Abstract
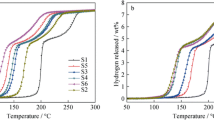
The hydrogen desorption properties of Li2BNH6 were improved by doping with cobalt. With the addition of CoCl2 (7 wt%), more than 8 wt% of hydrogen was released from Li2BNH6 at temperatures below 210°C, which is approximately 90°C lower than that of pristine Li2BNH6. X-ray diffraction, Fourier transform-infrared and Raman characterizations revealed that the dehydrogenation was a stepwise process with the formation of intermediates Li4BN3H10 and LiBH4 and final products of Li3BN2 and LiH. The introduction of Co greatly accelerated the dehydrogenation of Li4BN3H10. X-ray absorption near-edge structure measurements revealed that Co and CoB species formed during ball milling of CoCl2 with LiBH4 and LiNH2, which may function as catalyst in the subsequent dehydrogenation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Schlapbach L, Züttel A. Hydrogen-storage materials for mobile applications. Nature, 2001, 414: 353–358
Bogdanović B, Schwickardi M. Ti-doped alkali metal aluminium hydrides as potential novel reversible hydrogen storage materials. J Alloys Compd, 1997, 253: 1–9
Züttel A, Wenger P, Rentsch S, et al. LiBH4—A new hydrogen storage material. J Power Sources, 2003, 118: 1–7
Chen P, Xiong Z T, Luo J Z, et al. Interaction of hydrogen with metal nitrides and imides. Nature, 2002, 420: 302–304
Leng H Y, Ichikawa T, Hino S, et al. New metal-N-H system composed of Mg(NH2)2 and LiH for hydrogen storage. J Phys Chem B, 2004, 108: 8763–8765
Pinkerton F E, Meisner G P, Meyer M S, et al. Hydrogen desorption exceeding ten weight percent from the new quaternary hydride Li3BN2H8. J Phys Chem B, 2005, 109: 6–8
Aoki M, Miwa K, Noritake T, et al. Destabilization of LiBH4 by mixing with LiNH2. Appl Phys A: Mater Sci Proc, 2005, 80: 1409–1412
Wolf G, Baumann J, Baitalow F, et al. Calorimetric process monitoring of thermal decomposition of B-N-H compounds. Thermochim Acta, 2000, 343: 19–25
Gutowska A, Li L Y, Shin Y S, et al. Nanoscaffold mediates hydrogen release and the reactivity of ammonia borane. Angew Chem Int Ed, 2005, 44: 3578–3582
Xiong Z T, Yong C K, Wu G T, et al. High-capacity hydrogen storage in lithium and sodium amidoboranes. Nat Mater, 2008, 7: 138–141
Diyabalanage H V K, Shrestha R P, Semelsberger T A, et al. Calcium amidotrihydroborate: A hydrogen storage material. Angew Chem Int Ed, 2007, 46: 8995–8997
Schlesinger H I, Brown H C. Metallo borohydrides. III. Lithium borohydride. J Am Chem Soc, 1940, 62: 3429–3435
Vajo J J, Skeith S L, Mertens F. Reversible storage of hydrogen in destabilized LiBH. J Phys Chem B, 2005, 109: 3719–3722
Yang J, Sudik A, Wolverton C. Destabilizing LiBH4 with a metal (M = Mg, Al, Ti, V, Cr, or Sc) or metal hydride (MH2, MgH2, TiH2, or CaH2). J Phys Chem C, 2007, 111: 19134–19140
Lu J, Fang Z G Z. Dehydrogenation of a combined LiAlH4/LiNH2 system. J Phys Chem B, 2005, 109: 20830–20834
Xiong Z T, Wu G T, Hu J J, et al. Reversible hydrogen storage by a Li-Al-N-H complex. Adv Funct Mater, 2007, 17: 1137–1142
Meisner G P, Scullin M L, Balogh M P, et al. Hydrogen release from mixtures of lithium borohydride and lithium amide: A phase diagram study. J Phys Chem B, 2006, 110: 4186–4192
Noritake T, Aoki M, Towata S, et al. Crystal structure analysis of novel complex hydrides formed by the combination of LiBH4 and LiNH2. Appl Phys A: Mater Sci Proc, 2006, 83: 277–279
Pinkerton F E, Meyer M S, Meisner G P, et al. Improved hydrogen release from LiB0.33N0.67H2.67 with metal additives: Ni, Fe, and Zn. J Alloys Compd, 2007, 433: 282–291
Tang W S, Wu G, Liu T, et al. Cobalt-catalyzed hydrogen desorption from the LiNH2-LiBH4 system. Dalton Trans, 2008: 2395-2399
Pinkerton F E, Meyer M S. Hydrogen desorption behavior of nickel-chloride-catalyzed stoichiometric Li4BN3H10. J Phys Chem C, 2009, 113: 11172–11176
He T, Xiong Z T, Wu G T, et al. Nanosized Co- and Ni-catalyzed ammonia borane for hydrogen storage. Chem Mater, 2009, 21: 2315–2318
Chu H L, Xiong Z T, Wu G T, et al. Hydrogen storage properties of Ca(BH4)2-LiNH2 system. Chem Asian J, 2010, 5: 1594–1599
Chater P A, David W I F, Johnson S R, et al. Synthesis and crystal structure of Li4BH4(NH2)3. Chem Commun, 2006: 2439-2441
Kulinich S A, Zhukov A N, Sevast’yanova L G, et al. On some alkali- and alkaline-earth-metal boron nitrides, unsaturated with boron. Diam Relat Mater, 1999, 8: 2152–2158
Yamane H, Kikkawa S, Horiuchi H, et al. Structure of a new polymorph of lithium boron nitride, Li3BN2. J Solid State Chem, 1986, 65: 6–12
Somer M, Herterich U, Curda J, et al. Ternary nitridoborates. 1. LiMg[BN2] and Ba4[BN2] 2O, compounds with the anion [N-B-N]3−: Syntheses, crystal structures, and vibrational spectra. Z Anorg Allg Chem, 1997, 623: 18–24
Chater P A, David W I F, Anderson P A. Synthesis and structure of the new complex hydride Li2BH4NH2. Chem Commun, 2007: 4770–4772
Pinkerton F E, Meyer M S, Meisner G P, et al. Improved hydrogen release from LiB0.33N0.67H2.67 with noble metal additions. J Phys Chem B, 2006, 110: 7967–7974
Orimo S, Nakamori Y, Kitahara G, et al. Dehydriding and rehydriding reactions of LiBH4. J Alloys Compd, 2005, 404–406: 427–430
Pei Y, Guo P J, Qiao M H, et al. The modification effect of Fe on amorphous CoB alloy catalyst for chemoselective hydrogenation of crotonaldehyde. J Catal, 2007, 248: 303–310
Xu Q, Chandra M. Catalytic activities of non-noble metals for hydrogen generation from aqueous ammonia-borane at room temperature. J Power Sources, 2006, 163: 364–370
Liu Y F, Luo K, Zhou Y F, et al. Diffusion controlled hydrogen desorption reaction for the LiBH4/2LiNH2 system. J Alloys Compd, 2009, 481: 473–479
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Zheng, X., Wu, G., He, T. et al. Improved hydrogen desorption properties of Co-doped Li2BNH6 . Chin. Sci. Bull. 56, 2481–2485 (2011). https://doi.org/10.1007/s11434-011-4466-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-011-4466-3