Abstract
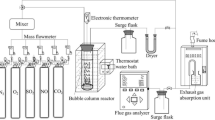
Performance of simultaneous desulfurization and dinitration using the solution of NaClO2 and NaClO as new-style complex absorbent was investigated experimentally in self-designed bench scale bubbling reactor. The effects of main parameters, such as the concentrations of NaClO2 and of NaClO, solution pH and reaction temperature and so on, on removal efficiencies of SO2 and NOx, were examined, then the optimal conditions were established, in which the molar ratio of NaClO to NaClO2 was 1:1, the reaction temperature was 50°C and the solution pH was 5.5. The removal efficiencies of SO2 and NO under the optimal conditions were 100% and 89.2%, respectively. The mechanism of simultaneous removal based on complex absorbent was proposed by analyzing the removal products and the electrode potentials of related species, namely SO2 and NO are oxidized by chlorite anion, hypochlorite, chlorine dioxide and chlorine contained in complex absorbent. In thermodynamic aspect, simultaneous desulfurization and denitration reactions in liquid phase can happen spontaneously and completely, and are all exothermic reactions. It was confirmed by kinetics that for simultaneous desulfurization and dinitration, the reaction order and average activation energy of SO2 were 1 and 21.6 kJ·mol−1, respectively, and those of NO were 1 and 8.2 kJ·mol−1, respectively.
Similar content being viewed by others
References
Wang Q, Hu J J, Zou P. Researches about NaClO2 wet flue gas desulfurization and denitration technology (in Chinese). Jiangxi Electr Pow, 2004, 28: 14–16
Teramoto M, Ikeda M, Teranishi H. Absorption rates of NO in mixed aqueous solutions of NaClO2 and NaOH. Kagaku Kogaku Ronbunshu, 1976, 2: 637–640
Sada E, Kumazawa H, Kudo I, et al. Absorption of NO in aqueous mixed solution of NaClO2 and NaOH. Chem Eng Sci, 1978, 33: 315–318
Sada E, Kumazawa H, Yamanake H, et al. Kinetics of absorption of sulfur dioxide and nitric oxide in aqueous mixed solutions of sodium chlorite and sodium hydroxide. J Chem Eng Jpn, 1978, 11: 276–282
Sada E, Kumazawa H, Kudo I, et al. Absorption of lean NOx in aqueous solutions of NaClO2 and NaOH. Ind Eng Chem Process Des Dev, 1979, 18: 275–278
Brogren C, Karlsson H, Bjerle I. Absorption of NO in an aqueous solution of NaClO2. Chem Eng Technol, 1999, 21: 61–70
Hsu H W, Lee C J, Chou K S. Absorption of NO by NaClO2 solution: Performance characteristics. Chem Eng Commun, 1998, 170: 67–81
Chien T W, Chu H. Removal of SO2 and NO from flue gas by wet scrubbing using an aqueous NaClO2 solution. J Hazard Mater, 2000, 80: 43–57
Chu H, Chien T W, Twu B W. The absorption kinetics of NO in NaClO2/NaOH solutions. J Hazard Mater, 2001, 84: 241–252
Chien T W, Chu H, Hsueh H T. Kinetic study on absorption of SO2 and NOx with acidic NaClO2 solutions using the spraying column. J Environ Eng, 2003, 129: 967–974
Adewuyi Y G, He X D, Shaw H, et al. Simultaneous absorption and oxidation of NO and SO2 by aqueous solutions of sodium chlorite. Chem Eng Commun, 1999, 174: 21–51
Zhao Y, Liu F, Guo T X, et al. Experiments and reaction characteristics of liquid phase simultaneous removal of SO2 and NO. Sci China Ser E-Tech Sci, 2009, 52: 1768–1775
Lehtimaa T, Tarvo V, Mortha G, et al. Reactions and kinetics of Cl(III) decomposition. Ind Eng Chem Res, 2008, 47: 5284–5290
Kormanyos B, Nagypal I, Peintler G, et al. Effect of chloride ion on the kinetics and mechanism of the reaction between chlorite ion and hypochlorous acid. Inorg Chem, 2008, 47: 7914–7920
Deshwal B R, Jin D S, Lee S H, et al. Removal of NO from flue gas by aqueous chlorine-dioxide scrubbing solution in a lab-scale bubbling reactor. J Hazard Mater, 2008, 150: 649–655
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, Y., Guo, T., Liu, F. et al. Performance of simultaneous desulfurization and denitration in liquid phase with new-style complex absorbent. Sci. China Technol. Sci. 54, 3009–3016 (2011). https://doi.org/10.1007/s11431-011-4529-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11431-011-4529-3