Abstract
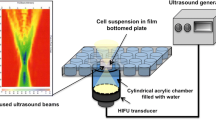
The objective of the present study was to investigate whether ultrasound combined with microbubbles induces apoptotic cell death in androgen-independent prostate cancer cells and to identify the probable mechanism. We used ultrasound in continuous wave mode with a frequency of 21 kHz and a spatial-average temporal-average intensity of 46 mW/cm2. Ultrasound combined with microbubbles (200 μL SonoVue™) was used to treat androgen-independent human prostate cancer PC-3 cells for 30 s. PC-3 cells were divided into three groups: the control group, the ultrasound group and the ultrasound combined with microbubbles group. Immediately after treatment, trypan blue exclusion was used to assess cell viability. Cell apoptosis at 24 h after treatment was measured using transmission electron microscopy and flow cytometry. Western blotting was used to evaluate the expression of the apoptosis-related proteins, Bcl-2 and Bax. Ultrasound combined with microbubbles had a minimal effect on the viability of PC-3 cells and induced minimal levels of cell lysis. The level of apoptosis in PC-3 cells induced by this modality was significantly higher than in controls (12.77 ± 0.31% vs. 2.56 ± 0.22%, P<0.01). Treatment with ultrasound combined with microbubbles increased the expression of Bax, a pro-apoptotic protein, and decreased the expression of Bcl-2, an anti-apoptotic protein. It was concluded that ultrasound combined with microbubbles induces apoptotic cell death in human prostate cancer PC-3 cells through down-regulation of Bcl-2 and up-regulation of Bax.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Jemal A, Siegel R, Xu J, et al. Cancer statistics, 2010. CA Cancer J Clin, 2010, 60: 277–300
Xu A H, Hu Z M, Qu J B, et al. Cyclic bisbibenzyls induce growth arrest and apoptosis of human prostate cancer PC3 cells. Acta Pharmacol Sin, 2010, 31: 609–615
Mangar S A, Huddart R A, Parker C C, et al. Technological advances in radiotherapy for the treatment of localised prostate cancer. Eur J Cancer, 2005, 41: 908–921
Tanaka G, Hirata Y, Goldenberg S L, et al. Mathematical modelling of prostate cancer growth and its application to hormone therapy. Philos Transact A Math Phys Eng Sci, 2010, 368: 5029–5044
Lecornet E, Ahmed H U, Moore C, et al. Focal therapy for prostate cancer: A potential strategy to address the problem of overtreatment. Arch Esp Urol, 2010, 63: 845–852
Baumert H. Salvage treatments for prostatic radiation failure. Cancer Radiothe, 2010, 14: 442–445
Tabuchi Y, Takasaki I, Zhao Q L, et al. Genetic networks responsive to low-intensity pulsed ultrasound in human lymphoma U937 cells. Cancer Lett, 2008, 270: 286–294
Tang W, Liu Q, Zhang J, et al. In vitro activation of mitochondria-caspase signaling pathway in sonodynamic therapy-induced apoptosis in sarcoma 180 cells. Ultrasonics, 2010, 50: 567–576
Feng Y, Tian Z, Wan M. Bioeffects of low-intensity ultrasound in vitro: Apoptosis, protein profile alteration, and potential molecular mechanism. J Ultrasound Med, 2010, 29: 963–974
Ashush H, Rozenszajn L A, Blass M, et al. Apoptosis induction of human myeloid leukemic cells by ultrasound exposure. Cancer Res, 2000, 60: 1014–1020
Rosenthal I, Sostaric J Z, Riesz P. Sonodynamic therapy—A review of the synergistic effects of drugs and ultrasound. Ultrason Sonochem, 2004, 11: 349–363
Tian Z, Quan X, Xu C, et al. Hematoporphyrin monomethyl ether enhances the killing action of ultrasound on osteosarcoma in vivo. J Ultrasound Med, 2009, 28: 1695–1702
Kolarova H, Tomankova K, Bajgar R, et al. Photodynamic and sonodynamic treatment by phthalocyanine on cancer cell lines. Ultrasound Med Biol, 2009, 35: 1397–1404
Jackson J K, Pirmoradi F N, Wan C P, et al. Increased accumulation of paclitaxel and doxorubicin in proliferating capillary cells and prostate cancer cells following ultrasound exposure. Ultrasonics, 2011, 51: 932–939
Kawai N, Iino M. Molecular damage to membrane proteins induced by ultrasound. Ultrasound Med Biol, 2003, 29: 609–614
Marentis T C, Kusler B, Yaralioglu G G, et al. Microfluidic sonicator for real-time disruption of eukaryotic cells and bacterial spores for DNA analysis. Ultrasound Med Biol, 2005, 31: 1265–1277
Reed J C. Bcl-2 and the regulation of programmed cell death. J Cell Biol, 1994, 124: 1–6
Yang E, Korsmeyer S. Molecular thanatopsis: Adiscourse on the BCL2 family and cell death. Blood, 1996, 88: 386–401
Apakama I, Robinson M C, Walter N M, et al. Bcl-2 overexpression combined with p53 protein accumulation correlates with hormonerefractory prostate cancer. Br J Cancer, 1996, 74: 1258–1262
Hassan M A, Feril L B Jr, Suzuki K, et al. Evaluation and comparison of three novel microbubbles: Enhancement of ultrasound-induced cell death and free radicals production. Ultrason Sonochem, 2009, 16: 372–378
Lagneaux L, de Meulenaer E C, Delforge A, et al. Ultrasonic lowenergy treatment: A novel approach to induce apoptosis in human leukemic cells. Exp Hematol, 2002, 30: 1293–1301
Furusawa Y, Zhao Q L, Hassan M A, et al. Ultrasound-induced apoptosis in the presence of Sonazoid and associated alterations in gene expression levels: A possible therapeutic application. Cancer Lett, 2010, 288: 107–115
Sicklick J K, Li Y X, Jayaraman A, et al. Dysregulation of the Hedgehog pathway in human hepatocarcinogenesis. Carcinogenesis, 2006, 27: 748–757
Wang X B, Liu Q H, Wang P, et al. Enhancement of apoptosis by sonodynamic therapy with protoporphyrin IX in isolate sarcoma 180 cells. Cancer Biother Radiopharm, 2008, 23: 238–246
Fang H Y, Tsai K C, Cheng W H, et al. The effects of power on-off durations of pulsed ultrasound on the destruction of cancer cells. Int J Hyperthermia, 2007, 23: 371–380
Li Y C, Lin H J, Yang J H, et al. Baicalein-induced apoptosis via endoplasmic reticulum stress through elevations of reactive oxygen species and mitochondria dependent pathway in mouserat hybrid retina ganglion cells (N18). Neurochem Res, 2008, 34: 418–429
Samuel S, Miller D L, Fowlkes J B. The relationship of acoustic emission and pulse-repetition frequency in the detection of gas body stability and cell death. Ultrasound Med Biol, 2006, 32: 439–447
Chumakova O V, Liopo A V, Evers B M, et al. Effect of 5-fluorouracil, optison and ultrasound on MCF-7 cell viability. Ultrasound Med Biol, 2006, 32: 751–758
Kodama T, Tomita Y, Koshiyama K, et al. Transfection effect of microbubbles on cells in superposed ultrasound waves and behavior of cavitation bubble. Ultrasound Med Biol, 2006, 32: 905–914
Tsujimoto Y, Croce C M. Analysis of the structure, transcripts, and protein products of bcl-2, the gene involved in human follicular lymphoma. Proc Natl Acad Sci USA, 1986, 83: 5214–5218
Ku J H, Seo S Y, Kwak C, et al. The role of survivin and Bcl-2 in zinc-induced apoptosis in prostate cancer cells. Urol Oncol, 2013, in press
Malik A, Afaq S, Shahid M, et al. Influence of ellagic acid on prostate cancer cell proliferation: A caspase-dependent pathway. Asian Pac J Trop Med, 2011, 4: 550–555
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Bai, W., Yang, S., Shen, E. et al. Treatment of PC-3 cells with ultrasound combined with microbubbles induces distinct alterations in the expression of Bcl-2 and Bax. Chin. Sci. Bull. 58, 3535–3540 (2013). https://doi.org/10.1007/s11434-013-5753-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-013-5753-y