsou.html.
Henriksen, G., & Willoch, F. (2008). Imaging of opioid receptors in the central nervous system. Brain, 131(5), 1171–1196.
Horvitz, J. C. (2000). Mesolimbocortical and nigrostriatal dopamine responses to salient non- reward events. Neuroscience, 96(4), 651–656.
Hughes, A., Williams, M. R., Lipari, R. N., Bose, J., Copello, E. A., &Kroutil, L. A. (2015). Prescription Drug Use and Misuse in the United States: Results from the 2015 National Survey on Drug Use and Health. Retrieved from https://www.samhsa.gov/data/sites/default/files/NSDUH-FFR2-2015/NSDUH-FFR2-2015.htm.
Hutchinson, M. R., Lewis, S. S., Coats, B. D., Skyba, D. A., Crysdale, N. Y., Berkelhammer, D. L., Brzeski, A., Northcutt, A., Vietz, C. M., Judd, C. M., et al. (2009). Reduction of opioid withdrawal and potentiation of acute opioid analgesia by systemic AV411. Brain BehavImmun, 23, 240–250.
Ikemoto, A., & Panksepp, J. (1999). The role of nucleus accumbens dopamine in motivated behavior: a unifying interpretation with special reference to reward-seeking. Brain Research Reviews, 31, 6–41.
Inciardi, J. A., Surratt, H. L., Cicero, T. J., Rosenblum, A., Ahwah, C., Bailey, J. E., et al. (2010). Prescription drugs purchased through the internet: who are the end users? Drug and Alcohol Dependence, 110(1–2), 21–29.
Johnson, S. W., & North, R. A. (1992). Opioids excite dopamine neurons by hyperpolarization of local interneurons. The Journal of Neuroscience, 12(2), 483–488.
Johnston, L. D., O’Malley, P. M., Miech, R. A., Bachman, J. G., & Schulenberg, J. E. (2017). Monitoring the Future national survey results on drug use, 1975-2016: overview, key findings on adolescent drug use. Ann Arbor: Institute for Social Research, The University of Michigan.
Jones, C.M. (2013). Trends in the distribution of selected opioids by state, US, 1999–2011. Paper presented at: National Meeting Safe State Alliance, Baltimore, MD.
Jones, J. D., Vosburg, S. K., Manubay, J. M., & Comer, S. D. (2011). Oxycodone abuse in New York City: characteristics of intravenous and intranasal users. The American Journal on Addictions, 20(3), 190–195.
Jones, C. M., Paulozzi, L. J., & Mack, K. A. (2014). Sources of prescription opioid pain relievers by frequency of past-year nonmedical use. JAMA Internal Medicine, 174(5), 802.
Kahan, M., Srivastava, A., Wilson, L., Mailis-Gagnon, A., & Midmer, D. (2006). Opioids for managing chronic non-malignant pain. Canadian Family Physician, 52(9), 1091–1096.
Katz, N., & Fanciullo, G. J. (2002). Role of urine toxicology testing in the management of chronic opioid therapy. The Clinical Journal of Pain, 18, 76–82.
Katz, D. A., & Hays, L. R. (2004). Adolescent oxycontin abuse. Journal of the American Academy of Child & Adolescent Psychiatry, 43(2), 231–234.
Keller, C. E., Ashrafioun, L., Neumann, A. M., Klein, J. V., Fox, C. H., & Blondell, R. D. (2012). Practices, perceptions, and concerns of primary care physicians about opioid dependence associated with the treatment of chronic pain. Substance Abuse, 33(2), 103–113.
Kirkpatrick, S. L., & Bryant, C. D. (2015). Behavioral architecture of opioid reward and aversion in C57BL/6 substrains. Front BehavNeurosci, 8, 450.
Kokki, H., Rasanen, I., Reinikainen, M., Suhonen, P., Vanamo, K., & Ojanperä, I. (2004). Pharmacokinetics of oxycodone after intravenous, Buccal, intramuscular and gastric administration in children. Clinical Pharmacokinetics, 43(9), 613–622.
Kokki, H., Kokki, M., & Sjövall, S. (2012). Oxycodone for the treatment of postoperative pain. Expert OpinPharmacother, 13(7), 1045–1058.
Kuehn, B. M. (2013). SAMHSA: pain medication abuse a common path to heroin. JAMA, 310(14), 1433.
Lalovic, B., Kharasch, E., Hoffer, C., Risler, L., Liu-Chen, L.-Y., & Shen, D. D. (2006). Pharmacokinetics and pharmacodynamics of oral oxycodone in healthy human subjects: role of circulating active metabolites. ClinPharmacol Ther, 79, 461–479.
Lankenau, S. E., Teti, M., Silva, K., Bloom, J. J., Harocopos, A., & Treese, M. (2012). Patterns of prescription drug misuse among young injection drug users. Journal of Urban Health, 89(6), 1004–1016.
Le Merrer, J., Becker, J. A., Befort, K., & Kieffer, B. L. (2009). Reward processing by the opioid system in the brain. Physiological Reviews, 89(4), 1379–1412.
Leow, K. P., Wright, A. W., Cramond, T., & Smith, M. T. (1993). Determination of the serum protein binding of oxycodone and morphine using ultrafiltration. Therapeutic Drug Monitoring, 15(5), 440–447.
Leri, F., & Burns, B. H. (2005). Ultra-low-dose naltrexone reduces the rewarding potency of oxycodone and relapse vulnerability in rats. Pharmacology, Biochemistry, and Behavior, 82, 252–262.
Liu, Y. L., Liang, J. H., Yan, L. D., Su, R. B., Wu, C. F., & Gong, Z. H. (2005). Effects of l-tetrahydropalmatineon locomotor sensitization to oxycodone in mice. Acta Pharmacologica Sinica, 26, 533–538.
Mars, S. G., Bourgois, P., Karandinos, G., Montero, F., & Ciccarone, D. (2014). “Every ‘never’ I ever said came true”: transitions from opioid pills to heroin injecting. International Journal of Drug Policy, 25(2), 257–266.
Mccabe, S. E., Cranford, J. A., Boyd, C. J., & Teter, C. J. (2007). Motives, diversion and routes of administration associated with nonmedical use of prescription opioids. Addictive Behaviors, 32(3), 562–575.
Moradi, M., Esmaeili, S., Shoar, S., & Safari, S. (2012). Use of oxycodone in pain management. Anesthesiology and Pain Medicine, 1(4), 262–264.
Narita, M., Nakamura, A., Ozaki, M., Imai, S., Miyoshi, K., Suzuki, M., & Suzuki, T. (2007). Comparative pharmacological profiles of morphine and oxycodone under a neuropathic pain-like state in mice: evidence for less sensitivity morphine. Neuropsychopharmacology, 33(5), 1097–1112.
National Institute on Drug Abuse. (2016). Monitoring the future. Retrieved from https://www.drugabuse.gov/related-topics/trends-statistics/monitoring-future
Nielsen, C. K., Ross, F. B., & Smith, M. T. (2000). Incomplete, asymmetric, and route-dependent cross-tolerance between oxycodone and morphine in the dark agouti rat. The Journal of Pharmacology and Experimental Therapeutics, 295, 91–99.
Niikura, K., Ho, A., Kreek, M. J., & Zhang, Y. (2013). Oxycodone-induced conditioned place preference and sensitization of locomotor activity in adolescent and adult mice. PharmacolBiochemBehav, 110, 112–116.
Olkkola, K. T., Kontinen, V. K., Saari, T. I., & Kalso, E. A. (2013). Does the pharmacology of oxycodone justify its increasing use as an analgesic? Trends in Pharmacological Sciences, 34(4), 206–214.
Osborne, R., Joel, S., Trew, D., & Slevin, M. (1990). Morphine and metabolite behavior after different routes of morphine administration: demonstration of the importance of the active metabolite morphine-6-glucuronide. ClinPharmacolTher, 47, 12–19.
Osgood, E. D., Eaton, T. A., Trudeau, J. J., & Katz, N. P. (2012). A brief survey to characterize oxycodone abuse patterns in adolescents enrolled in two substance abuse recovery high schools. The American Journal of Drug and Alcohol Abuse, 38(2), 166–170.
Ostlund, S. B., & Balleine, B. W. (2008). On habits and addiction: an associative analysis of compulsive drug seeking. Drug Discov Today Dis Models, 5(4), 235–245.
Patrick, S. W., Schumacher, R. E., Benneyworth, B. D., Krans, E. E., McAllister, J. M., & Davis, M. M. (2012). Neonatal abstinence syndrome and associated health care expenditures: United States, 2000–2009. JAMA, 307(18), 1934–1940.
Peacock, A., Bruno, R., Cama, E., Kihas, I., Larance, B., Lintzeris, N., ... Degenhardt, L. (2015). Jurisdictional differences in opioid use, other licit and illicit drug use, and harms associated with substance use among people who tamper with pharmaceutical opioids. Drug and Alcohol Review, 34(6), 611–622.
Perrino, P. J., Colucci, S. V., Apseloff, G., & Harris, S. C. (2013). Pharmacokinetics, tolerability, and safety of intranasal administration of reformulated OxyContin® tablets compared with original OxyContin® tablets in healthy adults. Clinical Drug Investigation, 33(6), 441–449.
Pöyhiä, R., & Kalso, E. A. (1992). Antinociceptive effects and central nervous system depression caused by oxycodone and morphine in rats. PharmacolToxicol, 70(2), 125–130.
Rigg, K. K., Kurtz, S. P., & Surratt, H. L. (2012). Patterns of prescription medication diversion among drug dealers. Drugs: Education, Prevention and Policy, 19(2), 145–155.
Riley, J., Eisenberg, E., Müller-Schwefe, G., Drewes, A. M., & Arendt-Nielsen, L. (2008). Oxycodone: a review of its use in the management of pain. Current Medical Research and Opinion, 24(1), 175–192.
Rosenblum, A., Parrino, M., Schnoll, S. H., Fong, C., Maxwell, C., Cleland, C. M., et al. (2007). Prescription opioid abuse among enrollees into methadone maintenance treatment. Drug and Alcohol Dependence, 90(1), 64–71.
Ross, F. B., & Smith, M. T. (1997). The instrinsic antinociceptive effects of oxycodone appear to be kappa-opioid receptor mediated. Pain, 73, 151–157.
Ross, F. B., Wallis, S. C., & Smith, M. T. (2000). Co-administration of sub-antinociceptive doses of oxycodone and morphine produces marked antinociceptive synergy with reduced CNS side-effects in rats. Pain, 84(2), 421–428.
Rutten, K., De Vry, J., Robens, A., Tzschentke, T. M., & Van der Kam, E. L. (2011). Dissociation of rewarding, anti-aversive and anti-nociceptive effects of different classes of anti-nociceptives in the rat. European Journal of Pain, 15, 299–305.
Ruttenl, K., Vry, J., Robens, A., Tzschentke, T. M., & Kam, E. L. (2011). Dissociation of rewarding, anti-aversive and anti-nociceptive effects of different classes of anti-nociceptives in the rat. European Journal of Pain, 15(3), 299–305.
Sanchez, V., Carpenter, M. D., Yohn, N. L., & Blendy, J. A. (2016). Long-lasting effects of adolescent oxycodone exposure on reward-related behavior and gene expression in mice. Psychopharmacology, 233(23–24), 3991–4002.
Schultz, W., & Dickinson, A. (2000). Neuronal coding of prediction errors. Annu Rev Neuroscience, 23, 473–500.
Secci, M. E., Factor, J. A., Schindler, C. W., & Panlilio, L. V. (2016). Choice between delayed food and immediate oxycodone in rats. Psychopharmacology, 233(23–24), 3977–3989.
Seip-Cammack, K. M., & Shapiro, M. L. (2014). Behavioral flexibility and response selection are impaired after limited exposure to oxycodone. Learning & Memory, 21(12), 686–695.
Sellers, E. M., Schuller, R., Romach, M. K., & Horbay, G. L. (2006). Relative abuse potential of opioid formulations in Canada: a structured field study. Journal of Opioid Management, 2(4), 219–227.
Silvasti, M., Rosenberg, P., Seppälä, T., Svartling, N., & Pitkänen, M. (1998). Comparison of analgesic efficacy of oxycodone and morphine in postoperative intravenous patient-controlled analgesia. ActaAnaesthesiolScand, 42(5), 576–580.
Smith, H. S. (2009). Opioid metabolism. Mayo Clinic Proceedings, 87(7), 613–624.
Smith, M. Y., & Woody, G. (2005). Nonmedical use and abuse of scheduled medications prescribed for pain, pain-related symptoms, and psychiatric disorders: patterns, user characteristics, and management options. Curr.Psychiatry Rep., 7, 337–343.
Sng, B., Kwok, S., Mathur, D., Ithnin, F., Newton-Dunn, C., Assam, P., et al. (2016). Comparison of epidural oxycodone and epidural morphine for post-caesarean section analgesia: a randomised controlled trial. Indian Journal of Anaesthesia, 60(3), 187.
Sproule, B., Brands, B., Li, S., & Catz-Biro, L. (2009). Changing patterns in opioid addiction: characterizing users of oxycodone and other opioids. Canadian Family Physician, 55(1), 68–69.
Squeglia, L. M., Jacobus, J., & Tapert, S. F. (2009). The influence of substance use on adolescent brain development. Clinical EEG and Neuroscience, 40(1), 31–38.
Steketee, J. D., & Kalivas, P. W. (2011). FDrug wanting: Behavioral sensitization and relapse to drug-seeking behavior. Pharmacological Reviews, 63(2), 348–365.
Stoops, W. W., Hatton, K. W., Lofwall, M. R., Nuzzo, P. A., & Walsh, S. L. (2010). Intravenous oxycodone, hydrocodone, and morphine in recreational opioid users: abuse potential and relative potencies. Psychopharmacology, 212(2), 193–203.
Substance Abuse and Mental Health Services Administration (SAMHSA). (2013). National Survey on Drug use and Health 2011 and 2012. Rockville: Center for Behavioral Health Statistics and Quality.
Substance Abuse and Mental Health Services Administration (SAMHSA). (2017). Prescription Drug Misuse and Abuse. Retrieved from https://www.samhsa.gov/topics/prescription-drug-misuse-abuse
Thompson, C. M., Wojno, H., Greiner, E., May, E. L., Rice, K. C., & Selley, D. E. (2004). Activation of G-proteins by morphine and codeine congeners: insights to the relevance of O- and N-demethylated metabolites at mu- and delta-opioid receptors. The Journal of Pharmacology and Experimental Therapeutics, 308(2), 547–554.
United Nations. (2016). World drug report on international narcotics control board for 2016. Vienna: United Nations.
Upadhyay, J., et al. (2010). Alterations in brain structure and functional connectivity in prescription opioid-dependent patients. Brain, 133, 2098–2114.
U.S. Cong., Senate Caucus on International Narcotics Control. (2014, May 14). National Institute on Drug Abuse. (ND. Volkow, Author) [Cong. Rept.]. Retrieved from https://www.drugabuse.gov/about-nida/legislative-activities/testimony-to-congress/2016/americas-addiction-to-opioids-heroin-prescription-drug-abuse.
Vilà-Balló, A., Mas-Herrero, E., Ripollés, P., Simó, M., Miró, J., Cucurell, D., et al. (2017). Unraveling the role of the hippocampus in reversal learning. The Journal of Neuroscience, 37(28), 6686–6697.
Volkow, N. D., & Mclellan, A. T. (2016). Opioid abuse in chronic pain—misconceptions and mitigation strategies. New England Journal of Medicine, 374(13), 1253–1263.
Vosburg, S. K., Eaton, T. A., Sokolowska, M., Osgood, E. D., Ashworth, J. B., Trudeau, J. J., et al. (2016). Prescription opioid abuse, prescription opioid addiction, and heroin abuse among adolescents in a recovery high school: a pilot study. Journal of Child & Adolescent Substance Abuse, 25(2), 105–112.
Wade, C. L., Vendruscolo, L. F., Scholosburg, J. E., Hernandez, D. O., & Koob, G. F. (2015). Compulsive-like responding for opioid analgesics in rats with extended access. Neuropsychopharmacology, 40(2), 421–428.
Walsh, S. L., Nuzzo, P. A., Lofwall, M. R., & Holtman, J. R. (2008). The relative abuse liability of oral oxycodone, hydrocodone and hydromorphone assessed in prescription opioid abusers. Drug and Alcohol Dependence, 98(3), 191–202.
Webster, L. R., Bath, B., Medve, R. A., Marmon, T., & Stoddard, G. J. (2012). Randomized, double-blind, placebo-controlled study of the abuse potential of different formulations of oral oxycodone. Pain Medicine, 13(6), 790–801.
Weele, C. M., Porter-Stransky, K. A., Mabrouk, O. S., Lovic, V., Singer, B. F., Kennedy, R. T., & Aragona, B. J. (2014). Rapid dopamine transmission within the nucleus accumbens: dramatic difference between morphine and oxycodone delivery. European Journal of Neuroscience, 40(7), 3041–3054.
Wiebelhaus, J. M., Walentiny, D. M., & Beardsley, P. M. (2016). Effects of acute and repeated administration of oxycodone and naloxone-precipitated withdrawal on intracranial self-stimulation in rats. Behavioral Pharmacology, 356(1), 43–52.
Wise, R. A., & Koob, G. F. (2014). The development and maintenance of drug addiction. Neuropsychopharmacology, 39(2), 254–262.
Wisniewski, A. M., Purdy, C. H., & Blondell, R. D. (2008). The epidemiologic association between opioid prescribing, non-medical use, and emergency department visits. Journal of Addictive Diseases, 27(1), 1–11.
Wong, A., Macleod, D., Robinson, J., Koutsogiannis, Z., Graudins, A., & Greene, S. L. (2015). Oxycodone/naloxone preparation can cause acute withdrawal symptoms when misused parenterally or taken orally. ClinToxicol (Phila), 53(8), 815–818.
Wu, L., Pilowsky, D. J., & Patkar, A. A. (2008). Non-prescribed use of pain relievers among adolescents in the United States. Drug and Alcohol Dependence, 94, 1–11.
Yanagidate, F. (2004). Epidural oxycodone or morphine following gynaecologicalsurgery. British Journal of Anaesthesia, 93(3), 362–367.
Zacny, J. P., & Drum, M. (2010). Psychopharmacological effects of oxycodone in healthy volunteers: roles of alcohol-drinking status and sex. Drug and Alcohol Dependence, 107, 209–214.
Zhang, Y., Picetti, R., Butelman, E. R., Schlussman, S. D., Ho, A., & Kreek, M. J. (2009). Behavioral and neurochemical changes induced by oxycodone differ between adolescent and adult mice. Neuropsychopharmacology, 34, 912–922.
Zhang, Y., Brownstein, A., Buonora, M., Niikura, K., Ho, A., Rosa, J. C., et al. (2015). Self-administration of oxycodone alters synaptic plasticity gene expression in the hippocampus differentially in male adolescent and adult mice. Neuroscience, 285, 34–46.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Meenu Minhas and Dr. Francesco Leri declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Minhas, M., Leri, F. A Multifaceted Analysis of Oxycodone Addiction. Int J Ment Health Addiction 16, 1016–1032 (2018). https://doi.org/10.1007/s11469-017-9827-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11469-017-9827-y