Abstract
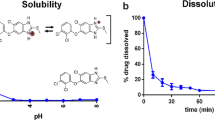
Artemisinin (ART) is a sesquiterpene lactone with an endo-peroxide bridge that is thought to be responsible for its antimalarial activity. It has low oral bioavailability because of aqueous insolubility, which leads to local toxicity at the site of aggregation. The present work focused on increasing its solubility and evaluating its permeation across a model membrane to mimic transdermal delivery that bypasses the hepatic metabolism. For this purpose, physical mixtures (PM), solid dispersions (SD) and lyophilized dispersions (LD) with different drug-polymer ratios (1:0.5, 1:1, 1:2, 1:4 and 1:9) were prepared using the hydrophilic polymer polyvinylpyrrolidone (PVP). Drug-polymer dispersions were characterized using X-ray diffraction (XRD) and Fourier transform infrared spectroscopy (FTIR). Solubility was measured in three solvents: de-ionized water, phosphate buffered saline (PBS) and methanol. The toluene-water partition coefficient was evaluated and compared with the literature and calculated logP values. In vitro diffusion of ART was studied across a polydimethylsiloxane membrane from a saturated solution of drug-polymer dispersions. XRD patterns showed a gradual decrease in crystallinity of ART with increasing polymer concentration, while FTIR confirmed no interactions between ART and PVP. Solubility was increased up to 4-, 5- and 8-fold for LD in water, PBS and methanol, respectively. The logP for toluene-water was 2.65 ± 0.3, which is in good agreement with literature and calculated logP values. Permeation was enhanced, which is attributed to the decrease in crystallinity and increase in wettability of the drug. The ART flux was significantly higher than that of pure ART (0.12 ± 0.01) with increasing PVP concentration for SD and LD formulations. In conclusion, drug-polymer dispersions with PVP improve the pharmaceutical properties of ART in the order LD>SD>PM.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Svensson U S H, Sandström R, Carlborg Ö, et al. High in situ rat intestinal permeability of artemisinin unaffected by multiple dosing and with no evidence of P-glycoprotein involvement. Drug Metab Dispos, 1999, 27: 227–232
Thaithong S, Beale G H. Susceptibility of Thai isolates of Plasmodium falciparum to artemisinine [qinghaosu] and artemether. Bull World Health Organ, 1985, 63: 617–619
Klayman D L. Qinghaosu [artemisinin]: An antimalarial drug from China. Science, 1985, 228: 1049–1055
Titulaer H, Zuidema J, Kager P, et al. The pharmacokinetics of artemisinin after oral, intramuscular and rectal administration to volunteers. J Pharma Pharmacol, 1990, 42: 810–813
Wong J, Yuen K. Improved oral bioavailability of artemisinin through inclusion complexation with (beta)-and (gamma)-cyclodextrins. Int J Pharm, 2001, 227: 177–185
Sahoo N G, Kakran M, Li L, et al. Dissolution enhancement of a poorly water-soluble antimalarial drug by means of a modified multi-fluid nozzle pilot spray drier. Mat Sci Eng C, 2011, 31: 391–399
Stoughton R B. Percutaneous absorption. South Med J, 1962, 55: 1134–1138
Kim J H, Choi H K. Effect of additives on the crystallization and the permeation of ketoprofen from adhesive matrix. Int J Pharm, 2002, 236: 81–85
Barry B W. Skin transport. In: Dermatological Formulations Percutaneous Absorption. New York: Marcel Dekker, 1983. 95–126
Iwasa A, Irimoto K, Kasai S, et al. Effect of nonionic surfactants on percutaneous absorption of diclofenac sodium. Yakuzaigaku, 1991, 51: 16–21
Javadzadeh Y, Shokri J, Hallaj-Nezhadi S, et al. Enhancement of percutaneous absorption of Finasteride by cosolvents, cosurfactant and surfactants. Pharm Dev Technol, 2009: 1–7
Pathan I B, Setty C M. Chemical penetration enhancers for transdermal drug delivery systems. Trop J Pharm Res, 2009, 8: 173–179
Karavas E, Ktistis G, Xenakis A, et al. Effect of hydrogen bonding interactions on the release mechanism of felodipine from nanodispersions with polyvinylpyrrolidone. Eur J Pharm Biopharm, 2006, 63: 103–114
Ruan L P, Yu B Y, Fu G M, et al. Improving the solubility of ampelopsin by solid dispersions and inclusion complexes. J Pharm Biomed Anal, 2005, 38: 457–464
Tantishaiyakul V, Kaewnopparat N, Ingkatawornwong S. Properties of solid dispersions of piroxicam in polyvinylpyrrolidone K-30. Int J Pharm, 1996, 143: 59–66
Van Nijlen T, Brennan K, Van den Mooter G, et al. Improvement of the dissolution rate of artemisinin by means of supercritical fluid technology and solid dispersions. Int J Pharm, 2003, 254: 173–181
Akbuga J, Gürsoy A, Kendi E. The preparation and stability of fast release furosemide-PVP solid dispersion. Drug Dev Ind Pharm, 1988, 14: 1439–1464
Margarit M V, Marín M T, Contreras M D. Solubility of solid dispersions of pizotifen malate and povidone. Drug Dev Ind Pharm, 2001, 27: 517–522
El-Badry M, Fathy M. Enhancement of the dissolution and permeation rates of meloxicam by formation of its freeze-dried solid dispersions in polyvinylpyrrolidone K-30. Drug Dev Ind Pharm, 2006, 32: 141–150
Zhao S S, Zeng M Y. Spektrometrische Hochdruck-Flüssigkeits-Chromatographische [HPLC] Untersuchungen zur Analytik von Qinghaosu. Planta Med, 1985, 51: 233–237
Ansari M T, Iqbal I, Sunderland V B. Dihydroartemisinin-cyclodextrin complexation: Solubility and stability. Arch Pharmacal Res, 2009, 32: 155–165
Franz T J. Percutaneous absorption: On the relevance of in vitro data. J Invest Dermatol, 1975, 64: 190–195
Avery M A, Bonk J D, Chong W K M, et al. Structure-activity relationships of the antimalarial agent artemisinin. 2. Effect of heteroatom substitution at O-11: Synthesis and bioassay of N-alkyl-11-aza-9-desmethylartemisinins. J Med Chem, 1995, 38: 5038–5044
Sammour O A, Hammad M A, Megrab N A, et al. Formulation and optimization of mouth dissolve tablets containing rofecoxib solid dispersion. AAPS PharmSciTech, 2006, 7: 167–175
Chiou W L, Riegelman S. Pharmaceutical applications of solid dispersions. J Pharm Sci, 1971, 60: 1281–1302
Abdelkader H, Abdallah O Y, Salem H S. Comparison of the effect of tromethamine and polyvinylpyrrolidone on dissolution properties and analgesic effect of nimesulide. AAPS PharmSciTech, 2007, 8: 110–117
Shah J, Vasanti S, Anroop B, et al. Enhancement of dissolution rate of valdecoxib by solid dispersions technique with PVP K 30 & PEG 4000: Preparation and in vitro evaluation. J Incl Phen Macro, 2009, 63: 69–75
Liu C, Desai K G H. Enhancement of dissolution rate of valdecoxib using solid dispersions with polyethylene glycol 4000. Drug Dev Ind Pharm, 2005, 31: 1–10
Modi A, Tayade P. Enhancement of dissolution profile by solid dispersion [kneading] technique. AAPS PharmSciTech, 2006, 7: 87–92
Betageri G V, Makarla K R. Enhancemnet of dissolution of glyburide by solid dispersion and lyophilisation techniques. Int J Pharm, 1995, 126: 155–160
Barry B W. Properties that influence. In: Dermatological Formulations Percutaneous Absorption. New York: Marcel Decker, 1983. 127–233
Chow D. Concentration dependent enhancement of 1-dodecylazacy-cloheptan-2-one on the percutaneous penetration kinetics of triamcinolone acetonide. J Pharm Sci, 1984, 73: 1794–1799
Huang Y B, Wu P C, Ko H M, et al. Cardamom oil as a skin permeation enhancer for indomethacin, piroxicam and diclofenac. Int J Pharm, 1995, 126: 111–117
Dias M, Hadgraft J, Lane M E. Influence of membrane-solvent-solute interactions on solute permeation in model membranes. Int J Pharm, 2007, 336: 108–114
Watkinson A C, Joubin H, Green D M, et al. The influence of vehicle on permeation from saturated solutions. Int J Pharm, 1995, 121: 27–36
Raghavan S, Kiepfer B, Davis A, et al. Membrane transport of hydrocortisone acetate from supersaturated solutions; the role of polymers. Int J Pharm, 2001, 221: 95–105
Kim J H, Lee C H, Choi H K. Transdermal delivery of physostigmine: Effects of enhancers and pressure-sensitive adhesives. Drug Dev Ind Pharm, 2002, 28: 833–839
Zhang J, Liu Z, Du H, et al. A novel hydrophilic adhesive matrix with self-enhancement for drug percutaneous permeation through rat skin. Pharm Res, 2009, 26: 1398–1406
Author information
Authors and Affiliations
Corresponding author
Additional information
This Article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Shahzad, Y., Shah, S.N.H., Ansari, M.T. et al. Effects of drug-polymer dispersions on solubility and in vitro diffusion of artemisinin across a polydimethylsiloxane membrane. Chin. Sci. Bull. 57, 1685–1692 (2012). https://doi.org/10.1007/s11434-012-5094-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-012-5094-2