Abstract
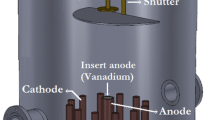
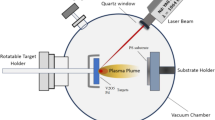
Novel nano-structured films of V2O5 are prepared by pulsed laser deposition method. Nanoscaled V2O5 ridges lie on SrTiO3 substrate and construct into grid-textured structures. Structural properties of the films have been analyzed by scanning electron microscope, X-ray diffraction and transmission electron microscope. The films have enlarged surface-to-volume ratio due to the ridge-channel structure which makes them applicable to gas sensing. Therefore, gas sensors based on the V2O5 films have been assembled which present reliable sensing properties to gaseous acetone, and ethanol at room temperature. The physical-chemical reactions between adsorbed O2– and testing gases are the possible reason for this property.
Similar content being viewed by others
References
Hans M, Jos P H B, Udo D, et al. Progress of the EUVL alpha tool. In: SPIE Symposium on Emerging Lithographic Technologies V. Volume 4343. Santa Clara, 2001. 38–51
Hans M, Vadim B, Noreen H, et al. Development of the ASML EUV alpha demo tool. In: Proceeding SPIE on Emerging Lithographic Technologies IX. Volume 5751. San Jose, California, 2005. 90–101
Wu Y N, Jiang T, Shi T L, et al. Au modified ZnO nanowires for ethanol gas sensing. Sci China Tech Sci, 2017, 60: 71–77
Navale Y H, Navale S T, Ramgir N S, et al. Zinc oxide hierarchical nanostructures as potential NO2 sensors. Sensor Actuat B-Chem, 2017, 251: 551–563
Shan H, Liu C B, Liu L, et al. Synthesis and acetone gas sensing properties of α-Fe2O3 nanotubes. Sci China Chem, 2013, 56: 1722–1726
Zeng W, Liu T, Wang Z. Enhanced gas sensing properties by SnO2 nanosphere functionalized TiO2 nanobelts. J Mater Chem, 2012, 22: 3544–3548
Kaur N, Comini E, Zappa D, et al. Nickel oxide nanowires: Vapor liquid solid synthesis and integration into a gas sensing device. Nanotechnology, 2016, 27: 205701
Guan L, Pang H, Wang J, et al. Fabrication of novel comb-like Cu2O nanorod-based structures through an interface etching method and their application as ethanol sensors. Chem Commun, 2010, 46: 7022–7024
Liu B, Yang H, Zhao H, et al. Synthesis and enhanced gas-sensing properties of ultralong NiO nanowires assembled with NiO nanocrystals. Sensor Actuat B-Chem, 2011, 156: 251–262
Kolmakov A, Moskovits M. Chemical sensing and catalysis by onedimensional metal-oxide nanostructures. Annu Rev Mater Res, 2004, 34: 151–180
Comini E. Metal oxide nano-crystals for gas sensing. Anal Chim Acta, 2006, 568: 28–40
Liu Y, Dong J, Hesketh P J, et al. Synthesis and gas sensing properties of ZnO single crystal flakes. J Mater Chem, 2005, 15: 2316–2320
Schierbaum K D, Weimar U, Göpel W. Comparison of ceramic, thickfilm and thin-film chemical sensors based upon SnO2. Sensor Actuat B-Chem, 1992, 7: 709–716
Rao B B. Zinc oxide ceramic semi-conductor gas sensor for ethanol vapour. Mater Chem Phys, 2000, 64: 62–65
Mosset A, Lecante P, Galy J, et al. Structural analysis of amorphous V2O5 by large-angle X-ray scattering. Philos Mag B, 1982, 46: 137–149
Dziembaj R. Oxygen equilibrium pressure above V2O5−x and thermodynamic properties of this oxide system. J Solid State Chem, 1978, 26: 159–165
Meyer J, Zilberberg K, Riedl T, et al. Electronic structure of vanadium pentoxide: An efficient hole injector for organic electronic materials. J Appl Phys, 2011, 110: 033710
Liu J, Wang X, Peng Q, et al. Vanadium pentoxide nanobelts: Highly selective and stable ethanol sensor materials. Adv Mater, 2005, 17: 764–767
Isabelle R, Marko B, Ulrich S, et al. V2O5 nanofibres: Novel gas sensors with extremely high sensitivity and selectivity to amines. Sensor Actuat B-Chem, 2005, 106: 730–735
Yu H Y, Kang B H, Pi U H, et al. V2O5 nanowire-based nanoelectronic devices for helium detection. Appl Phys Lett, 2005, 86: 253102
Demeter M, Neumann M, Reichelt W. Mixed-valence vanadium oxides studied by XPS. Surf Sci, 2000, 454–456: 41–44
Sawatzky G A, Post D. X-ray photoelectron and Auger spectroscopy study of some vanadium oxides. Phys Rev B, 1979, 20: 1546–1555
Silversmit G, Depla D, Poelman H, et al. Determination of the V2p XPS binding energies for different vanadium oxidation states (V5+ to V0+). J Electron Spectr Related Phenomena, 2004, 135: 167–175
Hryha E, Rutqvist E, Nyborg L. Stoichiometric vanadium oxides studied by XPS. Surf Interface Anal, 2012, 44: 1022–1025
Chen Y J, Xue X Y, Wang Y G, et al. Synthesis and ethanol sensing characteristics of single crystalline SnO2 nanorods. Appl Phys Lett, 2005, 87: 233503
Liu Y, Koep E, Liu M. A highly sensitive and fast-responding SnO2 sensor fabricated by combustion chemical vapor deposition. Chem Mater, 2005, 17: 3997–4000
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by the National Key Project for Basic Research (Grant No. 2014CB921002), the National Natural Science Foundation of China (Grant No. 11374225), and the Research Grant Council of Hong Kong (Grant No. 702112).
Rights and permissions
About this article
Cite this article
Cheng, R., Huang, W., Liu, X. et al. The preparation of grid-textured V2O5 films and their potential applications in gas sensing. Sci. China Technol. Sci. 63, 467–471 (2020). https://doi.org/10.1007/s11431-018-9474-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11431-018-9474-6