Abstract
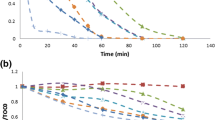
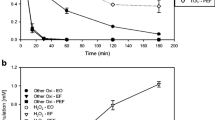
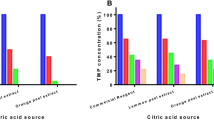
In this study, photo-Fenton oxidation was applied to degradation of sulfamonomethoxine sodium (SMMS) in aqueous solution. The operation parameters of pH, temperature, and concentrations of H2O2, Fe2+ and SMMS were investigated. The optimum conditions for the photo-Fenton process were determined as follows: [SMMS]=4.53 mg/L, pH 4.0, [H2O2]=0.49 mmol/L, [Fe2+]= 19.51 μmol/L and T=25°C. Under these conditions 98.5% of the SMMS degraded. The kinetics were also studied, and degradation of SMMS by the photo-Fenton process could be described by first-order kinetics. The apparent activation energy was calculated as 23.95 kJ/mol. Mineralization of the process was investigated by measuring the chemical oxygen demand (COD), and the COD decreased by 99% after 120 min. This process could be used as a pretreatment method for wastewater containing sulfa-monomethoxine sodium.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Halling-Sorensen B, Nielsen S N, Lanzky P F, et al. Occurrence, fate and effects of pharmaceutical substances in the environment-A review. Chemosphere, 1998, 36: 357–394
Daugthon C G, Ternes T A. Pharmaceuticals and personal care products in the environment: Agents of subtle change? Environ Health Perspect, 1999, 107(Suppl 6): 907–938
Latch D E, Stender B L, Packer J L, et al. Photochemical fate of pharmaceuticals in the environment: Cimetidine and ranitidine. Environ Sci Technol, 2003, 37: 3342–3350
Boreen A L, Arnold W A, McNeill K. Photochemical fate of sulfa drugs in the aquatic environment: Sulfa drugs containing five-membered heterocyclic groups. Environ Sci Technol, 2004, 38: 3933–3940
Richardson B J, Lam P K S, Martin M. Emerging chemicals of concern: Pharmaceuticals and personal care products (PPCPs) in Asia, with particular reference to southern China. Mar Pollut Bull, 2005, 50: 913–920
Matamoros V, Bayona J M. Elimination of pharmaceuticals and personal care products in subsurface flow constructed wetlands. Environ Sci Technol, 2006, 40: 5811–5816
Edhlund B L, Arnold W A, McNeill K. Aquatic photochemistry of nitrofuran antibiotics. Environ Sci Technol, 2006, 40: 5422–5427
Xu W H, Zhang G, Li X D, et al. Occurrence and elimination of antibiotics at four sewage treatment plants in the Pearl River Delta (PRD), South China. Water Res, 2007, 41: 4526–4534
Matamoros V, Arias C, Brix H, et al. Removal of pharmaceuticals and personal care products (PPCPs) from urban wastewater in a pilot vertical flow constructed wetland and a sand filter. Environ Sci Technol, 2007, 41: 8171–8177
Miege C, Choubert J M, Ribeiro L, et al. Fate of pharmaceuticals and personal care products in wastewater treatment plants-Conception of a database and first results. Environ Pollut, 2009, 157: 1721–1726
Heberer T. Occurrence, fate, and removal of pharmaceutical residues in the aquatic environment: A review of recent research data. Toxicol Lett, 2002, 131: 5–17
Balmer M E, Buser H R, Müller M D, et al. Occurrence of some organic UV filters in wastewater, in surface waters, and in fish from Swiss lakes. Environ Sci Technol, 2005, 39: 953–962
Lishman L, Smyth S A, Sarafin K, et al. Occurrence and reductions of pharmaceuticals and personal care products and estrogens by municipal wastewater treatment. Sci Total Environ, 2006, 367: 544–558
Xu W H, Zhang G, Zou S C, et al. Determination of selected antibiotics in the Victoria Harbour and the Pearl River, South China using high-performance liquid chromatography-electrospray ionization tandem mass spectrometry. Environ Pollut, 2007, 145: 672–679
Lee J, Lee B C, Ra J S, et al. Comparison of the removal efficiency of endocrine disrupting compounds in pilot scale sewage treatment processes. Chemosphere, 2008, 71: 1582–1592
Zhang Y J, GeiBen S U, Gal C. Carbamazepine and diclofenac: Removal in wastewater treatment plants and occurrence in water bodies. Chemosphere, 2008, 73: 1151–1161
Zhao J L, Ying G G, Wang L, et al. Determination of phenolic endocrine disrupting chemicals and acidic pharmaceuticals in surface water of the Pearl Rivers in South China by gas chromatography-negative chemical ionization-mass spectrometry. Sci Total Environ, 2009, 407: 962–974
Carballa M, Omil F, Lema L M, et al. Behavior of pharmaceuticals, cosmetics and hormones in a sewage treatment plant. Water Res, 2004, 38: 2918–2926
Peng X Z, Wang Z D, Kuang W X, et al. A preliminary study on the occurrence and behavior of sulfonamides, ofloxacin and chloramphenicol antimicrobials in wastewaters of two sewage treatment plants in Guangzhou, China. Sci Total Environ, 2006, 371: 314–322
Benitez F J, Acero J L, Real F J, et al. The role of hydroxyl radicals for the decomposition of p-hydroxyl phenyl acetic acid in aqueous solutions. Water Res, 2001, 35: 1338–1343
Malik P K, Saha S K. Oxidation of direct dyes with hydrogen peroxide using ferrous ion as catalyst. Sep Purif Technol, 2003, 31: 241–250
Anotai J, Lu M C, Chewpreecha P. Kinetics of aniline degradation by Fenton and electro-Fenton processes. Water Res, 2006, 40: 1841–1847
Chen J X, Zhu L Z. Heterogeneous UV-Fenton catalytic degradation of dyestuff in water with hydroxyl-Fe pillared bentonite. Catal Today, 2007, 126: 463–470
Zazo J A, Casas J A, Mohedano A F, et al. Chemical pathway and kinetics of phenol oxidation by Fenton’s reagent. Environ Sci Technol, 2005, 39: 9295–9302
Perez M, Torrades F, Domenech X, et al. Fenton and photo-Fenton oxidation of textile effluents. Water Res, 2002, 36: 2703–2710
Zhao Y P, Hu J Y, Jin W. Transformation of oxidation products and reduction of estrogenic activity of 17β-estradiol by a heterogeneous photo-Fenton reaction. Environ Sci Technol, 2008, 42: 5277–5284
Sun S P, Li C J, Sun J H, et al. Decolorization of an azo dye Orange G in aqueous solution by Fenton oxidation process: Effect of system parameters and kinetic study. J Hazard Mater, 2009, 161: 1052–1057
Kim I, Tanaka H. Photodegradation characteristics of PPCPs in water with UV treatment. Environ Int, 2009, 35: 793–802
Lucas M S, Peres J A. Decolorization of the azo dye Reactive Black 5 by Fenton and photo-Fenton oxidation. Dyes Pigm, 2006, 71: 236–244
Ge L K, Chen J W, Qiao X L, et al. Light-source-dependent effects of main water constituents on photodegradation of phenicol antibiotics: Mechanism and kinetics. Environ Sci Technol, 2009, 43: 3101–3107
Sun J H, Li X Y, Feng J L, et al. Oxone/Co2+ oxidation as an advanced oxidation process: Comparison with traditional Fenton oxidation for treatment of landfill leachate. Water Res, 2009, 43: 4363–4369
Modirshahla N, Behnajady M A, Ghanbary F. Decolorization and mineralization of C.I. Acid Yellow 23 by Fenton and photo-Fenton processes. Dyes Pigm, 2007, 73: 305–310
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Sun, J., Feng, J., Shi, S. et al. Degradation of the antibiotic sulfamonomethoxine sodium in aqueous solution by photo-Fenton oxidation. Chin. Sci. Bull. 57, 558–564 (2012). https://doi.org/10.1007/s11434-011-4887-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-011-4887-z