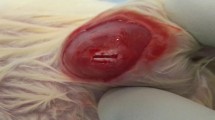
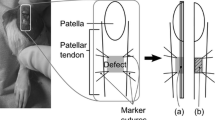
Abstract
The repair of injured tendons remains a great challenge because of the poor intrinsic healing capacity of tendons. In this study, we examined the spatiotemporal expression pattern of the bone morphogenetic protein 2 (bmp-2) gene in normal and experimentally injured rat patellar tendons. We also investigated the ability of exogenously applied BMP-2 to promote patellar tendon healing. In situ hybridization with bmp-2 and alk-6 (bmp-2 receptor) antisense riboprobes revealed that both genes were normally expressed at low levels in intact rat tendons. However, bmp-2 expression was significantly upregulated in tenocytes found in the wound site at 7 d and later following tendon injury. In addition, it was found that bmp-2 was expressed in cultured patellar tenocytes. Application of exogenous BMP-2 to the tendon wound site significantly enhanced tendon repair. Moreover, in vitro and in vivo studies further demonstrated that BMP-2 enhanced tenocyte proliferation and migration to the wound site. The detectable amount of BMP-2 in normal tendons suggests that a basal level of bmp-2 expression was likely present to maintain the normal functions of the patellar tendon. Injury to the tendon induced increased bmp-2 expression intrinsically, but the expression level was insufficient for proper tendon repair. Our findings indicate that it is possible to significantly improve tendon healing by applying exogenous BMP-2 to the wound site, inferring that this protein could be developed as a potential therapeutic reagent for the treatment of damaged tendons.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Hui C, Salmon L J, Kok A, et al. Fifteen-year outcome of endoscopic anterior cruciate ligament reconstruction with patellar tendon autograft for “isolated” anterior cruciate ligament tear. Am J Sports Med, 2011, 39: 89–98
Barber F A, Aziz-Jacobo J, Oro F B. Anterior cruciate ligament reconstruction using patellar tendon allograft: An age-dependent outcome evaluation. Arthroscopy, 2010, 26: 488–493
Noyes F R, Barber-Westin S D. Anterior cruciate ligament reconstruction with autogenous patellar tendon graft in patients with articular cartilage damage. Am J Sports Med, 1997, 25: 626–634
Arnoczky S P, Warren R F, Ashlock M A. Replacement of the anterior cruciate ligament using a patellar tendon allograft: An experimental study. J Bone Joint Surg Am, 1986, 68: 376–385
Adriani E, Mariani P P, Maresca G, et al. Healing of the patellar tendon after harvesting of its mid-third for anterior cruciate ligament reconstruction and evolution of the unclosed donor site defect. Knee Surg Sports Traumatol Arthrosc, 1995, 3: 138–143
Marumoto J M, Mitsunaga M M, Richardson A B, et al. Late patellar tendon ruptures after removal of the central third for anterior cruciate ligament reconstruction: A report of two cases. Am J Sports Med, 1996, 24: 698–701
Schippinger G, Passler J M, Seibert F J, et al. Are complications in cruciate ligament replacement operations with patellar tendon transplantation dependent on surgical technique and surgical timing? Swiss Surg, 1997, 3: 154–159
Kamps B S, Linder L H, DeCamp C E, et al. The influence of immobilization versus exercise on scar formation in the rabbit patellar tendon after excision of the central third. Am J Sports Med, 1994, 22: 803–811
Chan B P, Chan K M, Maffulli N, et al. Effect of basic fibroblast growth factor: An in vitro study of tendon healing. Clin Orthop Relat Res, 1997, 342: 239–247
Mosmann T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J Immunol Methods, 1983, 65: 55–63
Cook H, Davies K J, Harding K G, et al. Defective extracellular matrix reorganization by chronic wound fibroblasts is associated with alterations in TIMP-1, TIMP-2 and MMP-2 activity. J Invest Dermatol, 2000, 115: 225–233
Webb S E, Lee K K, Tang M K, et al. Fibroblast growth factors 2 and 4 stimulate migration of mouse embryonic limb myogenic cells. Dev Dyn, 1997, 209: 206–216
Celeste A J, Iannazzi J A, Taylor R C, et al. Identification of transforming growth factor beta family members present in bone-inductive protein purified from bovine bone. Proc Natl Acad Sci USA, 1990, 87: 9843–9847
Tachi K, Takami M, Sato H, et al. Enhancement of bone morphogenetic protein-2-induced ectopic bone formation by transforming growth factor-β1. Tissue Eng Part A, 2011, 17: 597–606
Kingsley D M. The TGF-beta superfamily: New members, new receptors, and new genetic tests of function in different organisms. Genes Dev, 1994, 8: 133–146
Wozney J M, Rosen V, Byrne M, et al. Growth factors influencing bone development. J Cell Sci Suppl, 1990, 13: 149–156
Blader P, Rastegar S, Fischer N, et al. Cleavage of the BMP-4 antagonist chordin by zebrafish tolloid. Science, 1997, 278: 1937–1940
Croteau S, Rauch F, Silvestri A, et al. Bone morphogenetic proteins in orthopedics: From basic science to clinical practice. Orthopedics, 1999, 22: 686–695, quiz 696–697
Chen D, Harris M A, Rossini G, et al. Bone morphogenetic protein 2 (BMP-2) enhances BMP-3, BMP-4, and bone cell differentiation marker gene expression during the induction of mineralized bone matrix formation in cultures of fetal rat calvarial osteoblasts. Calcif Tissue Int, 1997, 60: 283–290
De Luca F, Barnes K M, Uyeda J A, et al. Regulation of growth plate chondrogenesis by bone morphogenetic protein-2. Endocrinology, 2001, 142: 430–436
Duprez D, Bell E J, Richardson M K, et al. Overexpression of BMP-2 and BMP-4 alters the size and shape of developing skeletal elements in the chick limb. Mech Dev, 1996, 57: 145–157
Katagiri T, Yamaguchi A, Komaki M, et al. Bone morphogenetic protein-2 converts the differentiation pathway of C2C12 myoblasts into the osteoblast lineage. J Cell Biol, 1994, 127: 1755–1766
Katagiri T, Akiyama S, Namiki M, et al. Bone morphogenetic protein-2 inhibits terminal differentiation of myogenic cells by suppressing the transcriptional activity of MyoD and myogenin. Exp Cell Res, 1997, 230: 342–351
Kingsley D M, Bland A E, Grubber J M, et al. The mouse short ear skeletal morphogenesis locus is associated with defects in a bone morphogenetic member of the TGF beta superfamily. Cell, 1992, 71: 399–410
Kingsley D M. What do BMPs do in mammals? Clues from the mouse short-ear mutation. Trends Genet, 1994, 10: 16–21
Hay E, Lemonnier J, Fromigue O, et al. Bone morphogenetic protein-2 promotes osteoblast apoptosis through a Smad-independent, protein kinase C-dependent signaling pathway. J Biol Chem, 2001, 276: 29028–29036
Zhang H, Bradley A. Mice deficient for BMP2 are nonviable and have defects in amnion/chorion and cardiac development. Development, 1996, 122: 2977–2986
Chang H, Huylebroeck D, Verschueren K, et al. Smad5 knockout mice die at mid-gestation due to multiple embryonic and extraembryonic defects. Development, 1999, 126: 1631–1642
Yang X, Castilla L H, Xu X, et al. Angiogenesis defects and mesenchymal apoptosis in mice lacking SMAD5. Development, 1999, 126: 1571–1580
Wang E A, Rosen V, D’Alessandro J S, et al. Recombinant human bone morphogenetic protein induces bone formation. Proc Natl Acad Sci USA, 1990, 87: 2220–2224
Martinek V, Latterman C, Usas A, et al. Enhancement of tendonbone integration of anterior cruciate ligament grafts with bone morphogenetic protein-2 gene transfer: A histological and biomechanical study. J Bone Joint Surg Am, 2002, 84-A: 1123–1131
Bi Y, Ehirchiou D, Kilts T M, et al. Identification of tendon stem/progenitor cells and the role of the extracellular matrix in their niche. Nat Med, 2007, 13: 1219–1227
Rui Y F, Lui P P, Ni M, et al. Mechanical loading increased BMP-2 expression which promoted osteogenic differentiation of tendonderived stem cells. J Orthop Res, 2011, 29: 390–396
Lee K K, Cai D Q, Tang M K, et al. Growth arrest-specific 2 gene expression during patellar tendon healing. Cells Tissues Organs, 2003, 173: 138–146
Ryoo H M, Lee M H, Kim Y J. Critical molecular switches involved in BMP-2-induced osteogenic differentiation of mesenchymal cells. Gene, 2006, 366: 51–57
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Li, Z., Shen, X., Cao, L. et al. Bone morphogenetic protein 2 improves patellar tendon healing by promoting migration and proliferation of tenocytes. Chin. Sci. Bull. 56, 1361–1369 (2011). https://doi.org/10.1007/s11434-011-4438-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-011-4438-7