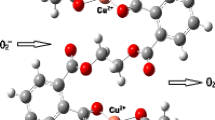
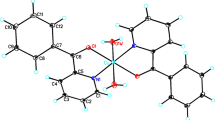
Abstract
Two new transition metal (Cu, Ni) complexes with amino-Schiff base ligand, (C9H7NO3)Cu(C14H12N2)·H2O (1) and (C9H7NO3)Ni(C3H4N2)3·H2O (2), have been designed and synthesized in ethanol solution at room temperature. Both of the complexes have been characterized by elemental analysis, IR spectra, UV-vis spectroscopy and X-ray single crystal diffraction. For complex 1, the coordination environment of the central copper atom is a distorted square pyramid, and one-dimensional chain is formed through the inter-molecular hydrogen bonds (O4-H2W⋯O3, O4-H2W⋯O3#1 (#1: −x+1, y, −z+3/2)) and weak interactions (π-π stacking interaction) between the phenyl rings. For complex 2, the nickel atom is 6-coordinated and in a distorted octahedral environment, and a discrete hydrogen-bond cluster (four molecules are connected by hydrogen bonds into a group) is formed via two types of intra-molecular hydrogen bonds (O-H⋯O, N-H⋯O) and inter-molecular hydrogen bonds (O-H⋯O, N-H⋯O).
Similar content being viewed by others
References
Michacl R, Wagner F, Walker A, et al. Spectroscopic of 1:1 copper (II) complexes with schiff base ligands derived from salicylablehrde and L-histidine and its analogues. Inorg Chem, 1983, 22: 3021–3028
Xiong Y, Li D F, Zheng X F, et al. Synthesis, characterization and superoxide dismutase activity of some SOD model complexes. J Central China Nomal Univ (Natl Sci Ed), 1997, 31(2): 179–181
Koh L L, Ranford J O, Robinson W T, et al. Model for the reduced schiff Base intermediate between amino acids and pyridoxal: Copper( II) complexes of N-(2-Hydroxybenzyl)amino acids with nonpolar side chains and the crystal structures of [Cu(N-(2-hydroxybenzyl)-D,L-alanine)(phen)]·H2O and Cu(N-(2-hydroxybenzyl)-D,L-alanine) (imidazole)]. Inorg Chem, 1996, 35(22): 6466–6472
Das S, Pal S. Copper(II) complexes with tridentate N-(benzoyl)-N′-(salicylidine)-hydrazine and monodentate N-heterocycles: Investigations of intermolecular interactions in the solid state. J Mol Structure, 2005, 753(1–3): 68–79
Mukhopadhyay A, Padmaja G, Pal S. Square-planar nickel(II) complexes with a tridentate Schiff base and monodentate heterocycles: self-assembly to dimeric and one-dimensional array via hydrogen bonding. Inorg Chem Commun, 2003, 6(4): 381–386
Deshmukh M M, Bartolotti L J, Gadre S R. Intramolecular hydrogen bonding and cooperative interactions in carbohydrates via the molecular tailoring approach. J Phys Chem, A, 2008, 112(2): 312–321
Baxter P N W, Lehn J-M, Kneisel B O, et al. Multicomponent selfassembly: Preferential generation of a rectangular [2 × 3]G grid by mixed-ligand recognition. Angew Chem Int Ed Engl, 1997, 36(18): 1978–1981
Braga D, Grepioni F, Desiraju G R. The molecular mechanism of autoxidation for myoglobin and hemoglobin: A venerable puzzle. Chem Rev, 1998, 98(4): 1357–1374
Sangeetha N R, Pal N, Anson C E, et al. A one-dimensional assembly of copper(II) polyhedra via dual use of hydrogen-bonding and π-π interaction. Inorg Chem Commun, 2000, 3(8): 415–419
Zhang J P, Kitagawa S. Supramolecular isomerism, framework flexibility, unsaturated metal center, and porous property of Ag(I)/Cu(I) 3,3′S,5,5′-tetrametyl-4,4′-bipyrazolate. J Am Chem Soc, 2008, 130(3): 907–917
Gadad A K, Noolvi M N, Karpoormath R V. Synthesis and anti-tubercular activity of a series of 2-sulfonamido/trifluoromethyl-6-substituted imidazo[2,1-b]-1,3,4-thiadiazole derivatives, Bioorg Med Chem, 2004, 12(21): 5651–5659
Shin K J, Koo K D, Yoo K H, et al. Synthesis and biological properties of new 1β-methylcarbapenems containing heteroaromatic thioether moiety. Bioorg Med Chem Lett, 2001, 11 (17): 2397–2399
Abrahams B F, Batten S R, Hamit H, et al. A wellsian “three-dimensional” racemate: eight interpenetrating, enantiomorphic (10,3)-a nets, four right- and four left-handed. J Chem Soc, Chem Commun, 1996, 11: 1313–1314
Piguet C, Bernardinelli G, Hopfgartner G. Helicates as versatile supramolecular complexes. Chem Rev, 1997, 97(6): 2005–2062
Aspinall H C, Bickley J F, Dwyer J L M, et al. Lithium binaphtholates: chiral chains and clusters. Angew Chem Int Ed, 2000, 39(16): 2858–2861
Rowan A E, Nolte R J M. Molecular programming of helicity. Angew Chem Int Ed, 1998, 110: 65–71
Lawrence D S, Jiang T, Levett M, Self-assembling supramolecular complexes. Chem Rev, 1995, 95(6): 2229–2260
Panchal P K, Patel M N. Synthesis, structural characterization, and antibacterial studies of some mixed-ligand first row d-transition metal complexes. Synth React Inorg Met-Org Chem, 2004, 34(7): 1277–1289.
Addison A W, Rao T N, Reedijik J, et al. Synthesis, structure, and spectroscopic properties of copper(II) compounds containing nitrogen-sulphur donor ligands; the crystal and molecular structure of aqua[1,7-bis(N-methylbenzimidazol-2′-yl)-2,6-dithiaheptane]copper (II) perchlorate. J Chem Soc, Dalton Trans, 1984, 7: 1349–1356
Xing Y H, Han J, Zhou G H, et al. Syntheses and structures of mononuclear and binuclear transition metal complexes (Cu, Zn, Ni) with salicylideneglycine and imidazole). J Coord Chem, 2008, 61(5): 715–730
Marklund S, Marklund G. Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem, 1974, 47(3): 469–474
Bhirud G R, Srivastava T S. Synthesis, characterization and superoxide dismutase activity of some ternary copper(II) dipeptide-2,2′-bipyridine, 1,10-phenanthroline and 2,9-dimethyl-1,10-phenanthroline complexes. Inorg Chim Acta, 1991, 179(1–2): 125–131
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China (Grant Nos. 20771051 and 20633050), Education Foundation of Liaoning Province (Grant No. 05L212) and SRF for ROCS, SEM
About this article
Cite this article
Han, J., Bai, F., Zhao, H. et al. Synthesis, structure and superoxide dismutase activity of two self-assembly transition metal complexes containing a tridentate amino-Schiff base deviating from salicylaldehyde with glycine. Chin. Sci. Bull. 54, 3508–3514 (2009). https://doi.org/10.1007/s11434-009-0449-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-009-0449-z