Abstract
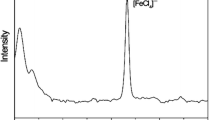
A series of iron(III)-containing imidazolium salts of the general formula [DRim][FeX4] (R = 2,6-diisopropylphenyl, IPr, X = Cl, 1; R = IPr, X = Br, 2; R = tertbutyl, tBu, X = Cl, 3; R = isopropyl, iPr, X = Cl, 4; R = benzyl, Bn, X = Cl, 5; R = Bn, X = Br, 6) have been prepared in high yields via reactions of anhydrous ferric halides with equivalent of the corresponding N,N-dihydrocarby-limidazolium halides, where 2–6 are novel ones. All of the complexes were characterized by elemental analysis, Raman spectroscopy, electrospray ionization mass spectroscopy, and X-ray crystallography for 1 and 2. All of them were non-hygroscopic and air-stable, with four of them existing as solids (1–4) and two as liquids (5 and 6) at room temperature. A preliminary catalytic study on the coupling of 4-tolylmagnesium bromide with cyclohexyl bromide revealed that 1 and 3 possessed the highest activity. In comparison, 2, 4 and 5 exhibited moderate activity and the least active complex was 6.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bolm C, Legros J, Paih J L, et al. Iron-catalyzed reactions in organic synthesis. Chem Rev, 2004, 104: 6217–6254
Martin R, Fürstner A. Cross-coupling of alkyl halides with aryl Grignard reagents catalyzed by a low-valent iron complex. Angew Chem Int Ed, 2004, 43: 3955–3957
Fürstner A, Martin R, Krause H, et al. Preparation, structure, and reactivity of nonstabilized organoiron compounds. Implications for iron-catalyzed cross coupling reactions. J Am Chem Soc, 2008, 130: 8773–8787
Bedford R B, Bruce D W, Frost R M, et al. Iron(III) salen-type catalysts for the cross-coupling of aryl Grignards with alkyl halides bearing β-hydrogens. Chem Commum, 2004, 2822–2823
Martin R B, Betham M, Bruce D W, et al. Iron nanoparticles in the coupling of alkyl halides with aryl Grignard reagents. Chem Commun, 2006, 1398–1400
Chowdhury R R, Crane A K, Fowler C, et al. Iron(III) aminebis(phenolate) complexes as catalysts for the coupling of alkyl halides with aryl Grignard reagents. Chem Commun, 2008, 94–96
Qian X, Dawe L N, Kozak C M. Catalytic alkylation of aryl Grignard reagents by iron(III) amine-bis(phenolated) complexes. Dalton Trans, 2011, 40: 933–943
Reckling A M, Martin D, Dawe L N, et al. Structure and C-C cross-coupling reactivity of iron(III) complexes of halogenated amine-bis(phenolate) ligands. J Organomet Chem, 2011, 696: 787–794
Hasan K, Dawe L N, Kozak C M. Synthesis, structure, and C-C cross-coupling activity of (amine)bis(phenolato)iron(acac) complexes. Eur J Inorg Chem, 2011, 4610–4621
Nagano T, Hayashi T. Iron-catalyzed Grignard cross-coupling with alkyl halides possessing β-hydrogens. Org Lett, 2004, 6: 1297–1299
Cahiez G, Habiak V, Duplais C, et al. Iron-catalyzed alkylations of aromatic Grignard reagents. Angew Chem Int Ed, 2007, 46: 4364–4366
Bica K, Gaertner P. An iron-containing ionic liquid as recyclable catalyst for aryl Grignard cross-coupling of alkyl halides. Org Lett, 2006, 8: 733–735
Alexznder M V, Khandekar A C, Samant S D. Sulfonylation reactions of aromatics using FeCl3-based ionic liquids. J Mol Catal A, 2004, 223: 75–83
Chen X, Peng Y. Chloroferrate(III) ionic liquid: Efficient and recyclable catalyst for solvent-free synthesis of 3,4-dihydropyrimidin-2(1H)-ones. Catal Lett, 2008, 122: 310–313
Nguyen M D, Nguyen L V, Jeon E H, et al. Fe-containing ionic liquids as catalysts for the dimerization of bicycle[2.2.1]hepta-2,5-diene. J Catal, 2008, 258: 5–13
Li H M, Zhu W S, Wang Y, et al. Deep oxidative desulfurization of fuels in redox ionic liquids based on iron chloride. Green Chem, 2009, 11: 810–815
Wang H, Yan R Y, Li Z X, et al. Fe-containing magnetic ionic liquid as an effective catalyst for the glycolysis of poly(ethylene terephthalate). Catal Commun, 2010, 11: 763–767
Gao J, Wang J Q, Song Q W, et al. Iron(III)-based ionic liquid-catalyzed regioselective benzylation of arenas and heteroarenes. Green Chem, 2011, 13: 1182–1186
Olivier-Bourbigou H, Magna L, Morvan D. Ionic liquids and catalysis: Recent progress from knowledge to applications. Appl Catal A, 2010, 373: 1–56
Hu X B, Sun Y, Mao J Y, et al. Theoretical study on the structure-reactivity relationships of acetylacetone-Fe catalyst modified by ionic compound in C-H activation reaction. J Catal, 2010, 272: 320–332
Abbott A P, Frisch G, Ryder K S. Metal complexation in ionic liquids. Annu Rep Prog Chem, Sect A, 2008, 104: 21–45
Bäcker T, Breunig O, Valldor M, et al. In-situ crystal growth and properties of the magnetic. Cryst Growth Des, 2011, 11: 2564–2571
Wang K F, Zhang L, Zhuang R R, et al. An iron(III)-containing ionic liquid: Characterization, magnetic. Transition Met Chem, 2011, 36: 785–791
Yan C H, Wang L L, Gao H H, et al. An efficient and recyclable iron(III)-containing imidazolium salt catalyst for cross-coupling of aryl Grignard reagents with alkyl halides. Chin Sci Bull, 2012, 57: 1953–1958
Gao H H, Yan C H, Tao X P, et al. Synthesis of anionic iron(II) complex bearing an N-heterocyclic carbine ligand and its catalysis for aryl Grignard cross-coupling of alkyl halides. Organometallics, 2010, 29: 4189–4192
Jafarpour L, Stevens E D, Nolan S P. A sterically demanding nucleophilic carbene: 1,3-bis(2,6-diisopropylphenyl)imidazol-2-ylidene). Thermochemistry and catalytic application in olefin metathesis. J Organomet Chem, 2000, 606: 49–54
Herrmann W A, Böhm V P W, Gstöttmayr C W K, et al. Synthesis, structure and catalytic application of palladium(II) complexes bearing N-heterocyclic carbenes and phosphines. J Organomet Chem, 2001, 617–618: 616–628
Arduengo III A J. Preparation of 1,3-disubstituted imidazolium salts. US Pat, 5 077 414, 1991-12-31
Sitze M S, Schreiter E R, Patterson E V, et al. Ionic liquids based on FeCl3 and FeCl2. Raman scattering and ab initio calculations. Inorg Chem, 2001, 40: 2298–2304
Avery J S, Burbride C D, Goodume D M L. Raman spectra of tetrahalo-anions of FeIII, MnII, FeII, CuII and ZnII. Spectrochimica Acta, 1968, 24A: 1721–1726
Lowe C B, Schultz A J, Shaviv R, et al. Magnetochemistry of the tetrahaloferrate(III) ions. 7. Crystal structure and magnetic ordering in (pyridinium)3Fe2Br9. Inorg Chem, 1994, 33: 3051–3054
Steiner T. Hydrogen-bond distances to halide ions in organic and organometallic crystal structures: Up-to-date database study. Acta Cryst, 1998, B54: 456–463
Beford R B, Betham M, Bruce D W, et al. Iron-phosphine, -phosphite, -arsine, and -carbene catalysts for the coupling of primary and secondary alkyl halides with aryl Grignard reagents. J Org Chem, 2006, 71: 1104–1110
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Xia, Y., Yan, C., Li, Z. et al. Synthesis, structure of iron(III)-containing imidazolium salts and their catalytic activity in the alkylation of aryl Grignard reagents. Chin. Sci. Bull. 58, 493–499 (2013). https://doi.org/10.1007/s11434-012-5513-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-012-5513-4