Abstract
A new reduced chemical kinetic model of 1-pentene is developed based on a detailed chemical kinetic model of 1-pentene at high temperature (194 species and 1266 reactions). By means of sensitivity and flow rate analyses, 32 important reactions are determined, on the basis of which a reduced chemical kinetic model consisting 50 species and 251 reactions is proposed through addition of other important species and reactions. The results show that the simulated cylinder pressure and temperature calculated using the reduced model agree well with those from the detailed model. The reduced model requires less computing time than the detailed model, which enables coupled computation of the chemical kinetic model with computational fluid dynamics.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Jiang Y, Qiu R, Fan W C. A kinetic modeling study of pollutant formation in premixed hydrocarbon flames. Chin Sci Bull, 2005, 50: 276–281
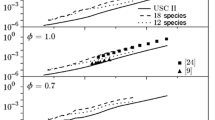
Touchard S, Buda F, Dayma G, et al. Experimental and modeling study of the oxidation of 1-pentene at high temperature. Int J Chem Kinet, 2005, 37: 451–463
Heyberger B, Belmekki N, Conraud V, et al. Oxidation of small alkenes at high temperature. Int J Chem Kinet, 2002, 34: 666–677
Heyberger B, Battin-leclerc F, Warth V, et al. Comprehensive mechanism for the gas-phase oxidation of propene. Combust Flame, 2001, 126: 1780–1802
Touchard S, Fournet R, Glaude P A, et al. Computer aided generation of kinetic mechanism-application to the oxidation of large alkenes at low-temperature. In: Chauveau C, Vovelle C, eds. Proceedings of the European Combustion Meeting, Orleans, France, 2003. 1–6
Touchard S, Fournet R, Glaude P A, et al. Modeling of the oxidation of large alkenes at low temperature. P Combust Inst, 2004, 30: 1073–1081
Ribaucour M, Minetti R, Sochet L R. Autoignition of n-pentane and 1-pentene: Experimental data and kinetic modeling. P Combust Inst, 1998, 27: 345–351
Prabhu S K, Bhat R K, Miller D L, et al. 1-pentene oxidation and its interaction with nitric oxide in the low and negative temperature coefficient regions. Combust Flame, 1996, 104: 377–390
Alatorre G G, Böhm H, Burak A, et al. Experimental and modelling study of 1-pentene combustion at fuel-rich conditions. Phys Chem, 2001, 215: 981–995
Huang H Z, Su W H. Development and optimization of a new reduced chemical kinetics for n-heptane combustion (in Chinese). J Eng Thermophysi, 2006, 27: 883–886
Zhao C P, Chen S Q, Li Y L, et al. Reduction of a detailed chemical kinetics model of n-heptane combustion and its validity analysis (in Chinese). Trans Chin Soc Inter Combust Engines, 2008, 26: 346–352
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Feng, H., Meng, X., Cheng, G. et al. Analysis and reduction of a detailed chemical kinetic model of 1-pentene combustion at high temperature. Chin. Sci. Bull. 58, 1072–1078 (2013). https://doi.org/10.1007/s11434-012-5616-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-012-5616-y