Abstract
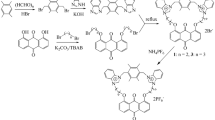
A series of novel imidazolium based ionic liquids containing the urea moiety were designed and synthesized for anion recognition. 1-Ethylurea-3-methylimidazolium acetate ([Eumim]OAc) was used as the receptor for the halides and complex anions (BF4 −, PF6 −, BPh4 −). 1H NMR spectra showed that the urea protons and imidazolium C(2) proton of the receptor ([Eumim]OAc) moved upfield on addition of various anions. A Job plot showed that the [Eumim]OAc receptor formed a 1:1 complex with BPh4 −. X-ray diffraction analysis and the molecular modeling study revealed that the conformations of [Eumim]OAc and [Eumim]BPh4 were different. The conformational change of the cation was caused by anion exchange, and may provide an alternative to current methods for recognition of anions.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Martinez-Manez R, Sancenon F. Fluorogenic and chromogenic chemosensors and reagents for anions. Chem Rev, 2003, 103: 4419–4476
Bondy C R, Loeb S J. Amide based receptors for anions. Coord Chem Rev, 2003, 240: 77–99
Beer P D, Gale P A. Anion recognition and sensing: The state of the art and future perspectives. Angew Chem Int Ed, 2001, 40: 486–516
Kryshtal G V, Zhdankina G M, Zlotin S G. Recoverable phase-transfer catalysts with fluorinated anions: Generation and reactions of dichlorocarbene and CCl3 anion in the heterogeneous system KOH(s)/CHCl3/nBu4NPF6. Eur J Org Chem, 2008, 73: 1777–1782
Shao J, Wang Y H, Lin H, et al. A novel anion receptor employing the pyrrole and amide moieties in the cooperative binding of anions. J Incl Phenom Macrocycl Chem, 2008, 62: 377–381
Bao X P, Yuan J H, Zhou Y H. Selective colorimetric sensing for F− by a cleft-shaped anion receptor containing amide and hydroxyl as recognition units. Sensors Actuators B, 2009, 140: 467–472
Nielsen K A, Cho W S, Jeppesen J O, et al. Tetra-TTF calix[4] pyrrole: A rationally designed receptor for electron-deficient neutral guests. J Am Chem Soc, 2004, 126: 16296–16297
Sessler J L, Katayev E, Pantos G D, et al. Fine tuning the anion binding properties of 2,6-diamidopyridine dipyrromethane hybrid macrocycles. J Am Chem Soc, 2005, 127: 11442–11446
Schug K A, Lindner W. Noncovalent binding between guanidinium and anionic groups: Focus on biological- and synthetic-based arginine/guanidinium interactions with phosph[on]ate and sulf[on]ate residues. Chem Rev, 2005, 105: 67–113
Yoon J, Kim S K, Singh N J, et al. Imidazolium receptors for the recognition of anions. Chem Soc Rev, 2006, 35: 355–360
Xu Z, Kim S K, Yoon J. Revisit to imidazolium receptors for the recognition of anions: Highlighted research during 2006–2009. Chem Soc Rev, 2010, 39: 1457–1466
Amendola V, Esteban-Góamez D, Fabbrizzi L, et al. What anions do to N-H-containing receptors. Acc Chem Res, 2006, 39: 343–353
Zhang Z, Schreiner P R. (Thio)urea organocatalysis—What can be learnt from anion recognition. Chem Soc Rev, 2009, 38: 1187–1198
Welton T. Room-temperature ionic liquids. solvents for synthesis and catalysis. Chem Rev, 1999, 99: 2071–2083
Haumann M, Riisager A. Hydroformylation in room temperature ionic liquids (RTILs): Catalyst and process developments. Chem Rev, 2008, 108: 1474–1497
Zhao D, Wu M, Kou Y, et al. Ionic liquids: Applications in catalysis. Catal Today, 2002, 74: 157–189
Hapiot P, Lagros C. Electrochemical reactivity in room-temperature ionic liquids. Chem Rev, 2008, 108: 2238–2264
Gutowski K E, Broker G A, Willauer H D, et al. Controlling the aqueous miscibility of ionic liquids: Aqueous biphasicsystems of water-miscible ionic liquids and water-structuring salts for recycle, metathesis, and separations. J Am Chem Soc, 2003, 125: 6632–6633
Tsuzuki S, Tokuda H, Hayamizu K, et al. Magnitude and directionality of interaction in ion pairs of ionic liquids: Relationship with ionic conductivity. J Phys Chem B, 2005, 109: 16474–16481
Yu G R, Zhang S J, Zhou G H, et al. Structure, interaction and property of amino-functionalized imidazolium ILs by molecular dynamics simulation and ab initio calculation. AICHE J, 2007, 53: 3210–3221
Dong K, Zhang S J, Wang D X, et al. Hydrogen bonds in imidazolium ionic liquids. J Phys Chem A, 2006, 110: 9775–9782
Frisch M J, Trucks G W, Schlegel H B, et al. Gaussian 03[CP]. Pittsburgh PA: Gaussian, Inc., 2003
Sheldrick G M. Program for Empirical Absorption Correction of Area Detector Data. Göttingen: University of Gottingen, 1996
Sheldrick G M. SHELXTL, Version 5.1. Madison, WI, USA: Bruker Analytical X-ray Instruments Inc., 1998
Visser A E, Swatloski R P, Reichert W M, et al. Task-specific ionic liquids for the extraction of metal ions from aqueous solutions. Chem Commun, 2001, 1: 135–136
Kim Y J, Kwak H, Lee S J, et al. Urea/thiourea-based colorimetric chemosensors for the biologically important ions: Efficient and simple sensors. Tetrahedron, 2006, 62: 9635–9640
Huang W W, Lin H, Cai Z S, et al. A novel anthracene-based receptor: Highly sensitive fluorescent and colorimetric receptor for fluoride. Talanta, 2010, 81: 967–971
Hou X H, Kobiro K. Versatility of an intramolecularly hydrogen-bonded 4-(N,N-dimethylamino)benzoate group as a signaling subunit for anion recognition. Tetrahedron, 2005, 61: 5866–5875
Yoon S Y, Lai Y H. 10a-(4-Biphenyl)-10b-methyl-10a, 10b-dihydropyrene: A conformational study and ring current effect. Tetrahedron Lett, 2008, 49: 4282–4285
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Dong, Z., Yang, Y., Zhang, L. et al. Ionic liquids containing the urea moiety for recognition of halides and complex anions. Chin. Sci. Bull. 57, 473–478 (2012). https://doi.org/10.1007/s11434-011-4722-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-011-4722-6