Abstract
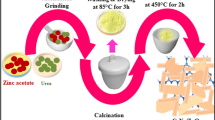
Ag-loaded TiO2 (Ag/TiO2) nanocomposites were prepared by microwave-assisted chemical reduction method using tetrabutyl titanate as the Ti source. The prepared samples were characterized by X-ray diffraction, scanning electron microscopy, transmission electron microscopy, N2 adsorption–desorption isotherms, UV–vis absorption spectrum, X-ray photoelectron spectrum, photoluminescence spectrum, and Raman scattering spectrum, respectively. Results revealed that Ag nanoparticles (NPs) were successfully deposited on TiO2 by reduction of Ag+, and the visible light absorption and Raman scattering of TiO2 were enhanced by Ag NPs based on its surface plasmon resonance effect. Besides, Ag NPs could also effectively restrain the recombination of photogenerated electrons and holes with a longer luminescence life time. In addition, photocatalytic reduction of CO2 with H2O on the composites was conducted to obtain methanol. Experimental results indicated that Ag-loaded TiO2 had better photocatalytic activity than pure TiO2 due to the synergistic effect between UV light excitation and surface plasmon resonance enhancement, and 2.5 % Ag/TiO2 exhibited the best activity; the corresponding energy efficiency was about 0.5 % and methanol yield was 405.2 μmol/g-cat, which was 9.4 times higher than that of pure TiO2. Additionally, an excitation enhancement synergistic mechanism was proposed to explain the experimental results of photocatalytic reduction of CO2 under different reaction conditions.













Similar content being viewed by others
References
Fujishima A, Honda K (1972) Electrochemical photolysis of water at a semiconductor electrode. Nature 238:37–38. doi:10.1038/238037a0
Hoffmann MR, Martin ST, Choi W, Bahnemann DW (1995) Environmental applications of semiconductor photocatalysis. Chem Rev 95:69–96. doi:10.1021/cr00033a004
Khan SU, Al-Shahry M, Ingler WB (2002) Efficient photochemical water splitting by a chemically modified n-TiO2. Science 297:2243–2245. doi:10.1126/science.1075035
Wu X, Chen Z, Lu GQM, Wang L (2011) Nanosized anatase TiO2 single crystals with tunable exposed (001) facets for enhanced energy conversion efficiency of dye-sensitized solar cells. Adv Funct Mater 21:4167–4172. doi:10.1002/adfm.201100828
Yaron P (2010) Application of TiO2 photocatalysis for air treatment: patents' overview. Appl Catal B: Environ 99:448–460. doi:10.1016/j.apcatb.2010.05.011
Xiang Q, Yu J, Jaroniec M (2012) Synergetic effect of MoS2 and graphene as cocatalysts for enhanced photocatalytic H2 production activity of TiO2 nanoparticles. J Am Chem Soc 134:6575–6578. doi:10.1021/ja302846n
O'Regan B, Xiaoe L, Ghaddar T (2012) Dye adsorption, desorption, and distribution in mesoporous TiO2 films and its effects on recombination losses in dye sensitized solar cells. Energy Environ Sci 5:7203–7215. doi:10.1039/C2EE21341A
Wang M, Bai J, Le Formal F, Moon SJ, Cevey-Ha L, Humphry-Baker R, Grätzel C, Zakeeruddin SM, Grätzel M (2012) Solid-state dye-sensitized solar cells using ordered TiO2 nanorods on transparent conductive oxide as photoanodes. J Phys Chem C 116:3266–3273. doi:10.1021/jp209130x
Sun T, Fan J, Liu E, Liu L, Wang Y, Dai H, Yang Y, Hou W, Hu X, Jiang Z (2012) Fe and Ni co-doped TiO2 nanoparticles prepared by alcohol-thermal method: application in hydrogen evolution by water splitting under visible light irradiation. Powder Technol 228:210–218. doi:10.1016/j.powtec.2012.05.018
Fan J, Liu E, Tian L, Hu X, He Q, Sun T (2010) Synergistic effect of N and Ni2+ on nanotitania in photocatalytic reduction of CO2. J Environ Eng 137:171–176. doi:10.1061/(ASCE)EE.1943-7870.0000311
Li Y, Chen L, Guo Y, Sun X, Wei Y (2012) Preparation and characterization of WO3/TiO2 hollow microsphere composites with catalytic activity in dark. Chem Eng J 181–182:734–739. doi:10.1016/j.cej.2010.12.007
Kalanur SS, Lee SH, Hwang YJ, Joo OS (2013) Enhanced photoanode properties of CdS nanoparticle sensitized TiO2 nanotube arrays by solvothermal synthesis. J Photochem Photobiol A 259:1–9. doi:10.1016/j.jphotochem.2013.02.018
Bian Z, Tachikawa T, Kim W, Choi W, Majima T (2012) Superior electron transport and photocatalytic abilities of metal-nanoparticle-loaded TiO2 superstructures. J Phys Chem C 116:25444–25453. doi:10.1021/jp309683f
Yanagida S, Makino M, Ogaki T, Yasumori A (2012) Preparation of Pd-Pt co-loaded TiO2 thin films by sol–gel method for hydrogen gas sensing. J Electrochem Soc 159:845–849. doi:10.1149/2.059212jes
Li X, Zhuang Z, Li W, Pan H (2012) Photocatalytic reduction of CO2 over noble metal-loaded and nitrogen-doped mesoporous TiO2. Appl Catal A 429–430:31–38. doi:10.1016/j.apcata.2012.04.001
Sun L, Li J, Wang C, Li S, Lai Y, Chen H, Lin C (2009) Ultrasound aided photochemical synthesis of Ag loaded TiO2 nanotube arrays to enhance photocatalytic activity. J Hazard Mater 171:1045–1050. doi:10.1016/j.jhazmat.2009.06.115
Houšková V, Stengl V, Bakardjieva S, Murafa N, Tyrpekl V (2009) Efficient gas phase photodecomposition of acetone by Ru-doped Titania. Appl Catal, B 89:613–619. doi:10.1016/j.apcatb.2009.01.023
Wu TS, Wang KX, Li GD, Sun SY, Sun J, Chen JS (2010) Montmorillonite-supported Ag/TiO2 nanoparticles: an efficient visible-light bacteria photodegradation material. ACS Appl Mater Interfaces 2:544–550. doi:10.1021/am900743d
Christopher P, Ingram DB, Linic S (2010) Enhancing photochemical activity of semiconductor nanoparticles with optically active Ag nanostructures: photochemistry mediated by Ag surface plasmons. J Phys Chem C 114:9173–9177. doi:10.1021/jp101633u
Logar M, Jančar B, Šturm S, Suvorov D (2010) Weak polyion multilayer-assisted in situ synthesis as a route toward a plasmonic Ag/TiO2 photocatalyst. Langmuir 26:12215–12224. doi:10.1021/la101124q
Awazu K, Fujimaki M, Rockstuhl C, Tominaga J, Murakami H, Ohki Y, Yoshida N, Watanabe T (2008) A plasmonic photocatalyst consisting of silver nanoparticles embedded in titanium dioxide. J Am Chem Soc 130:1676–1680. doi:10.1021/ja076503n
Chen JJ, Wu JC, Wu PC, Tsai DP (2010) Plasmonic photocatalyst for H2 evolution in photocatalytic water splitting. J Phys Chem C 115:210–216. doi:10.1021/jp1074048
Wu F, Hu X, Fan J, Liu E, Sun T, Kang L, Hou W, Zhu C, Liu H (2012) Photocatalytic activity of Ag/TiO2 nanotube arrays enhanced by surface plasmon resonance and application in hydrogen evolution by water splitting. Plasmonics 1–8:502–508. doi:10.1007/s11468-012-9418-5
Zhou M, Zhang J, Cheng B, Yu H (2012) Enhancement of visible-light photocatalytic activity of mesoporous Au-TiO2 nanocomposites by surface plasmon resonance. Int J Photoenergy. doi:10.1155/2012/532843
Kochuveedu ST, Kim DP, Kim DH (2012) Surface-plasmon-induced visible light photocatalytic activity of TiO2 nanospheres decorated by Au nanoparticles with controlled configuration. J Phys Chem C 116:2500–2506. doi:10.1021/jp209520m
Yu J, Dai G, Huang B (2009) Fabrication and characterization of visible-light-driven plasmonic photocatalyst Ag/AgCl/TiO2 nanotube arrays. J Phys Chem C 113:16394–16401. doi:10.1021/jp905247j
Liang YQ, Cui ZD, Zhu SL, Liu Y, Yang XJ (2011) Silver nanoparticles supported on TiO2 nanotubes as active catalysts for ethanol oxidation. J Catal 278:276–287. doi:10.1016/j.jcat.2010.12.011
Zielińska A, Kowalska E, Sobczak JW, Łącka I, Gazda M, Ohtani B, Hupka J, Zaleska A (2010) Silver-doped TiO2 prepared by microemulsion method: surface properties, bio- and photoactivity. Sep Purif Technol 72:309–318. doi:10.1016/j.seppur.2010.03.002
Amendola V, Bakr OM, Stellacci F (2010) A study of the surface plasmon resonance of silver nanoparticles by the discrete dipole approximation method: effect of shape, size, structure, and assembly. Plasmonics 5:85–97. doi:10.1007/s11468-009-9120-4
Ma YW, Wu ZW, Zhang LH, Zhang J, Jian GS, Pan S (2013).Theoretical study of the local surface plasmon resonance properties of silver nanosphere clusters. Plasmonics 1–10. doi:10.1007/s11468-013-9541-y
Zhang YX, Li GH, Jin YX, Zhang Y, Zhang J, Zhang LD (2002) Hydrothermal synthesis and photoluminescence of TiO2 nanowires. Chem Phys Lett 365:300–304. doi:10.1016/S0009-2614(02)01499-9
Yu J, Yu H, Ao CH, Lee SC, Yu JC, Ho W (2006) Preparation, characterization and photocatalytic activity of in situ Fe-doped TiO2 thin films. Thin Solid Films 49:273–280. doi:10.1016/j.tsf.2005.08.352
Kremenović A, Antić B, Blanuša J, Čomor M, Colomban P, Mazerolles L, Bozin ES (2011) Heterogeneity and disorder in Ti1-xFeyO2-d nanocrystal rutile-based flowerlike aggregates: detection of anatase. J Phys Chem C 115:4395–4403. doi:10.1021/jp109437b
Swamy V, Kuznetsov A, Dubrovinsky LS, Caruso RA, Shchukin DG, Muddle BC (2005) Finite-size and pressure effects on the Raman spectrum of nanocrystalline anatase TiO2. Phys Rev B 71:184302–184312. doi:10.1103/PhysRevB.71.184302
Wu JC (2009) Photocatalytic reduction of greenhouse gas CO2 to fuel. Catal Surv Asia 13:30–40. doi:10.1007/s10563-009-9065-9
Anpo M, Yamashita H, Ichihashi Y, Ehara S (1995) Photocatalytic reduction of CO2 with H2O on various titanium oxide catalysts. J Electroanal Chem 396:21–26. doi:10.1016/0022-0728(95)04141-A
Tan SS, Zou L, Hu E (2006) Photocatalytic reduction of carbon dioxide into gaseous hydrocarbon using TiO2 pellets. Catal Today 115:269–273. doi:10.1016/j.cattod.2006.02.057
Zhang QH, Han WD, Hong YJ, Yu JG (2009) Photocatalytic reduction of CO2 with H2O on Pt-loaded TiO2 catalyst. Catal Today 148:335–340. doi:10.1016/j.cattod.2009.07.081
Acknowledgments
This work was supported by the National natural Science Foundation of China (no. 21176199), the Scientific Research Foundation of Northwest University (no. 12NW19), the Scientific Research Staring Foundation of Northwest University (no. PR12216), the Shaanxi Provincial Research Foundation for Basic Research, China (grant no. 2013JQ2003), and the Scientific Research Foundation of the Education Department of Shaanxi Provincial Government, China (no. 2013JK0693).
Author information
Authors and Affiliations
Corresponding author
Additional information
Enzhou Liu and Limin Kang are the co-first authors.
Rights and permissions
About this article
Cite this article
Liu, E., Kang, L., Wu, F. et al. Photocatalytic Reduction of CO2 into Methanol over Ag/TiO2 Nanocomposites Enhanced by Surface Plasmon Resonance. Plasmonics 9, 61–70 (2014). https://doi.org/10.1007/s11468-013-9598-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11468-013-9598-7