Abstract
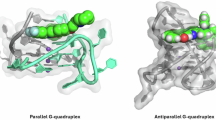
G-quadruplexes attract more and more attention in recent years. Numerous small molecules which can induce or stabilize the formation of G-quadruplexes have been investigated on the purpose of anticancer drug development. As a motif existed in physiological condition, flanking sequences are an important part of G-quadruplexes but the study on the impact of flanking sequences on (G-quadruplex)-ligand binding is rarely reported. In this paper, the effects of flanking sequences on binding affinity between a series of unimolecular parallel-stranded G-quadruplex sequences derived from c-myc oncogene promoter (termed as c-myc G-quadruplexes) and their ligands are discussed in detail. The results showed that the flanking sequences on c-myc G-quadruplexes play key roles in (G-quadruplex)-ligand interaction. When a c-myc G-quadruplex is bound to its ligands, the flanking sequences might form a binding cavity above the terminal G-quartet, which could provide a suitable site for ligands to dock in. Moreover, the bases on flanking sequences could interact with ligand through π-π stacking, and finally form a sandwich-stacking mode (terminal G-quartet, ligand and bases on the flanking sequence). This mode could stabilize the (G-quadruplex)-ligand complex effectively and enhance the binding affinity dramatically. However, flanking sequences are also found to exhibit steric hindrance effect which could impede the (G-quadruplex)-ligand binding.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Hurley L H, Siddiqui-Jain A, Grand C L, et al. Direct evidence for a G-quadruplex in a promoter region and its targeting with a small molecule to repress c-myc transcription. Proc Natl Acad Sci USA, 2002, 99: 11593–11598
Sun D Y, Thompson B, Cathers B E, et al. Inhibition of human telomerase by a G-quadruplex-interactive compound. J Med Chem, 1997, 40: 2113–2116
Neidle S, Burge S, Parkinson G N, et al. Quadruplex DNA: Sequence, topology and structure. Nucleic Acids Res, 2006, 34: 5402–5415
Xiao J H, Davidson I, Macchi M, et al. In vitro binding of several cell-specific and ubiquitous nuclear proteins to the Gt-I motif of the sv40 enhancer. Gene Dev, 1987, 1: 794–807
Kuryavyi V V, Jovin T M. Triad-DNA-A model for trinucleotide repeats. Nat Genet, 1995, 9: 339–341
Huppert J L, Balasubramanian S. G-quadruplexes in promoters throughout the human genome. Nucleic Acid Res, 2007, 35: 406–413
Wang Y, Patel D J. Guanine residues in d(T2AG3) and d(T2G4) form parallel-stranded potassium cation stabilized G-quadruplexes with antiglycosidic torsion angles in solution. Biochemistry, 1992, 31: 8112–8119
Izbicka E, Nishioka D, Marcell V, et al. Telomere-interactive agents affect proliferation rates and induce chromosomal destabilization in sea urchin embryos. Anti Canc Drug Des, 1999, 14: 355–365
Williamson J R. G-quartet structures in telomeric DNA. Annu Rev Biophys Biomol Struct, 1994, 23: 703–730
Parkinson G N, Ghosh R, Neidle S. Structural basis for binding of porphyrin to human telomeres. Biochemistry, 2007, 46: 2390–2397
Han F X G, Wheelhouse R T, Hurley L H. Interactions of TMPyP4 and TMPyP2 with quadruplex DNA. Structural basis for the differential effects on telomerase inhibition. J Am Chem Soc, 1999, 121: 3561–3570
Seenisamy J, Bashyam S, Gokhale V, et al. Design and synthesis of an expanded porphyrin that has selectivity for the c-myc G-quadruplex structure. J Am Chem Soc, 2005, 127: 2944–2959
Dash J, Waller Z A E, Pantos G D, et al. Synthesis and binding studies of novel diethynyl-pyridine amides with genomic promoter DNA G-quadruplexes. Chem-Eur J, 2011, 17: 4571–4581
Mcguire R, Mcmillin D R. Steric effects direct the binding of porphyrins to tetramolecular quadruplex DNA. Chem Commun, 2009, 7393–7395
Xu W, Tan J H, Chen S B, et al. Studies on the binding of 5-N-methylated quindoline derivative to human telomeric G-quadruplex. Biochem Biophys Res Commun, 2011, 406: 454–458
Pilch D S, Barbieri C M, Rzuczek S G, et al. Targeting human telomeric G-quadruplex DNA with oxazole-containing macrocyclic compounds. Biochimie, 2008, 90: 1233–1249
Rahman K M, Reszka A P, Gunaratnam M, et al. Biaryl polyamides as a new class of DNA quadruplex-binding ligands. Chem Commun, 2009, 4097–4099
Sparapani S, Bellini S, Gunaratnam M, et al. Bis-guanylhydrazone diimidazo 1,2-a:1,2-c pyrimidine as a novel and specific G-quadruplex binding motif. Chem Commun, 2010, 46: 5680–5682
Dash J, Shirude P S, Hsu S T D, et al. Diarylethynyl amides that recognize the parallel conformation of genomic promoter DNA G-quadruplexes. J Am Chem Soc, 2008, 130: 15950–15956
Martino L, Virno A, Pagano B, et al. Structural and thermodynamic studies of the interaction of distamycin A with the parallel quadruplex structure [d(TGGGGT)]4. J Am Chem Soc, 2007, 129: 16048–16056
Sun H X, Tang Y L, Xiang J F, et al. Spectroscopic studies of the interaction between quercetin and G-quadruplex DNA. Bioorg Med Chem Lett, 2006, 16: 3586–3589
Baraldi P G, Bovero A, Fruttarolo F, et al. DNA minor groove binders as potential antitumor and antimicrobial agents. Med Res Rev, 2004, 24: 475–528
Nelson S M, Ferguson L R, Denny W A. Non-covalent ligand/DNA interactions: Minor groove binding agents. Mutat Res, 2007, 623: 24–40
Li Q, Xiang J F, Li X D, et al. Stabilizing parallel G-quadruplex DNA by a new class of ligands: Two non-planar alkaloids through interaction in lateral grooves. Biochimie, 2009, 91: 811–819
Guedin A, Gros J, Alberti P, et al. How long is too long? Effects of loop size on G-quadruplex stability. Nucleic Acids Res, 2010, 38: 7858–7868
Arora A, Maiti S. Stability and molecular recognition of quadruplexes with different loop length in the absence and presence of molecular crowding agents. J Phys Chem B, 2009, 113: 8784–8792
Kelly K, Cochran B H, Stiles C D, et al. Cell-specific regulation of the c-myc-gene by lymphocyte mitogens and platelet-derived growth-factor. Cell, 1983, 35: 603–610
Evan G I, Wyllie A H, Gilbert C S, et al. Induction of apoptosis in fibroblasts by c-myc protein. Cell, 1992, 69: 119–128
Kinzler K W, He T C, Sparks A B, et al. Identification of c-myc as a target of the APC pathway. Science, 1998, 281: 1509–1512
Steller H. Mechanisms and genes of cellular suicide. Science, 1995, 267: 1445–1449
Cosconati S, Marinelli L, Trotta R, et al. Tandem application of virtual screening and NMR experiments in the discovery of brand new DNA quadruplex groove binders. J Am Chem Soc, 2009, 131: 16336
Pelengaris S, Khan M. The c-myc oncoprotein as a treatment target in cancer and other disorders of cell growth. Expert Opin Ther Target, 2003, 7: 623–642
Gartel A L, Shchors K. Mechanisms of c-myc-mediated transcriptional repression of growth arrest genes. Exp Cell Res, 2003, 283: 17–21
Slamon D J, Dekernion J B, Verma I M, et al. Expression of cellular oncogenes in human malignancies. Science, 1984, 224: 256–262
Siebenlist U, Hennighausen L, Battey J, et al. Chromatin structure and protein binding in the putative regulatory region of the c-myc gene in Burkitt lymphoma. Cell, 1984, 37: 381–391
Davis T L, Firulli A B, Kinniburgh A J. Ribonucleoprotein and protein factors bind to an H-DNA-forming c-myc DNA element: Possible regulators of the c-myc gene. Proc Natl Acad Sci USA, 1989, 86: 9682–9686
Simonsson T, Pecinka P, Kubista M. DNA tetraplex formation in the control region of c-myc. Nucleic Acids Res, 1998, 26: 1167–1172
Siddiqui-Jain A, Grand C L, Bearss D J, et al. Direct evidence for a G-quadruplex in a promoter region and its targeting with a small molecule to repress c-myc transcription. Proc Natl Acad Sci USA, 2002, 99: 11593–11598
Patel D J, Phan A T, Kuryavyi V. Human telomere, oncogenic promoter and 5′-UTR G-quadruplexes: Diverse higher order DNA and RNA targets for cancer therapeutics. Nucleic Acids Res, 2007, 35: 7429–7455
Phan A T, Modi Y S, Patel D J. Propeller-type parallel-stranded G-quadruplexes in the human c-myc promoter. J Am Chem Soc, 2004, 126: 8710–8716
James T H, Mees C E K. The Theory of the Photographic Process. 4th ed. New York: Macmillan, 1977
Salvioli S, Ardizzoni A, Franceschi C, et al. JC-1, but not DiOC6(3) or rhodamine 123, is a reliable fluorescent probe to assess delta psi changes in intact cells: Implications for studies on mitochondrial functionality during apoptosis. FEBS Lett, 1997, 411: 77–82
Blatchford J W, Gustafson T L, Epstein A J, et al. Spatially and temporally resolved emission from aggregates in conjugated polymers. Phys Rev B Conden Matt, 1996, 54: R3683–R3686
Li N, Yu C, Huang F. Novel cyanine-AMP conjugates for efficient 5′ RNA fluorescent labeling by one-step transcription and replacement of [gamma-32P]ATP in RNA structural investigation. Nucleic Acid Res, 2005, 33: e37
Davila J, Harriman A, Gulliya K S. Photochemistry of merocyanine 540: The mechanism of chemotherapeutic activity with cyanine dyes. Photochem Photobiol, 1991, 53: 1–11
Yang Q, Xiang J F, Yang S, et al. Verification of intramolecular hybrid/parallel G-quadruplex structure under physiological conditions using novel cyanine dye H-aggregates: Both in solution and on Au film. Anal Chem, 2010, 82: 9135–9137
Sun H X, Xiang J F, Zhang Y Z, et al. Spectroscopic studies of the interaction between methylene blue and G-quadruplex. Chin Sci Bull, 2006, 51: 1687–1692
Hamer F M. The Cyanine Dyes and Related Compounds. New York: Interscience Publishers, 1964
Ficken G E. The Chemistry of Synthetic Dyes. New York: Academic Press, 1971
Mergny J L, Phan A T, Lacroix L. Following G-quartet formation by UV-spectroscopy. FEBS Lett, 1998, 435: 74–78
Balagurumoorthy P, Brahmachari S K, Mohanty D, et al. Hairpin and parallel quartet structures for telomeric sequences. Nucleic Acid Res, 1992, 20: 4061–4067
Yang Q F, Xiang J F, Yang S, et al. Verification of specific G-quadruplex structure by using a novel cyanine dye supramolecular assembly: II. The binding characterization with specific intramolecular G-quadruplex and the recognizing mechanism. Nucleic Acid Res, 2010, 38: 1022–1033
Bean R C, Shepherd W C, Kay R E, et al. Spectral changes in a cationic dye due to interaction with macromolecules. III. Stoichiometry and mechanism of the complexing reaction1. J Phy Chem, 1965, 69: 4368–4379
Mcrae E G, Kasha M. Enhancement of phosphoescence ability upon aggregation of dye molecules. J Chem Phys, 1958, 28: 721–722
Herz A H. Aggregation of sensitizing dyes in solution and their adsorption onto silver halides. Adv Colloid Interface Sci, 1977, 8: 237–298
Chen C P, Zhou B M, Li D H, et al. Electron-transfer events in solutions of cyanine dyes. J Photochem Photobiol A Chem, 1995, 89: 25–29
Guo C, Xiang J, Feng J, et al. Effect of TiO2 colloids on the fluorescence behavior of two cyanine dyes. J Colloid Interface Sci, 2002, 246: 401–409
Ambrus A, Chen D, Dai J X, et al. Solution structure of the biologically relevant g-quadruplex element in the human c-myc promoter implications for G-quadruplex stabilization. Biochemistry, 2005, 44: 2048–2058
Phan A T, Kuryavyi V, Gaw H Y, et al. Small-molecule interaction with a five-guanine-tract G-quadruplex structure from the human MYC promoter. Nat Chem Biol, 2005, 1: 167–173
Kettani A, Basu G, Gorin A, et al. A two-stranded template-based approach to G.(C-A) triad formation: Designing novel structural elements into an existing DNA framework. J Mol Biol, 2000, 301: 129–146
Kettani A, Bouaziz S, Skripkin E, et al. Interlocked mismatch-aligned arrowhead DNA motifs. Structure, 1999, 7: 803–815
Kettani A, Gorin A, Majumdar A, et al. A dimeric DNA interface stabilized by stacked A.(G.G.G.G).A hexads and coordinated monovalent cations. J Mol Biol, 2000, 297: 627–644
Zhang Z J, Dai J X, Veliath E, et al. Structure of a two-G-tetrad intramolecular G-quadruplex formed by a variant human telomeric sequence in K+ solution: Insights into the interconversion of human telomeric G-quadruplex structures. Nucleic Acids Res, 2010, 38: 1009–1021
Author information
Authors and Affiliations
Corresponding authors
Additional information
This article is published with open access at Springerlink.com
Electronic supplementary material
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Gai, W., Yang, Q., Xiang, J. et al. Roles of flanking sequences in the binding between unimolecular parallel-stranded G-quadruplexes and ligands. Chin. Sci. Bull. 58, 731–740 (2013). https://doi.org/10.1007/s11434-012-5639-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-012-5639-4