Abstract
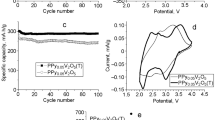
Graphite oxide (GO)/polypyrrole (PPy) nanocomposites (GPYs) were synthesized using in situ polymerization. The effect of the feeding ratios of pyrrole and GO on the structure and electrochemical performances of GPYs was investigated. The structure was characterized via Fourier-transform infrared spectroscopy, scanning electron microscopy, transmission electron microscopy and X-ray diffraction. The electrochemical performance was characterized via cyclic voltammetry, galvanostatic charge-discharge and electrochemical impedance spectroscopy. The results indicate that the more pyrrole is added to GO (with GO concentrations of 20% and 50%), the more agglomeration of both PPy and GO layers occurs. This is detrimental to the capacitance utilization of PPy. When the feeding ratio of GO:pyrrole is 80:20, PPys with nanofibrils are dispersed homogenously in/on the exfoliated layer of GO and the conductivity is enhanced. The capacitance utilization of PPy in a composite with a GO concentration of 80% (383 F/g) is higher than that of pure PPy (201 F/g), which indicates the presence of a synergistic effect between GO and PPy.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Park S, Ruoff R S. Chemical methods for production of graphenes. Nat Nanotechnol, 2009, 4: 217–224
He H, Klinowski J, Forster M, et al. A new structural model for graphite oxide. Chem Phys Lett, 1997, 287: 53–56
Lerf A, He H, Riedl T, et al. 13C and 1H MAS NMR studies of graphite oxide and its chemically modified derivatives. Solid State Ionics, 1997, 101-103: 857–862
Lerf A, He H, Forster M, et al. Structure of graphite oxide revisited. J Phys Chem B, 1998, 102: 4477–4482
Hontoria-Lucas C, Lopez-Peinado A J, Lopez-Gonzalez J D, et al. Study of oxygen-containing groups in a series of graphite oxides: Physical and chemical characterization. Carbon, 1995, 33: 1585–1592
Szabo T, Tombacz E, Illes E, et al. Enhanced acidity and pH dependent surface charge characterization of successively oxidized graphite oxides. Carbon, 2006, 44: 537–545
Nishide H, Oyaizu K. Materials science-Toward flexible batteries. Science, 2008, 319: 737–738
Lakard B, Segut O, Lakard S, et al. Potentiometric miniaturized pH sensors based on polypyrrole films. Sens Actuator B-Chem, 2007, 122: 101–108
Sharma R K, Rastogi A C, Desu S B. Pulse polymerized polypyrrole electrodes for high energy density electrochemical supercapacitor. Electrochem Commun, 2008, 10: 268–272
Cheng Y, Zhao Z S, Zhang F, et al. Preparation of Fe3O4-SiO2-polypyrrole core-shell nanoparticles, and their adsorption of Cr2O 2−7 . Chinese Sci Bull, 2010, 55: 2904–2909
Huang L Y, Hou W B, Liu Z O, et al. Polypyrrole-coated styrene-butyl acrylate copolymer composite particles with tunable conductivity. Chinese Sci Bull, 2005, 50: 971–975
Li F, Shi J J, In X. Synthesis and supercapacitor characteristics of PANI/CNTs composites. Chinese Sci Bull, 2010, 55: 1100–1106
Wang X X, Yang T, Jiao K. Controllable fabrication of Au micro/nanostructures on self-doped polyaniline nanofibers via electrochemical deposition and its application for DNA immobilization. Chinese Sci Bull, 2010, 55: 4125–4131
Han Y Q, Lu Y. Characterization and electrical properties of conductive polymer/colloidal graphite oxide nanocomposites. Composite Sci Technol, 2009, 69: 1231–1237
Gu Z M, Li C Z, Wang G C, et al. Synthesis and characterization of polypyrrole/graphite oxide composite by in situ emulsion polymerization. J Polym Sci Pol Phys, 2010, 48: 1329–1335
Gu Z, Zhang L, Li C. Preparation of highly conductive polypyrrole/graphite oxide composites via in situ polymerization. J Macromol Sci B, 2009, 48: 1093–1102
Han Y Q, Lu Y. Preparation and characterization of graphite oxide/polypyrrole composites. Carbon, 2007, 45: 2394–2399
Hummers W S, Offeman R E. Preparation of graphite oxide. J Am Chem Soc, 1958, 80: 1339
Nakajima T, Mabuchi A, Hagiwara R. A new structure model of graphite oxide. Carbon, 1988, 26: 357–361
Kovtyukhova N I, Ollivier P J, Martin B R, et al. Layer-by-layer assembly of ultrathin composite films from micron-sized graphite oxide sheets and polycations. Chem Mater, 1999, 11: 771–778
Liu Z, Wang Z M, Yang X, et al. Intercalation of organic ammonium ions into layered graphite oxide. Langmuir, 2002, 18: 4926–4932
Bissessur R, Liu P K Y, Scully S F. Intercalation of polypyrrole into graphite oxide. Synthetic Met, 2006, 156: 1023–1027
Liu Y C, Lin Y T. Strategy and characteristics of polypyrrole deposited on silver substrates with silver-containing nanocomplexes. J Phys Chem B, 2003, 107: 11370–11375
Zhang W X, Wen X G, Yang S H. Synthesize and characterization of uniform arrays of copper sulfide nanorods coated with nanolayers of polypyrrole. Langmuir, 2003, 19: 4420–4426
Jiang J H, Kucernak A. Electrochemical supercapacitor material based on manganese oxide: Preparation and characterization. Electrochim Acta, 2002, 47: 2381–2386
Kalpana D. New, low-cost, high-power poly(o-anisidine-co-metanilic acid)/activated carbon electrode for electrochemical supercapacitors. J Power Sources, 2009, 190: 587–591
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Han, Y., Ding, B. & Zhang, X. Effect of feeding ratios on the structure and electrochemical performance of graphite oxide/polypyrrole nanocomposites. Chin. Sci. Bull. 56, 2846–2852 (2011). https://doi.org/10.1007/s11434-011-4646-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-011-4646-1