Abstract
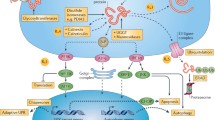
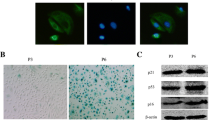
To investigate proteins expressed in the renal tissue of the passive Heymann nephritis (pHN) rat model, we prepared pHN rat models with anti-FxA1 serum and analyzed the proteins differentially expressed in the kidney tissue with label-free liquid chromatography- tandem mass spectrometry. We then analyzed in depth the endoplasmic reticulum stress (ERS)-related protein using an online bioinformatics platform. Forty-one differential proteins and their annotations were obtained. Gene Ontology (GO) function analysis showed that 16 proteins were involved in cellular metabolism and 22 were proteins related to catalytic activity, including protein folding or ATPase. Protein-GO networks indicated that VCP could interact with the ERS marker HSPa5, with both involved in a single pathway. On inhibition of podocyte VCP by RNAi under normal conditions, the HSPa5 expression level did not change, but when the cell was subjected to ERS by tunicamycin, HSPa5 expression significantly increased with RNAi of VCP when compared with the tunicamycin-treated group. Our results showed that ERS plays an important role in podocyte injury of membranous nephropathy and is mediated by an HSPa5-VCP signaling pathway, in which the most predominant proteins are those related to cellular metabolism and catalytic activity.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Sharpstone P, Ogg C S, Cameron J S. Nephrotic syndrome due to primary renal disease in adults: I. Survey of incidence in south east England. Br Med J, 1969, 2: 533–535
Rivera F, Lopez-Gomez J M, Perez-Garcia R. Clinicopathologic correlations of renal pathology in Spain. Kidney Int, 2004, 66: 898–904
Chen Z H, Qin W S, Zeng C H, et al. Triptolide reduces proteinuria in experimental membranous nephropathy and protects against C5b-9-induced podocyte injury in vitro. Kidney Int, 2010, 77: 974–988
Nangaku M, Pippin J, Richardson C A, et al. Beneficial effects of systemic immunoglobulin in experimental membranous nephropathy. Kidney Int, 1996, 50: 2054–2062
Jefferson J A, Pippin J W, Shankland S J. Experimental models of membranous nephropathy. Drug Discov Today Dis Models, 2010, 7: 27–33
Wu Q, Fan K, Sha W, et al. Highly sensitive detection of melamine based on reversed phase liquid chromatography mass spectrometry. Chin Sci Bull, 2009, 54: 732–737
Old W M, Meyer-Arendt K, Aveline-Wolf L, et al. Comparison of label-free methods for quantifying human proteins by shotgun proteomics. Mol Cell Proteomics, 2005, 4: 1487–1502
Asara J M, Christofk H R, Freimark L M, et al. A label-free quantification method by MS/MS TIC compared to SILAC and spectral counting in a proteomics screen. Proteomics, 2008, 8: 994–999
Dayarathna M K, Hancock W S, Hincapie M. A two step fractionation approach for plasma proteomics using immunodepletion of abundant proteins and multi-lectin affinity chromatography: Application to the analysis of obesity, diabetes, and hypertension diseases. J Sep Sci, 2008, 31: 1156–1166
Patel V J, Thalassinos K, Slade S E, et al. A comparison of labeling and label-free mass spectrometry-based proteomics approaches. J Proteome Res, 2009, 8: 3752–3759
Ke C Y, Geng X D. A new chromatographic method for fast separation of active proteins. Chin Sci Bull, 2008, 53: 1113–1116
Chant S, Katz A, Silverman M. Pathogenicity of a highly purified brush border membrane preparation in Heymann nephritis. J Clin Lab Immunol, 1980, 4: 133–140
Noble B, Van Liew J B, Andres G A, et al. Factors influencing susceptibility of LEW rats to Heymann nephritis. Clin Immunol Immunopathol, 1984, 30: 241–254
Bhan A K, Schneeberger E E, Baird L G, et al. Studies with monoclonal antibodies against brush border antigens in Heymann nephritis. Lab Invest, 1985, 53: 421–432
Shankland S J, Pippin J W, Reiser J, et al. Podocytes in culture: Past, present, and future. Kidney Int, 2007, 72: 26–36
Quan H, Peng X, Yang L, et al. An in-depth analysis of proteomics expression profiling in rat glomeruli utilizing LC-MS. Chin Sci Bull, 2010, 55: 2142–2151
Apweiler R, Bairoch A, Wu C H, et al. UniProt: The Universal Protein knowledgebase. Nucleic Acids Res, 2004, 32: D115–119
Yuan J S, Reed A, Chen F, et al. Statistical analysis of real-time PCR data. BMC Bioinformatics, 2006, 7: 85
Fauci A S, Braunwald E, Kasper D L, et al. Harrison’s Principles of Internal Medicine. 17th ed. New York: McGraw-Hill, 2008. 1789–1790
Heymann W, Hackel D B, Harwood S, et al. Production of nephrotic syndrome in rats by Freund’s adjuvants and rat kidney suspensions. Proc Soc Exp Biol Med, 1959, 100: 660–664
Bonegio R G, Fuhro R, Wang Z, et al. Rapamycin ameliorates proteinuria-associated tubulointerstitial inflammation and fibrosis in experimental membranous nephropathy. J Am Soc Nephrol, 2005, 16: 2063–2072
Weissinger E M, Wittke S, Kaiser T, et al. Proteomic patterns established with capillary electrophoresis and mass spectrometry for diagnostic purposes. Kidney Int, 2004, 65: 2426–2434
Ngai H H, Sit W H, Jiang P P, et al. Serial changes in urinary proteome profile of membranous nephropathy: Implications for pathophysiology and biomarker discovery. J Proteome Res, 2006, 5: 3038–3047
Wang X, Eno C O, Altman B J, et al. ER stress modulates cellular metabolism. Biochem J, 2011, 435: 285–296
Johnston J A, Ward C L, Kopito R R. Aggresomes: A cellular response to misfolded proteins. J Cell Biol, 1998, 143: 1883–1898
Katiyar S, Li G, Lennarz W J. A complex between peptide: N-glycanase and two proteasome-linked proteins suggests a mechanism for the degradation of misfolded glycoproteins. Proc Natl Acad Sci USA, 2004, 101: 13774–13779
Lilley B N, Ploegh H L. Multiprotein complexes that link dislocation, ubiquitination, and extraction of misfolded proteins from the endoplasmic reticulum membrane. Proc Natl Acad Sci USA, 2005, 102: 14296–14301
Tavaria M, Gabriele T, Anderson R L, et al. Localization of the gene encoding the human heat shock cognate protein, HSP 73, to chromosome 11. Genomics, 1995, 29: 266–268
Yang Y, Turner R S, Gaut J R. The chaperone BiP/GRP78 binds to amyloid precursor protein and decreases Abeta40 and Abeta42 secretion. J Biol Chem, 1998, 273: 25552–25555
Heymann J B, Iwasaki K, Yim Y I, et al. Visualization of the binding of Hsc70 ATPase to clathrin baskets: Implications for an uncoating mechanism. J Biol Chem, 2005, 280: 7156–7161
Rapoport I, Boll W, Yu A, et al. A motif in the clathrin heavy chain required for the Hsc70/auxilin uncoating reaction. Mol Biol Cell, 2008, 19: 405–413
Pleasure I T, Black M M, Keen J H. Valosin-containing protein, VCP, is a ubiquitous clathrin-binding protein. Nature, 1993, 365: 459–462
Shi J, Dixon R A, Gonzales R A, et al. Identification of cDNA clones encoding valosin-containing protein and other plant plasma membrane-associated proteins by a general immunoscreening strategy. Proc Natl Acad Sci USA, 1995, 92: 4457–4461
Nagahama M, Suzuki M, Hamada Y, et al. SVIP is a novel VCP/p97-interacting protein whose expression causes cell vacuolation. Mol Biol Cell, 2003, 14: 262–273
Heo J M, Livnat-Levanon N, Taylor E B, et al. A stress-responsive system for mitochondrial protein degradation. Mol Cell, 2010, 40: 465–480
Tresse E, Salomons F A, Vesa J, et al. VCP/p97 is essential for maturation of ubiquitin-containing autophagosomes and this function is impaired by mutations that cause IBMPFD. Autophagy, 2010, 6: 217–227
Cunningham P N, Hack B K, Ren G, et al. Glomerular complement regulation is overwhelmed in passive Heymann nephritis. Kidney Int, 2001, 60: 900–909
Glassock R J. The pathogenesis of idiopathic membranous nephropathy: A 50-year odyssey. Am J Kidney Dis, 2010, 56: 157–167
Inagi R, Nangaku M, Onogi H, et al. Involvement of endoplasmic reticulum (ER) stress in podocyte injury induced by excessive protein accumulation. Kidney Int, 2005, 68: 2639–2650
He F, Chen S, Wang H, et al. Regulation of CD2-associated protein influences podocyte endoplasmic reticulum stress-mediated apoptosis induced by albumin overload. Gene, 2011, 484: 18–25
Lai M T, Huang K L, Chang W M, et al. Geldanamycin induction of grp78 requires activation of reactive oxygen species via ER stress responsive elements in 9L rat brain tumour cells. Cell Signal, 2003, 15: 585–595
Matthews J A, Belof J L, Acevedo-Duncan M, et al. Glucosamine-induced increase in Akt phosphorylation corresponds to increased endoplasmic reticulum stress in astroglial cells. Mol Cell Biochem, 2007, 298: 109–123
Kammoun H L, Chabanon H, Hainault I, et al. GRP78 expression inhibits insulin and ER stress-induced SREBP-1c activation and reduces hepatic steatosis in mice. J Clin Invest, 2009, 119: 1201–1215
Vij N, Fang S, Zeitlin P L. Selective inhibition of endoplasmic reticulum-associated degradation rescues DeltaF508-cystic fibrosis transmembrane regulator and suppresses interleukin-8 levels: Therapeutic implications. J Biol Chem, 2006, 281: 17369–17378
Lass A, McConnell E, Nowis D, et al. A novel function of VCP (valosin-containing protein; p97) in the control of N-glycosylation of proteins in the endoplasmic reticulum. Arch Biochem Biophys, 2007, 462: 62–73
Zhong X, Shen Y, Ballar P, et al. AAA ATPase p97/valosin-containing protein interacts with gp78, a ubiquitin ligase for endoplasmic reticulum-associated degradation. J Biol Chem, 2004, 279: 45676–45684
Wojcik C, Rowicka M, Kudlicki A, et al. Valosin-containing protein (p97) is a regulator of endoplasmic reticulum stress and of the degradation of N-end rule and ubiquitin-fusion degradation pathway substrates in mammalian cells. Mol Biol Cell, 2006, 17: 4606–4618
Author information
Authors and Affiliations
Corresponding authors
Additional information
These authors contributed equally to this work.
Recommended by Prof. ZHANG Xue (Editorial Board Member)
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Huang, Z., Hong, Q., Xue, P. et al. A proteome-wide screen identifies valosin-containing protein as an essential regulator of podocyte endoplasmic reticulum stress. Chin. Sci. Bull. 57, 2493–2505 (2012). https://doi.org/10.1007/s11434-012-5250-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-012-5250-8