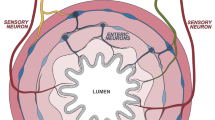
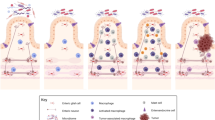
Abstract
The gut is the largest digestive and absorptive organ, which is essential for induction of mucosal and systemic immune responses, and maintenance of metabolic-immune homeostasis. The intestinal components contain the epithelium, stromal cells, immune cells, and enteric nervous system (ENS), as well as the outers, such as gut microbiota, metabolites, and nutrients. The dyshomeostasis of intestinal microenvironment induces abnormal intestinal development and functions, even colon diseases including dysplasia, inflammation and tumor. Several recent studies have identified that ENS plays a crucial role in maintaining the immune homeostasis of gastrointestinal (GI) microenvironment. The crosstalk between ENS and immune cells, mainly macrophages, T cells, and innate lymphoid cells (ILCs), has been found to exert important regulatory roles in intestinal tissue programming, homeostasis, function, and inflammation. In this review, we mainly summarize the critical roles of the interactions between ENS and immune cells in intestinal homeostasis during intestinal development and diseases progression, to provide theoretical bases and ideas for the exploration of immunotherapy for gastrointestinal diseases with the ENS as potential novel targets.
Similar content being viewed by others
References
Agirman, G., Yu, K.B., and Hsiao, E.Y. (2021). Signaling inflammation across the gut-brain axis. Science 374, 1087–1092.
Ahrends, T., Aydin, B., Matheis, F., Classon, C.H., Marchildon, F., Furtado, G.C., Lira, S.A., and Mucida, D. (2021). Enteric pathogens induce tissue tolerance and prevent neuronal loss from subsequent infections. Cell 184, 5715–5727.e12.
Amadei, G., Handford, C.E., Qiu, C., De Jonghe, J., Greenfeld, H., Tran, M., Martin, B.K., Chen, D.Y., Aguilera-Castrejon, A., Hanna, J.H., et al. (2022). Embryo model completes gastrulation to neurulation and organogenesis. Nature 610, 143–153.
Artis, D., and Spits, H. (2015). The biology of innate lymphoid cells. Nature 517, 293–301.
Baghdadi, M.B., Ayyaz, A., Coquenlorge, S., Chu, B., Kumar, S., Streutker, C., Wrana, J.L., and Kim, T.H. (2022). Enteric glial cell heterogeneity regulates intestinal stem cell niches. Cell Stem Cell 29, 86–100.e6.
Barnhoorn, M.C., Hakuno, S.K., Bruckner, R.S., Rogler, G., Hawinkels, L. J.A.C., and Scharl, M. (2020). Stromal cells in the pathogenesis of inflammatory bowel disease. J Crohns Colitis 14, 995–1009.
Bauché, D., Joyce-Shaikh, B., Jain, R., Grein, J., Ku, K.S., Blumenschein, W.M., Ganal-Vonarburg, S.C., Wilson, D.C., McClanahan, T.K., Malefyt, R.W., et al. (2018). LAG3+ regulatory T cells restrain interleukin-23-producing CX3CR1+ gut-resident macrophages during group 3 innate lymphoid cell-driven colitis. Immunity 49, 342–352.e5.
Bayrer, J.R., Castro, J., Venkataraman, A., Touhara, K.K., Rossen, N.D., Morrie, R.D., Maddern, J., Hendry, A., Braverman, K.N., Garcia-Caraballo, S., et al. (2023). Gut enterochromaffin cells drive visceral pain and anxiety. Nature 616, 137–142.
Becker, L., Nguyen, L., Gill, J., Kulkarni, S., Pasricha, P.J., and Habtezion, A. (2018). Age-dependent shift in macrophage polarisation causes inflammation-mediated degeneration of enteric nervous system. Gut 67, 827–836.
Cai, Y., Song, W., Li, J., Jing, Y., Liang, C., Zhang, L., Zhang, X., Zhang, W., Liu, B., An, Y., et al. (2022). The landscape of aging. Sci China Life Sci 65, 2354–2454.
Caruso, R., Lo, B.C., and Núñez, G. (2020). Host-microbiota interactions in inflammatory bowel disease. Nat Rev Immunol 20, 411–426.
Castro-Dopico, T., Fleming, A., Dennison, T.W., Ferdinand, J.R., Harcourt, K., Stewart, B.J., Cader, Z., Tuong, Z.K., Jing, C., Lok, L.S.C., et al. (2020). GM-CSF calibrates macrophage defense and wound healing programs during intestinal infection and inflammation. Cell Rep 32, 107857.
Chakarov, S., Lim, H.Y., Tan, L., Lim, S.Y., See, P., Lum, J., Zhang, X.M., Foo, S., Nakamizo, S., Duan, K., et al. (2019). Two distinct interstitial macrophage populations coexist across tissues in specific subtissular niches. Science 363.
Chalazonitis, A., and Rao, M. (2018). Enteric nervous system manifestations of neurodegenerative disease. Brain Res 1693, 207–213.
Chang, A.Y., Skirbekk, V.F., Tyrovolas, S., Kassebaum, N.J., and Dieleman, J.L. (2019). Measuring population ageing: an analysis of the Global Burden of Disease Study 2017. Lancet Public Health 4, e159–e167.
Chapman, N.M., Boothby, M.R., and Chi, H. (2020). Metabolic coordination of T cell quiescence and activation. Nat Rev Immunol 20, 55–70.
Cheng, S.X. (2012). Calcium-sensing receptor inhibits secretagogue-induced electrolyte secretion by intestine via the enteric nervous system. Am J Physiol Gastrointest Liver Physiol 303, G60–G70.
Chow, A.K., Grubišić, V., and Gulbransen, B.D. (2021). Enteric glia regulate lymphocyte activation via autophagy-mediated MHC-II expression. Cell Mol Gastroenterol Hepatol 12, 1215–1237.
Čiháková, D., Eaton, W.W., Talor, M.V., Harkus, U.H., Demyanovich, H., Rodriguez, K., Feldman, S., and Kelly, D.L. (2019). Gut permeability and mimicry of the Glutamate Ionotropic Receptor NMDA type Subunit Associated with protein 1 (GRINA) as potential mechanisms related to a subgroup of people with schizophrenia with elevated antigliadin antibodies (AGA IgG). Schizophrenia Res 208, 414–419.
Clevers, H. (2013). The intestinal crypt, a prototype stem cell compartment. Cell 154, 274–284.
Consortium, A.B., Bao, H., Cao, J., Chen, M., Chen, M., Chen, W., Chen, X., Chen, Y., Chen, Y., Chen, Y., et al. (2023). Biomarkers of aging. Sci China Life Sci 66, 893–1066.
Cornet, A., Savidge, T.C., Cabarrocas, J., Deng, W.L., Colombel, J.F., Lassmann, H., Desreumaux, P., and Liblau, R.S. (2001). Enterocolitis induced by autoimmune targeting of enteric glial cells: a possible mechanism in Crohn’s disease?. Proc Natl Acad Sci USA 98, 13306–13311.
Darmanis, S., Sloan, S.A., Zhang, Y., Enge, M., Caneda, C., Shuer, L.M., Hayden Gephart, M.G., Barres, B.A., and Quake, S.R. (2015). A survey of human brain transcriptome diversity at the single cell level. Proc Natl Acad Sci USA 112, 7285–7290.
Davies, C., Mishra, D., Eshraghi, R.S., Mittal, J., Sinha, R., Bulut, E., Mittal, R., and Eshraghi, A.A. (2021). Altering the gut microbiome to potentially modulate behavioral manifestations in autism spectrum disorders: a systematic review. Neurosci Biobehav Rev 128, 549–557.
De Schepper, S., Verheijden, S., Aguilera-Lizarraga, J., Viola, M.F., Boesmans, W., Stakenborg, N., Voytyuk, I., Schmidt, I., Boeckx, B., Dierckx de Casterlé, I., et al. (2018). Self-maintaining gut macrophages are essential for intestinal homeostasis. Cell 175, 400–415.e13.
Dogra, N., Mani, R.J., and Katare, D.P. (2022). The gut-brain axis: two ways signaling in Parkinson’s disease. Cell Mol Neurobiol 42, 315–332.
Drokhlyansky, E., Smillie, C.S., Van Wittenberghe, N., Ericsson, M., Griffin, G.K., Eraslan, G., Dionne, D., Cuoco, M.S., Goder-Reiser, M. N., Sharova, T., et al. (2020). The human and mouse enteric nervous system at single-cell resolution. Cell 182, 1606–1622.e23.
El-Salhy, M., Solomon, T., Hausken, T., Gilja, O.H., and Hatlebakk, J.G. (2017). Gastrointestinal neuroendocrine peptides/amines in inflammatory bowel disease. World J Gastroenterol 23, 5068–5085.
Fang, P., Kazmi, S.A., Jameson, K.G., and Hsiao, E.Y. (2020). The microbiome as a modifier of neurodegenerative disease risk. Cell Host Microbe 28, 201–222.
Fleming, M.A., Ehsan, L., Moore, S.R., and Levin, D.E. (2020). The enteric nervous system and its emerging role as a therapeutic target. Gastroenterol Res Pract 2020, 1–13.
Furness, J. B. (2012). The enteric nervous system and neurogastroenterology. Nat Rev Gastroenterol Hepatol 9, 286–294.
Gabanyi, I., Muller, P.A., Feighery, L., Oliveira, T.Y., Costa-Pinto, F.A., and Mucida, D. (2016). Neuro-immune interactions drive tissue programming in intestinal macrophages. Cell 164, 378–391.
Geremia, A., Biancheri, P., Allan, P., Corazza, G.R., and Di Sabatino, A. (2014). Innate and adaptive immunity in inflammatory bowel disease. Autoimmun Rev 13, 3–10.
Goode, T., O’Connor, T., Hopkins, A., Moriarty, D., O’Sullivan, G.C., Collins, J.K., O’Donoghue, D., Baird, A.W., O’Connell, J., and Shanahan, F. (2003). Neurokinin-1 receptor (NK-1R) expression is induced in human colonic epithelial cells by proinflammatory cytokines and mediates proliferation in response to substance P. J Cell Physiol 197, 30–41.
Goyal, R.K., and Hirano, I. (1996). The enteric nervous system. N Engl J Med 334, 1106–1115.
Graham, K.D., López, S.H., Sengupta, R., Shenoy, A., Schneider, S., Wright, C.M., Feldman, M., Furth, E., Valdivieso, F., Lemke, A., et al. (2020). Robust, 3-dimensional visualization of human colon enteric nervous system without tissue sectioning. Gastroenterology 158, 2221–2235.e5.
Gross, E.R., Gershon, M.D., Margolis, K.G., Gertsberg, Z.V., and Cowles, R.A. (2012). Neuronal serotonin regulates growth of the intestinal mucosa in mice. Gastroenterology 143, 408–417.e2.
Guan, X., Karpen, H.E., Stephens, J., Bukowski, J.T., Niu, S., Zhang, G., Stoll, B., Finegold, M.J., Holst, J.J., Hadsell, D.L., et al. (2006). GLP-2 receptor localizes to enteric neurons and endocrine cells expressing vasoactive peptides and mediates increased blood flow. Gastroenterology 130, 150–164.
Guerra, D.D., Bok, R., Vyas, V., Orlicky, D.J., Lorca, R.A., and Hurt, K.J. (2019). Akt phosphorylation of neuronal nitric oxide synthase regulates gastrointestinal motility in mouse ileum. Proc Natl Acad Sci USA 116, 17541–17546.
Hanahan, D., and Monje, M. (2023). Cancer hallmarks intersect with neuroscience in the tumor microenvironment. Cancer Cell 41, 573–580.
Hanč, P., Gonzalez, R.J., Mazo, I.B., Wang, Y., Lambert, T., Ortiz, G., Miller, E.W., and von Andrian, U.H. (2023). Multimodal control of dendritic cell functions by nociceptors. Science 379, eabm5658.
Hansen, M.B. (2003). The enteric nervous system II: gastrointestinal functions. Pharmacol Toxicol 92, 249–257.
Hao, M.M., Foong, J.P.P., Bornstein, J.C., Li, Z.L., Vanden Berghe, P., and Boesmans, W. (2016). Enteric nervous system assembly: functional integration within the developing gut. Dev Biol 417, 168–181.
Hayakawa, Y., Sakitani, K., Konishi, M., Asfaha, S., Niikura, R., Tomita, H., Renz, B.W., Tailor, Y., Macchini, M., Middelhoff, M., et al. (2017). Nerve growth factor promotes gastric tumorigenesis through aberrant cholinergic signaling. Cancer Cell 31, 21–34.
Hepworth, M.R., Monticelli, L.A., Fung, T.C., Ziegler, C.G.K., Grunberg, S., Sinha, R., Mantegazza, A.R., Ma, H.L., Crawford, A., Angelosanto, J.M., et al. (2013). Innate lymphoid cells regulate CD4+ T-cell responses to intestinal commensal bacteria. Nature 498, 113–117.
Hoff, S., Zeller, F., von Weyhern, C.W.H., Wegner, M., Schemann, M., Michel, K., and Röhl, A. (2008). Quantitative assessment of glial cells in the human and guinea pig enteric nervous system with an anti-Sox8/ 9/10 antibody. J Comp Neurol 509, 356–371.
Houser, M.C., and Tansey, M.G. (2017). The gut-brain axis: is intestinal inflammation a silent driver of Parkinson’s disease pathogenesis? npj Parkinsons Dis 3, 3.
Ibiza, S., García-Cassani, B., Ribeiro, H., Carvalho, T., Almeida, L., Marques, R., Misic, A.M., Bartow-McKenney, C., Larson, D.M., Pavan, W.J., et al. (2016). Glial-cell-derived neuroregulators control type 3 innate lymphoid cells and gut defence. Nature 535, 440–443.
Inclan-Rico, J.M., Ponessa, J.J., Valero-Pacheco, N., Hernandez, C.M., Sy, C.B., Lemenze, A.D., Beaulieu, A.M., and Siracusa, M.C. (2020). Basophils prime group 2 innate lymphoid cells for neuropeptide-mediated inhibition. Nat Immunol 21, 1181–1193.
Isidro, R.A., and Appleyard, C.B. (2016). Colonic macrophage polarization in homeostasis, inflammation, and cancer. Am J Physiol Gastrointest Liver Physiol 311, G59–G73.
Jarret, A., Jackson, R., Duizer, C., Healy, M.E., Zhao, J., Rone, J.M., Bielecki, P., Sefik, E., Roulis, M., Rice, T., et al. (2020). Enteric nervous system-derived IL-18 orchestrates mucosal barrier immunity. Cell 180, 50–63.e12.
Jiang, T.T., Shao, T.Y., Ang, W.X.G., Kinder, J.M., Turner, L.H., Pham, G., Whitt, J., Alenghat, T., and Way, S.S. (2017). Commensal fungi recapitulate the protective benefits of intestinal bacteria. Cell Host Microbe 22, 809–816.e4.
Ju, W., Zheng, R., Zhang, S., Zeng, H., Sun, K., Wang, S., Chen, R., Li, L., Wei, W., and He, J. (2023). Cancer statistics in Chinese older people, 2022: current burden, time trends, and comparisons with the US, Japan, and the Republic of Korea. Sci China Life Sci 66, 1079–1091.
Kermarrec, L., Durand, T., Neunlist, M., Naveilhan, P., and Neveu, I. (2016). Enteric glial cells have specific immunosuppressive properties. J Neuroimmunol 295–296, 79–83.
Köhler, C., Maes, M., Slyepchenko, A., Berk, M., Solmi, M., Lanctôt, K., and Carvalho, A. (2016). The gut-brain axis, including the microbiome, leaky gut and bacterial translocation: mechanisms and pathophysiological role in Alzheimer’s disease. Curr Pharm Des 22, 6152–6166.
Kritsilis, M., V. Rizou, S., Koutsoudaki, P., Evangelou, K., Gorgoulis, V., and Papadopoulos, D. (2018). Ageing, cellular senescence and neurodegenerative disease. Int J Mol Sci 19, 2937.
Lakhan, S.E., and Kirchgessner, A. (2010). Neuroinflammation in inflammatory bowel disease. J Neuroinflamm 7, 37.
Leonardi, I., Gao, I.H., Lin, W.Y., Allen, M., Li, X.V., Fiers, W.D., De Celie, M.B., Putzel, G.G., Yantiss, R.K., Johncilla, M., et al. (2022). Mucosal fungi promote gut barrier function and social behavior via type 17 immunity. Cell 185, 831–846.e14.
Li, H., Limenitakis, J.P., Greiff, V., Yilmaz, B., Schären, O., Urbaniak, C., Zünd, M., Lawson, M.A.E., Young, I.D., Rupp, S., et al. (2020). Mucosal or systemic microbiota exposures shape the B cell repertoire. Nature 584, 274–278.
Liu, H., Li, L., Hao, Y., Li, J., Liu, Z., Qi, J., Zhang, J., Wu, N., Wu, D., Gao, C., et al. (2023). Identification of two migratory colon ILC2 populations differentially expressing IL-17A and IL-5/IL-13. Sci China Life Sci 66, 67–80.
Liu, Q., Johnson, E.M., Lam, R.K., Wang, Q., Bo Ye, H., Wilson, E.N., Minhas, P.S., Liu, L., Swarovski, M.S., Tran, S., et al. (2019). Peripheral TREM1 responses to brain and intestinal immunogens amplify stroke severity. Nat Immunol 20, 1023–1034.
Ma, H., Tao, W., and Zhu, S. (2019). T lymphocytes in the intestinal mucosa: defense and tolerance. Cell Mol Immunol 16, 216–224.
MacDonald, T.T., Monteleone, I., Fantini, M.C., and Monteleone, G. (2011). Regulation of Homeostasis and Inflammation in the Intestine. Gastroenterology 140, 1768–1775.
Maes, M., Kanchanatawan, B., Sirivichayakul, S., and Carvalho, A.F. (2019). In schizophrenia, increased plasma IgM/IgA responses to gut commensal bacteria are associated with negative symptoms, neurocognitive impairments, and the deficit phenotype. Neurotox Res 35, 684–698.
Matheis, F., Muller, P.A., Graves, C.L., Gabanyi, I., Kerner, Z.J., CostaBorges, D., Ahrends, T., Rosenstiel, P., and Mucida, D. (2020). Adrenergic signaling in muscularis macrophages limits infection-induced neuronal loss. Cell 180, 64–78.e16.
Meroni, E., Stakenborg, N., Viola, M.F., and Boeckxstaens, G.E. (2019). Intestinal macrophages and their interaction with the enteric nervous system in health and inflammatory bowel disease. Acta Physiol 225, e13163.
Mertens, J., Herdy, J.R., Traxler, L., Schafer, S.T., Schlachetzki, J.C.M., Böhnke, L., Reid, D.A., Lee, H., Zangwill, D., Fernandes, D.P., et al. (2021). Age-dependent instability of mature neuronal fate in induced neurons from Alzheimer’s patients. Cell Stem Cell 28, 1533–1548.e6.
Mills, C.D. (2015). Anatomy of a Discovery: M1 and M2 macrophages. Front Immunol 6, 212.
Mizuno, S., Mikami, Y., Kamada, N., Handa, T., Hayashi, A., Sato, T., Matsuoka, K., Matano, M., Ohta, Y., Sugita, A., et al. (2014). Cross-talk between RORγt+ innate lymphoid cells and intestinal macrophages induces mucosal IL-22 production in Crohn’s disease. Inflamm Bowel Dis 20, 1426–1434.
Morarach, K., Mikhailova, A., Knoflach, V., Memic, F., Kumar, R., Li, W., Ernfors, P., and Marklund, U. (2021). Diversification of molecularly defined myenteric neuron classes revealed by single-cell RNA sequencing. Nat Neurosci 24, 34–46.
Mortha, A., Chudnovskiy, A., Hashimoto, D., Bogunovic, M., Spencer, S. P., Belkaid, Y., and Merad, M. (2014). Microbiota-dependent crosstalk between macrophages and ILC3 promotes intestinal homeostasis. Science 343, 1249288.
Mowat, A.M., and Agace, W.W. (2014). Regional specialization within the intestinal immune system. Nat Rev Immunol 14, 667–685.
Muller, P.A., Koscsó, B., Rajani, G.M., Stevanovic, K., Berres, M.L., Hashimoto, D., Mortha, A., Leboeuf, M., Li, X.M., Mucida, D., et al. (2014). Crosstalk between muscularis macrophages and enteric neurons regulates gastrointestinal motility. Cell 158, 300–313.
Neunlist, M., Van Landeghem, L., Mahé, M.M., Derkinderen, P., des Varannes, S.B., and Rolli-Derkinderen, M. (2013). The digestive neuronal-glial-epithelial unit: a new actor in gut health and disease. Nat Rev Gastroenterol Hepatol 10, 90–100.
Nielsen, M.M., Witherden, D.A., and Havran, W.L. (2017). γδ5 T cells in homeostasis and host defence of epithelial barrier tissues. Nat Rev Immunol 17, 733–745.
Nowicki, P.T., Caniano, D.A., Hammond, S., Giannone, P.J., Besner, G.E., Reber, K.M., and Nankervis, C.A. (2007). Endothelial nitric oxide synthase in human intestine resected for necrotizing enterocolitis. J Pediatr 150, 40–45.
Obata, Y., and Pachnis, V. (2016). The effect of microbiota and the immune system on the development and organization of the enteric nervous system. Gastroenterology 151, 836–844.
Obermayr, F., Hotta, R., Enomoto, H., and Young, H.M. (2013). Development and developmental disorders of the enteric nervous system. Nat Rev Gastroenterol Hepatol 10, 43–57.
Okumura, R., and Takeda, K. (2017). Roles of intestinal epithelial cells in the maintenance of gut homeostasis. Exp Mol Med 49, e338.
Ost, K.S., O’Meara, T.R., Stephens, W.Z., Chiaro, T., Zhou, H., Penman, J., Bell, R., Catanzaro, J.R., Song, D., Singh, S., et al. (2021). Adaptive immunity induces mutualism between commensal eukaryotes. Nature 596, 114–118.
Pawolski, V., and Schmidt, M.H.H. (2020). Neuron-glia interaction in the developing and adult enteric nervous system. Cells 10, 47.
Pearson, C., Thornton, E.E., McKenzie, B., Schaupp, A.L., Huskens, N., Griseri, T., West, N., Tung, S., Seddon, B.P., Uhlig, H.H., et al. (2016). ILC3 GM-CSF production and mobilisation orchestrate acute intestinal inflammation. Elife 5, e10066.
Peterson, L.W., and Artis, D. (2014). Intestinal epithelial cells: regulators of barrier function and immune homeostasis. Nat Rev Immunol 14, 141–153.
Pochard, C., Coquenlorge, S., Jaulin, J., Cenac, N., Vergnolle, N., Meurette, G., Freyssinet, M., Neunlist, M., and Rolli-Derkinderen, M. (2016). Defects in 15-HETE production and control of epithelial permeability by human enteric glial cells from patients with Crohn’s disease. Gastroenterology 150, 168–180.
Progatzky, F., and Pachnis, V. (2022). The role of enteric glia in intestinal immunity. Curr Opin Immunol 77, 102183.
Progatzky, F., Shapiro, M., Chng, S.H., Garcia-Cassani, B., Classon, C.H., Sevgi, S., Laddach, A., Bon-Frauches, A.C., Lasrado, R., Rahim, M., et al. (2021). Regulation of intestinal immunity and tissue repair by enteric glia. Nature 599, 125–130.
Renz, H., Brandtzaeg, P., and Hornef, M. (2011). The impact of perinatal immune development on mucosal homeostasis and chronic inflammation. Nat Rev Immunol 12, 9–23.
Ribot, J.C., Lopes, N., and Silva-Santos, B. (2021). γδ T cells in tissue physiology and surveillance. Nat Rev Immunol 21, 221–232.
Round, J.L., and Mazmanian, S.K. (2009). The gut microbiota shapes intestinal immune responses during health and disease. Nat Rev Immunol 9, 313–323.
Saldana-Morales, F.B., Kim, D.V., Tsai, M.T., and Diehl, G.E. (2021). Healthy intestinal function relies on coordinated enteric nervous system, immune system, and epithelium responses. Gut Microbes 13, 1–4.
Schlissel, M. (2013). B-cell development in the gut. Nature 501, 42–43.
Schneider, S., Wright, C.M., and Heuckeroth, R.O. (2019). Unexpected roles for the second brain: enteric nervous system as master regulator of bowel function. Annu Rev Physiol 81, 235–259.
Seillet, C., Luong, K., Tellier, J., Jacquelot, N., Shen, R.D., Hickey, P., Wimmer, V.C., Whitehead, L., Rogers, K., Smyth, G.K., et al. (2020). The neuropeptide VIP confers anticipatory mucosal immunity by regulating ILC3 activity. Nat Immunol 21, 168–177.
Seo, D., O’Donnell, D., Jain, N., Ulrich, J.D., Herz, J., Li, Y., Lemieux, M., Cheng, J., Hu, H., Serrano, J.R., et al. (2023). ApoE isoform- and microbiota-dependent progression of neurodegeneration in a mouse model of tauopathy. Science 379, eadd1236.
Sica, A., and Mantovani, A. (2012). Macrophage plasticity and polarization: in vivo veritas. J Clin Invest 122, 787–795.
Snippert, H.J., and Clevers, H. (2011). Tracking adult stem cells. EMBO Rep 12, 113–122.
Spits, H., Artis, D., Colonna, M., Diefenbach, A., Di Santo, J.P., Eberl, G., Koyasu, S., Locksley, R.M., McKenzie, A.N.J., Mebius, R.E., et al. (2013). Innate lymphoid cells—a proposal for uniform nomenclature. Nat Rev Immunol 13, 145–149.
Straub, R.H., Wiest, R., Strauch, U.G., Harle, P., and Scholmerich, J. (2006). The role of the sympathetic nervous system in intestinal inflammation. Gut 55, 1640–1649.
Sullivan, Z.A., Khoury-Hanold, W., Lim, J., Smillie, C., Biton, M., Reis, B. S., Zwick, R.K., Pope, S.D., Israni-Winger, K., Parsa, R., et al. (2021). γδ T cells regulate the intestinal response to nutrient sensing. Science 371.
Sun, Y., Wang, Q., Wang, Y., Ren, W., Cao, Y., Li, J., Zhou, X., Fu, W., and Yang, J. (2021). Sarm1-mediated neurodegeneration within the enteric nervous system protects against local inflammation of the colon. Protein Cell 12, 621–638.
Talbot, J., Hahn, P., Kroehling, L., Nguyen, H., Li, D., and Littman, D.R. (2020). Feeding-dependent VIP neuron-ILC3 circuit regulates the intestinal barrier. Nature 579, 575–580.
Tarazi, S., Aguilera-Castrejon, A., Joubran, C., Ghanem, N., Ashouokhi, S., Roncato, F., Wildschutz, E., Haddad, M., Oldak, B., Gomez-Cesar, E., et al. (2022). Post-gastrulation synthetic embryos generated ex utero from mouse naive ESCs. Cell 185, 3290–3306.e25.
Toumi, F., Neunlist, M., Cassagnau, E., Parois, S., Laboisse, C.L., Galmiche, J.P., and Jarry, A. (2003). Human submucosal neurones regulate intestinal epithelial cell proliferation: evidence from a novel co-culture model. Neurogastroenterol Motil 15, 239–242.
Turco, F., Sarnelli, G., Cirillo, C., Palumbo, I., De Giorgi, F., D’Alessandro, A., Cammarota, M., Giuliano, M., and Cuomo, R. (2014). Enteroglial-derived S100B protein integrates bacteria-induced Toll-like receptor signalling in human enteric glial cells. Gut 63, 105–115.
Van der Perren, A., Gelders, G., Fenyi, A., Bousset, L., Brito, F., Peelaerts, W., Van den Haute, C., Gentleman, S., Melki, R., and Baekelandt, V. (2020). The structural differences between patient-derived α-synuclein strains dictate characteristics of Parkinson’s disease, multiple system atrophy and dementia with Lewy bodies. Acta Neuropathol 139, 977–1000.
van Furth, R., Cohn, Z.A., Hirsch, J.G., Humphrey, J.H., Spector, W.G., and Langevoort, H.L. (1972). The mononuclear phagocyte system: a new classification of macrophages, monocytes, and their precursor cells. Bull World Health Organ 46, 845–852.
Vivier, E., Artis, D., Colonna, M., Diefenbach, A., Di Santo, J.P., Eberl, G., Koyasu, S., Locksley, R.M., McKenzie, A.N.J., Mebius, R.E., et al. (2018). Innate lymphoid cells: 10 years on. Cell 174, 1054–1066.
Walsh, K.T., and Zemper, A.E. (2019). The enteric nervous system for epithelial researchers: basic anatomy, techniques, and interactions with the epithelium. Cell Mol Gastroenterol Hepatol 8, 369–378.
Wang, J., and Wang, J. (2022). Blood group-gut microbiome—health axis gains further support from landmark multi-omics study in swines. Sci China Life Sci 65, 2338–2340.
Wang, S., Xia, P., Chen, Y., Qu, Y., Xiong, Z., Ye, B., Du, Y., Tian, Y., Yin, Z., Xu, Z., et al. (2017). Regulatory innate lymphoid cells control innate intestinal inflammation. Cell 171, 201–216.e18.
Warner, K., and Ohashi, P.S. (2019). ILC regulation of T cell responses in inflammatory diseases and cancer. Semin Immunol 41, 101284.
Wu, N., Sun, H., Zhao, X., Zhang, Y., Tan, J., Qi, Y., Wang, Q., Ng, M., Liu, Z., He, L., et al. (2021). MAP3K2-regulated intestinal stromal cells define a distinct stem cell niche. Nature 592, 606–610.
Xu, H., Ding, J., Porter, C.B.M., Wallrapp, A., Tabaka, M., Ma, S., Fu, S., Guo, X., Riesenfeld, S.J., Su, C., et al. (2019). Transcriptional atlas of intestinal immune cells reveals that neuropeptide α-CGRP modulates group 2 innate lymphoid cell responses. Immunity 51, 696–708.e9.
Yan, Y., Ramanan, D., Rozenberg, M., McGovern, K., Rastelli, D., Vijaykumar, B., Yaghi, O., Voisin, T., Mosaheb, M., Chiu, I., et al. (2021). Interleukin-6 produced by enteric neurons regulates the number and phenotype of microbe-responsive regulatory T cells in the gut. Immunity 54, 499–513.e5.
Yang, D., Jacobson, A., Meerschaert, K.A., Sifakis, J.J., Wu, M., Chen, X., Yang, T., Zhou, Y., Anekal, P.V., Rucker, R.A., et al. (2022). Nociceptor neurons direct goblet cells via a CGRP-RAMP1 axis to drive mucus production and gut barrier protection. Cell 185, 4190–4205.e25.
Yoo, B.B., and Mazmanian, S.K. (2017). The enteric network: interactions between the immune and nervous systems of the gut. Immunity 46, 910–926.
Zeng, W., Yang, F., Shen, W.L., Zhan, C., Zheng, P., and Hu, J. (2022). Interactions between central nervous system and peripheral metabolic organs. Sci China Life Sci 65, 1929–1958.
Zhang, L., Yu, X., Zheng, L., Zhang, Y., Li, Y., Fang, Q., Gao, R., Kang, B., Zhang, Q., Huang, J.Y., et al. (2018). Lineage tracking reveals dynamic relationships of T cells in colorectal cancer. Nature 564, 268–272.
Zhang, W., Lyu, M., Bessman, N.J., Xie, Z., Arifuzzaman, M., Yano, H., Parkhurst, C.N., Chu, C., Zhou, L., Putzel, G.G., et al. (2022). Gut-innervating nociceptors regulate the intestinal microbiota to promote tissue protection. Cell 185, 4170–4189.e20.
Zhao, S.B., Wu, J.Y., He, Z.X., Song, Y.H., Chang, X., Xia, T., Fang, X., Li, Z.S., Xu, C., Wang, S.L., et al. (2021). Corticotropin releasing hormone promotes inflammatory bowel disease via inducing intestinal macrophage autophagy. Cell Death Discov 7, 377.
Zheng, M., and Zhu, J. (2022). Innate lymphoid cells and intestinal inflammatory disorders. Int J Mol Sci 23, 1856.
Zheng, W., Song, H., Luo, Z., Wu, H., Chen, L., Wang, Y., Cui, H., Zhang, Y., Wang, B., Li, W., et al. (2021). Acetylcholine ameliorates colitis by promoting IL-10 secretion of monocytic myeloid-derived suppressor cells through the nAChR/ERK pathway. Proc Natl Acad Sci USA 118, e2017762118.
Zhou, J., Yang, W., and Liu, Q. (2022a). Cancer challenges worldwide and in China: preparing for the inevitable. Sci China Life Sci 65, 442–444.
Zhou, L., Chong, M.M.W., and Littman, D.R. (2009a). Plasticity of CD4+ T cell lineage differentiation. Immunity 30, 646–655.
Zhou, L., Chu, C., Teng, F., Bessman, N.J., Goc, J., Santosa, E.K., Putzel, G.G., Kabata, H., Kelsen, J.R., Baldassano, R.N., et al. (2019). Innate lymphoid cells support regulatory T cells in the intestine through interleukin-2. Nature 568, 405–409.
Zhou, L., Kong, G., Palmisano, I., Cencioni, M.T., Danzi, M., De Virgiliis, F., Chadwick, J.S., Crawford, G., Yu, Z., De Winter, F., et al. (2022b). Reversible CD8 T cell-neuron cross-talk causes aging-dependent neuronal regenerative decline. Science 376, eabd5926.
Zhou, R., Niwa, S., Homma, N., Takei, Y., and Hirokawa, N. (2009b). KIF26A is an unconventional kinesin and regulates GDNF-Ret signaling in enteric neuronal development. Cell 139, 802–813.
Zhu, G., Hu, J., and Xi, R. (2021). The cellular niche for intestinal stem cells: a team effort. Cell Regen 10, 1.
Zirra, A., and Huxford, B. (2022). CD4+ T cells contribute to neurodegeneration in Lewy body dementia. Mov Disord 37, 268.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (82030042, 32070917, 82111540277, 82202017), the Chongqing International Institute for Immunology (2021YJC01), Ministry of Science and Technology of China (2021 YFA1100800), Shanghai Science and Technology Commission (20JC1417400, 201409005500, 20JC1410100), Shanghai Municipal Health Commission (2022JC001, 2022XD047), and China Postdoctoral Science Foundation (2021M702160). We would like to thank all members of the HB. L laboratory for discussion. We apologize to all scientists whose valuable contributions could not be cited in this review due to limitations of volume.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The author(s) declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Wang, X., Ding, C. & Li, HB. The crosstalk between enteric nervous system and immune system in intestinal development, homeostasis and diseases. Sci. China Life Sci. 67, 41–50 (2024). https://doi.org/10.1007/s11427-023-2376-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-023-2376-0