Abstract
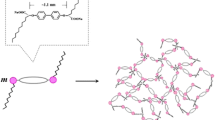
The critical micelle concentration (CMC) of cetyl trimethylammonium bromide (CTAB) in both water and ethanol-water-mixed solvent was determined using steady-state fluorescence techniques in order to investigate the effect of the self-assembling properties of the surfactant on the template synthesis of porous inorganic materials. Results indicated that the CMC increased with the increase of ethanol concentration in the mixed solvent. The CMC of CTAB is 0.0009 mol/L in water, while it is 0.24 mol/L in ethanol. Furthermore, the dissipative particle dynamics (DPD) was adopted to simulate the aggregation of CTAB in water and ethanol/water mixtures, and the energy difference was calculated for the surfactant tail groups after mixing with the solvent. The simulation results reflected a regularity similar to the experimental data, i.e., tail groups of CTAB interacted more strongly with ethanol than with groups of CTAB interacted more strongly with ethanol than with water, which elucidates the reason that the micelle is difficult to form in ethanol.
Similar content being viewed by others
References
Brown, S.P., Schnell I., Brand J.D., Müllen K., Spiess H.W., An Investigation of π-π Packing in a Columnar Hexabenzocoronene by Fast Magic-Angle Spinning and Double-Quantum 1H Solid-State NMR Spectroscopy. J. Am. Chem. Soc., 1999, 121(28): 6712–6718
McMahon C.A., Hawrylak B., Palepu R., Calorimetric and NMR Investigations of the Micellar Properties of Sodium Dodecyl Sulfate in Aqueous Mixtures of Isomeric Butanediols. Langmuir, 1999, 15(2): 429–436
Markey L., Stievenard D., Devos A., Lannoo M.; Demol F.; de Backer M., STM observations of self-assembled 1D and 2D nanoclusters of aromatic cryptand molecules deposited on highly oriented pyrolytic graphite. Supramole Science, 1997, 4(3–4): 375–379.
Huang Yaoxiong, Tan Runchu, Li Yonglong, Yang Yuquan, He Oicai, Effect of Salts on the Formation of C8-Lecithin Micelles in Aqueous Solution. J. Colloid and Interface Sci., 2001, 236(1): 28–34
Miguel M. G., Burrows H. D., Formosinho S. J., Lindman, B., Fluorescence studies of polymer-surfactant association. J. Mol. Struct., 2001, 563(1): 89–98
Lam Y. M., Goldbeck-Wood G., Mesoscale simulation of block copolymers in aqueous solution: parameterisation, micelle growth kinetics and the effect of temperature and concentration morphology. Polymer, 2003, 44(12): 3593–3605
Li Youyong., Guo Senli., Wang Kaixuan., Xu Xiaojie., Mesoscale computer simulation and its applications, Progress in Chemistry, 2000, 12(4) 361–375 (in Chinese)
Hansson P., Schneider S., Lindman B., Phase separation in polyelectrolyte gels interacting with surfactants of opposite charge. J. Phys. Chem. B., 2002, 106(38): 9777–9793
Yuan Shiling, Cai Zhengting, Xu Guiying, Mesoscopic simulation of aggregates in surfactant/oil/water systems. Chinese Journal of Chemistry, 2003, 21(2): 112–116 (in Chinese)
Yuan Shiling., Cai Zhengting., Xu Guiying., Dynamic simulation of aggregation morphology in surfactant solutions. Acta Chimica Sinica(in Chinese), 2002, 60(4), 241–245
Claesson P.M., Fielden M.L., Interactions between a 30 Charged Polyelec-trolyte and an Anionic Surfactant in Bulk and at a Solid-Liquid Interface. J. Phys. Chem. B., 1998, 102(7): 1270–1278
Groot R.D., Mesoscopic Simulation of Polymer-Surfactant Aggregation. Langmuir, 2000, 16(19): 7493–7502
Asefa T., Maclachian M. J., Coombs N., Ozin, G. A., Periodic mesoporous organosilicas with organic groups inside the channel walls. Nature, 1999, 402(6764): 867–881
Zhang Jinli, Li Wei, Meng, Xiang-kun, Wang, Li, Zhu Li. Synthesis of mesoporous silica membranes oriented by self-assembles of surfactants. Journal of Membrane Science, 2003, 222(1–2): 219–224
Author information
Authors and Affiliations
Corresponding author
Additional information
__________
Translated from Journal of Tianjin University, 2006, 39(1) (in Chinese)
About this article
Cite this article
Li, W., Zhang, M., Zhang, J. et al. Self-assembly of cetyl trimethylammonium bromide in ethanol-water mixtures. Front. Chem. China 1, 438–442 (2006). https://doi.org/10.1007/s11458-006-0069-y
Issue Date:
DOI: https://doi.org/10.1007/s11458-006-0069-y