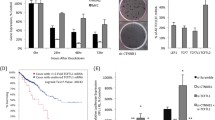
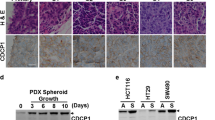
Abstract
The CREB1 gene encodes an exceptionally pleiotropic transcription factor that frequently dysregulated in human cancers. CREB1 can regulate tumor cell status of proliferation and/or migration; however, the molecular basis for this switch involvement in cell plasticity has not fully been understood yet. Here, we first show that knocking out CREB1 triggers a remarkable effect of epithelial-mesenchymal transition (EMT) and leads to the occurrence of inhibited proliferation and enhanced motility in HCT116 colorectal cancer cells. By monitoring 45 cellular signaling pathway activities, we find that multiple growth-related pathways decline significantly while inflammatory pathways including NF-κB are largely upregulated in comparing between the CREB1 wild-type and knocked out cells. Mechanistically, cells with CREB1 knocked out show downregulation of MYC as a result of impaired CREB1-dependent transcription of the oncogenic lncRNA CCAT1. Interestingly, the unbalanced competition between the coactivator CBP/p300 for CREB1 and p65 leads to the activation of the NF-κB pathway in cells with CREB1 disrupted, which induces an obvious EMT phenotype of the cancer cells. Taken together, these studies identify previously unknown mechanisms of CREB1 in CRC cell plasticity via regulating lncRNA CCAT1 and NF-κB pathways, providing a critical insight into a combined strategy for CREB1-targeted tumor therapies.
Similar content being viewed by others
References
Augenlicht, L.H., Davis, R., Lisanti, M.P., Zhurinsky, J., Troussard, A.A., D’Amico, M., Donehower, L.A., Takemaru, K.I., Moon, R.T., BenZe’ev, A., et al. (2000). The integrin-linked kinase regulates the cyclin D1 gene through glycogen synthase kinase 3β and cAMP-responsive element-binding protein-dependent pathways. J Biol Chem 275, 32649–32657.
Berkowitz, L.A., Riabowol, K.T., and Gilman, M.Z. (1989). Multiple sequence elements of a single functional class are required for cyclic AMP responsiveness of the mouse c-fos promoter. Mol Cell Biol 9, 4272–4281.
Boque-Sastre, R., Soler, M., Oliveira-Mateos, C., Portela, A., Moutinho, C., Sayols, S., Villanueva, A., Esteller, M., and Guil, S. (2015). Head-to-head antisense transcription and R-loop formation promotes transcriptional activation. Proc Natl Acad Sci USA 112, 5785–5790.
Boulon, S., Dantonel, J.C., Binet, V., Vie, A., Blanchard, J.M., Hipskind, R.A., and Philips, A. (2002). Oct-1 potentiates CREB-driven cyclin D1 promoter activation via a phospho-CREB- and CREB binding protein-independent mechanism. Mol Cell Biol 22, 7769–7779.
Bulun, S.E., Lin, Z., Zhao, H., Lu, M., Amin, S., Reierstad, S., and Chen, D. (2009). Regulation of aromatase expression in breast cancer tissue. Ann N Y Acad Sci 1155, 121–131.
Carlezon, W.A., Duman, R.S., and Nestler, E.J. (2005). The many faces of CREB. Trends Neuroscis 28, 436–445.
Chhabra, A., Fernando, H., Watkins, G., Mansel, R.E., and Jiang, W.G. (2007). Expression of transcription factor CREB1 in human breast cancer and its correlation with prognosis. Oncol Rep 18, 953–958.
Choi, M.J., Cho, K.H., Lee, S., Bae, Y.J., Jeong, K.J., Rha, S.Y., Choi, E.J., Park, J.H., Kim, J.M., Lee, J.S., et al. (2015). hTERT mediates norepinephrine-induced Slug expression and ovarian cancer aggressiveness. Oncogene 34, 3402–3412.
Chua, H.L., Bhat-Nakshatri, P., Clare, S.E., Morimiya, A., Badve, S., and Nakshatri, H. (2006). NF-κB represses E-cadherin expression and enhances epithelial to mesenchymal transition of mammary epithelial cells: potential involvement of ZEB-1 and ZEB-2. Oncogene 26, 711–724.
Dai, X., Xin, Y., Xu, W., Tian, X., Wei, X., and Zhang, H. (2021). CBP-mediated Slug acetylation stabilizes Slug and promotes EMT and migration of breast cancer cells. Sci China Life Sci 64, 563–574.
Feng, J., Liu, T., Qin, B., Zhang, Y., and Liu, X.S. (2012). Identifying ChIP-seq enrichment using MACS. Nat Protoc 7, 1728–1740.
Fusco, S., Leone, L., Barbati, S.A., Samengo, D., Piacentini, R., Maulucci, G., Toietta, G., Spinelli, M., McBurney, M., Pani, G., et al. (2016). A CREB-Sirt1-Hes1 circuitry mediates neural stem cell response to glucose availability. Cell Rep 14, 1195–1205.
Gao, C.F., Xie, Q., Su, Y.L., Koeman, J., Kean Khoo, S., Gustafson, M., Knudsen, B.S., Hay, R., Shinomiya, N., and Vande Woude, G.F. (2005). Proliferation and invasion: plasticity in tumor cells. Proc Natl Acad Sci USA 102, 10528–10533.
Gerritsen, M.E., Williams, A.J., Neish, A.S., Moore, S., Shi, Y., and Collins, T. (1997). CREB-binding protein/p300 are transcriptional coactivators of p65. Proc Natl Acad Sci USA 94, 2927–2932.
Ghosh, S., Lu, Y., and Hu, Y. (2008). A role of CREB in BRCA1 constitutive promoter activity and aromatase basal expression. Int J Biomed Sci 4, 260–265.
Goldman, M.J., Craft, B., Hastie, M., Repečka, K., McDade, F., Kamath, A., Banerjee, A., Luo, Y., Rogers, D., Brooks, A.N., et al. (2020). Visualizing and interpreting cancer genomics data via the Xena platform. Nat Biotechnol 38, 675–678.
Gonzalez-Mariscal, L., Tapia, R., Huerta, M., and Lopez-Bayghen, E. (2009). The tight junction protein ZO-2 blocks cell cycle progression and inhibits cyclin D1 expression. Ann N Y Acad Sci 1165, 121–125.
Guo, Y.H., Wang, L.Q., Li, B., Xu, H., Yang, J.H., Zheng, L.S., Yu, P., Zhou, A.D., Zhang, Y., Xie, S.J., et al. (2016). Wnt/β-catenin pathway transactivates microRNA-150 that promotes EMT of colorectal cancer cells by suppressing CREB signaling. Oncotarget 7, 42513–42526.
Hanahan, D. (2022). Hallmarks of cancer: new dimensions. Cancer Discov 12, 31–46.
Huang, S., Pettaway, C.A., Uehara, H., Bucana, C.D., and Fidler, I.J. (2001). Blockade of NF-κB activity in human prostate cancer cells is associated with suppression of angiogenesis, invasion, and metastasis. Oncogene 20, 4188–4197.
Huber, M.A., Azoitei, N., Baumann, B., Grünert, S., Sommer, A., Pehamberger, H., Kraut, N., Beug, H., and Wirth, T. (2004). NF-κB is essential for epithelial-mesenchymal transition and metastasis in a model of breast cancer progression. J Clin Invest 114, 569–581.
Huerta, M., Muñoz, R., Tapia, R., Soto-Reyes, E., Ramírez, L., Recillas-Targa, F., González-Mariscal, L., and López-Bayghen, E. (2007). Cyclin D1 is transcriptionally down-regulated by ZO-2 via an E box and the transcription factor c-Myc. Mol Biol Cell 18, 4826–4836.
Hummler, E., Cole, T.J., Blendy, J.A., Ganss, R., Aguzzi, A., Schmid, W., Beermann, F., and Schütz, G. (1994). Targeted mutation of the CREB gene: compensation within the CREB/ATF family of transcription factors. Proc Natl Acad Sci USA 91, 5647–5651.
Impey, S., McCorkle, S.R., Cha-Molstad, H., Dwyer, J.M., Yochum, G.S., Boss, J.M., McWeeney, S., Dunn, J.J., Mandel, G., and Goodman, R.H. (2004). Defining the CREB regulon: a genome-wide analysis of transcription factor regulatory regions. Cell 119, 1041–1054.
James, M.A., Lu, Y., Liu, Y., Vikis, H.G., and You, M. (2009). RGS17, an overexpressed gene in human lung and prostate cancer, induces tumor cell proliferation through the cyclic AMP-PKA-CREB pathway. Cancer Res 69, 2108–2116.
Jansen-Dürr, P., Meichle, A., Steiner, P., Pagano, M., Finke, K., Botz, J., Wessbecher, J., Draetta, G., and Eilers, M. (1993). Differential modulation of cyclin gene expression by MYC. Proc Natl Acad Sci USA 90, 3685–3689.
Kam, Y., Rubinstein, A., Naik, S., Djavsarov, I., Halle, D., Ariel, I., Gure, A.O., Stojadinovic, A., Pan, H.G., Tsivin, V., et al. (2014). Detection of a long non-coding RNA (CCAT1) in living cells and human adenocarcinoma of colon tissues using FIT-PNA molecular beacons. Cancer Lett 352, 90–96.
Kanehisa, M., Sato, Y., Furumichi, M., Morishima, K., and Tanabe, M. (2019). New approach for understanding genome variations in KEGG. Nucleic Acids Res 47, D590–D595.
Kim, T., Jeon, Y.J., Cui, R., Lee, J.H., Peng, Y., Kim, S.H., Tili, E., Alder, H., and Croce, C.M. (2015). Role of MYC-regulated long noncoding RNAs in cell cycle regulation and tumorigenesis. JNCI J Natl Cancer Inst 107, dju505.
Kunsch, C., Lang, R.K., Rosen, C.A., and Shannon, M.F. (1994). Synergistic transcriptional activation of the IL-8 gene by NF-kappa B p65 (RelA) and NF-IL-6. J Immunol 153, 153–164.
Lee, H.J.J., Mignacca, R.C., and Sakamoto, K.M. (1995). Transcriptional activation of egr-1 by granulocyte-macrophage colony-stimulating factor but not interleukin 3 requires phosphorylation of cAMP response element-binding protein (CREB) on serine 133. J Biol Chem 270, 15979–15983.
Lee, R.J., Albanese, C., Stenger, R.J., Watanabe, G., Inghirami, G., Haines Iii, G.K., Webster, M., Muller, W.J., Brugge, J.S., Davis, R.J., et al. (1999). pp60v-src induction of cyclin D1 requires collaborative interactions between the extracellular signal-regulated kinase, p38, and Jun kinase pathways. A role for cAMP response element-binding protein and activating transcription factor-2 in pp60v-src signaling in breast cancer cells. J Biol Chem 274, 7341–7350.
Lemberger, T., Parkitna, J.R., Chai, M., Schütz, G., and Engblom, D. (2008). CREB has a context-dependent role in activity-regulated transcription and maintains neuronal cholesterol homeostasis. FASEB J 22, 2872–2879.
Liu, B.D., Sun, L.J., and Song, E.W. (2013). Non-coding RNAs regulate tumor cell plasticity. Sci China Life Sci 56, 886–890.
Liu, H., Radisky, D.C., Yang, D., Xu, R., Radisky, E.S., Bissell, M.J., and Bishop, J.M. (2012). MYC suppresses cancer metastasis by direct transcriptional silencing of αv and β3 integrin subunits. Nat Cell Biol 14, 567–574.
Lonze, B.E., and Ginty, D.D. (2002). Function and regulation of CREB family transcription factors in the nervous system. Neuron 35, 605–623.
Merrell, A.J., and Stanger, B.Z. (2016). Adult cell plasticity in vivo: de-differentiation and transdifferentiation are back in style. Nat Rev Mol Cell Biol 17, 413–425.
Michael, M.D., Michael, L.F., and Simpson, E.R. (1997). A CRE-like sequence that binds CREB and contributes to cAMP-dependent regulation of the proximal promoter of the human aromatase P450 (CYP19) gene. Mol Cell Endocrinol 134, 147–156.
Nagata, D., Suzuki, E., Nishimatsu, H., Satonaka, H., Goto, A., Omata, M., and Hirata, Y. (2001). Transcriptional activation of the cyclin D1 gene is mediated by multiple cis-elements, including SP1 sites and a cAMP-responsive element in vascular endothelial cells. J Biol Chem 276, 662–669.
Nissan, A., Stojadinovic, A., Mitrani-Rosenbaum, S., Halle, D., Grinbaum, R., Roistacher, M., Bochem, A., Dayanc, B.E., Ritter, G., Gomceli, I., et al. (2012). Colon cancer associated transcript-1: a novel RNA expressed in malignant and pre-malignant human tissues. Int J Cancer 130, 1598–1606.
Perkins, N.D., and Gilmore, T.D. (2006). Good cop, bad cop: the different faces of NF-κB. Cell Death Differ 13, 759–772.
Philipp, A., Schneider, A., Väsrik, I., Finke, K., Xiong, Y., Beach, D., Alitalo, K., and Eilers, M. (1994). Repression of cyclin D1: a novel function of MYC. Mol Cell Biol 14, 4032–4043.
Ran, F.A., Hsu, P.D., Lin, C.Y., Gootenberg, J.S., Konermann, S., Trevino, A.E., Scott, D.A., Inoue, A., Matoba, S., Zhang, Y., et al. (2013). Double nicking by RNA-guided CRISPR Cas9 for enhanced genome editing specificity. Cell 154, 1380–1389.
Romano, G., Chagani, S., and Kwong, L.N. (2018). The path to metastatic mouse models of colorectal cancer. Oncogene 37, 2481–2489.
Sapio, L., Salzillo, A., Ragone, A., Illiano, M., Spina, A., and Naviglio, S. (2020). Targeting CREB in cancer therapy: a key candidate or one of many? An update. Cancers 12, 3166.
Seoane, J., Le, H.V., and Massagué, J. (2002). Myc suppression of the p21Cip1 Cdk inhibitor influences the outcome of the p53 response to DNA damage. Nature 419, 729–734.
Shankar, D.B., Cheng, J.C., Kinjo, K., Federman, N., Moore, T.B., Gill, A., Rao, N.P., Landaw, E.M., and Sakamoto, K.M. (2005). The role of CREB as a proto-oncogene in hematopoiesis and in acute myeloid leukemia. Cancer Cell 7, 351–362.
Shankar, D.B., and Sakamoto, K.M. (2004). The role of cyclic-AMP binding protein (CREB) in leukemia cell proliferation and acute leukemias. Leukemia Lymphoma 45, 265–270.
Solomon, D.L., Philipp, A., Land, H., and Eilers, M. (1995). Expression of cyclin D1 mRNA is not upregulated by Myc in rat fibroblasts. Oncogene 11, 1893–1897.
Staller, P., Peukert, K., Kiermaier, A., Seoane, J., Lukas, J., Karsunky, H., Möröy, T., Bartek, J., Massagué, J., Hänel, F., et al. (2001). Repression of p15INK4b expression by Myc through association with Miz-1. Nat Cell Biol 3, 392–399.
Subramanian, A., Tamayo, P., Mootha, V.K., Mukherjee, S., Ebert, B.L., Gillette, M.A., Paulovich, A., Pomeroy, S.L., Golub, T.R., Lander, E.S., et al. (2005). Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci USA 102, 15545–15550.
Swier, L.J.Y.M., Dzikiewicz-Krawczyk, A., Winkle, M., van den Berg, A., and Kluiver, J. (2019). Intricate crosstalk between MYC and non-coding RNAs regulates hallmarks of cancer. Mol Oncol 13, 26–45.
Tan, X., Wang, S., Zhu, L., Wu, C., Yin, B., Zhao, J., Yuan, J., Qiang, B., and Peng, X. (2012). cAMP response element-binding protein promotes gliomagenesis by modulating the expression of oncogenic microRNA-23a. Proc Natl Acad Sci USA 109, 15805–15810.
Tripathi, S., Pohl, M.O., Zhou, Y., Rodriguez-Frandsen, A., Wang, G., Stein, D.A., Moulton, H.M., DeJesus, P., Che, J., Mulder, L.C.F., et al. (2015). Meta- and orthogonal integration of influenza “OMICs” data defines a role for UBR4 in virus budding. Cell Host Microbe 18, 723–735.
Wang, J., Liu, X., Wu, H., Ni, P., Gu, Z., Qiao, Y., Chen, N., Sun, F., and Fan, Q. (2010). CREB up-regulates long non-coding RNA, HULC expression through interaction with microRNA-372 in liver cancer. Nucleic Acids Res 38, 5366–5383.
Wang, X.W., Hu, L.F., Hao, J., Liao, L.Q., Chiu, Y.T., Shi, M., and Wang, Y. (2019). A microRNA-inducible CRISPR-Cas9 platform serves as a microRNA sensor and cell-type-specific genome regulation tool. Nat Cell Biol 21, 522–530.
Wilson, B.E., Mochon, E., and Boxer, L.M. (1996). Induction of bcl-2 expression by phosphorylated CREB proteins during B-cell activation and rescue from apoptosis. Mol Cell Biol 16, 5546–5556.
Wood, L.D., Farmer, A.A., and Richmond, A. (1995). HMGI(Y) and Sp1 in addition to NF-κB regulate transcription of the MGSA/GROα gene. Nucl Acids Res 23, 4210–4219.
Wu, D., Zhau, H.E., Huang, W.C., Iqbal, S., Habib, F.K., Sartor, O., Cvitanovic, L., Marshall, F.F., Xu, Z., and Chung, L.W.K. (2007). cAMP-responsive element-binding protein regulates vascular endothelial growth factor expression: implication in human prostate cancer bone metastasis. Oncogene 26, 5070–5077.
Xiang, J.F., Yin, Q.F., Chen, T., Zhang, Y., Zhang, X.O., Wu, Z., Zhang, S., Wang, H.B., Ge, J., Lu, X., et al. (2014). Human colorectal cancer-specific CCAT1-L lncRNA regulates long-range chromatin interactions at the MYC locus. Cell Res 24, 513–531.
Xin, Y., Li, Z., Shen, J., Chan, M.T.V., and Wu, W.K.K. (2016). CCAT1: a pivotal oncogenic long non-coding RNA in human cancers. Cell Prolif 49, 255–260.
Yang, W., Shen, J., Wu, M., Arsura, M., FitzGerald, M., Suldan, Z., Kim, D.W., Hofmann, C.S., Pianetti, S., Romieu-Mourez, R., et al. (2001). Repression of transcription of the p27Kip1 cyclin-dependent kinase inhibitor gene by c-Myc. Oncogene 20, 1688–1702.
Yuan, S., Norgard, R.J., and Stanger, B.Z. (2019). Cellular plasticity in cancer. Cancer Discov 9, 837–851.
Yuan, Y., Li, H., Pu, W., Chen, L., Guo, D., Jiang, H., He, B., Qin, S., Wang, K., Li, N., et al. (2022). Cancer metabolism and tumor microenvironment: fostering each other? Sci China Life Sci 65, 236–279.
Zhang, C., Wang, L., Wu, D., Chen, H., Chen, Z., Thomas-Ahner, J.M., Zynger, D.L., Eeckhoute, J., Yu, J., Luo, J., et al. (2011). Definition of a FoxA1 cistrome that is crucial for G1 to S-phase cell-cycle transit in castration-resistant prostate cancer. Cancer Res 71, 6738–6748.
Zhang, X., Odom, D.T., Koo, S.H., Conkright, M.D., Canettieri, G., Best, J., Chen, H., Jenner, R., Herbolsheimer, E., Jacobsen, E., et al. (2005). Oenome-wide analysis of cAMP-response element binding protein occupancy, phosphorylation, and target gene activation in human tissues. Proc Natl Acad Sci USA 102, 4459–4464.
Zhao, Y.J., Zhang, J., Wang, Y.C., Wang, L., and He, X.Y. (2021). MiR-450a-5p inhibits gastric cancer cell proliferation, migration, and invasion and promotes apoptosis via targeting CREB1 and inhibiting AKT/OSK-3β signaling pathway. Front Oncol 11, 633366.
Zhou, K.R., Liu, S., Sun, W.J., Zheng, L.L., Zhou, H., Yang, J.H., and Qu, L.H. (2017). ChIPBase v2.0: decoding transcriptional regulatory networks of non-coding RNAs and protein-coding genes from ChIPseq data. Nucleic Acids Res 45, D43–D50.
Zong, W.X., Edelstein, L.C., Chen, C., Bash, J., and Oélinas, C. (1999). The prosurvival Bcl-2 homolog Bfl-1/A1 is a direct transcriptional target of NF-kappa B that blocks TNFalpha -induced apoptosis. Genes Dev 13, 382–387.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (31970604, 31900903, 31770879), the Major Research Plan of the National Natural Science Foundation of China (91940000), the National Key Research and Development Program of China (2017YFA0504400), in part by the Guangdong Province Key Laboratory of Computational Science (13lgjc05), the Guangdong Province Computational Science Innovative Research Team (14lgjc18), and the Fundamental Research Funds for the Central Universities (20lgpy112, 2021qntd26).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Compliance and ethics The author(s) declare that they have no conflict of interest.
Electronic supplementary material
11427_2022_2108_MOESM2_ESM.xls
Supplementary Table S1. Genes differentially expressed upon knocking out of CREB1 in HCT116 cells identified by the RNA-seq analysis.
Rights and permissions
About this article
Cite this article
Li, B., Zheng, L., Ye, J. et al. CREB1 contributes colorectal cancer cell plasticity by regulating lncRNA CCAT1 and NF-κB pathways. Sci. China Life Sci. 65, 1481–1497 (2022). https://doi.org/10.1007/s11427-022-2108-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-022-2108-x