Abstract
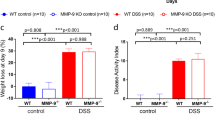
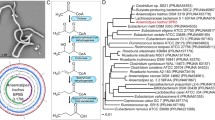
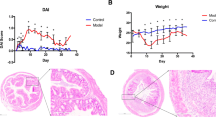
The dextran sulfate sodium (DSS)-induced colitis model is a widely applied mouse model, but controversial results have been obtained from experiments using the same mouse strain under the same conditions. Because the gut microbiota play an important role in DSS-induced colitis, it is essential to evaluate the influence of the initial gut microbiota in this model. Here, we identified significant variations in the initial gut microbiota of different batches of mice and found that the initial intestinal microbiota had a profound influence on DSS-induced colitis. We performed three independent trials using the same C57BL/6J mouse model with DSS treatment and used high-throughput 16S rRNA gene sequencing to analyze the gut microbiota. We found that the structure and composition of the gut microbiota in mice with severe colitis, as compared with mice with milder colon damage, had unique features, such as an increase in Akkermansia bacteria and a decrease in Barnesiella spp. Moreover, these varied gut bacteria in the different trials also showed different responses to DSS treatment. Our work suggests that, in studies using mouse models, the gut microbiota must be considered when examining mechanisms of diseases, to ensure that comparable results are obtained.
Similar content being viewed by others
References
Bauer, C., Duewell, P., Mayer, C., Lehr, H.A., Fitzgerald, K.A., Dauer, M., Tschopp, J., Endres, S., Latz, E., and Schnurr, M. (2010). Colitis induced in mice with dextran sulfate sodium (DSS) is mediated by the NLRP3 inflammasome. Gut 59, 1192–1199.
Berry, D., and Widder, S.(2014). Deciphering microbial interactions and detecting keystone species with co-occurrence networks. Front Microbiol 5, 219.
Caporaso, J.G., Kuczynski, J., Stombaugh, J., Bittinger, K., Bushman, F.D., Costello, E.K., Fierer, N., Peña, A.G., Goodrich, J.K., Gordon, J.I., Huttley, G.A., Kelley, S.T., Knights, D., Koenig, J.E., Ley, R.E., Lozupone, C.A., McDonald, D., Muegge, B.D., Pirrung, M., Reeder, J., Sevinsky, J.R., Turnbaugh, P.J., Walters, W.A., Widmann, J., Yatsunenko, T., Zaneveld, J., and Knight, R. (2010). QIIME allows analysis of high-throughput community sequencing data. Nat Meth 7, 335–336.
Chassaing, B., Srinivasan, G., Delgado, M.A., Young, A.N., Gewirtz, A.T., and Vijay-Kumar, M. (2012). Fecal lipocalin 2, a sensitive and broadly dynamic non-invasive biomarker for intestinal inflammation. PLoS ONE 7, e44328.
Collins, F.S., and Tabak, L.A. (2014). Policy: NIH plans to enhance reproducibility. Nature 505, 612–613.
Elinav, E., Strowig, T., Kau, A.L., Henao-Mejia, J., Thaiss, C.A., Booth, C.J., Peaper, D.R., Bertin, J., Eisenbarth, S.C., Gordon, J.I., and Flavell, R.A. (2011). NLRP6 inflammasome regulates colonic microbial ecology and risk for colitis. Cell 145, 745–757.
Endesfelder, D., zu Castell, W., Ardissone, A., Davis-Richardson, A.G., Achenbach, P., Hagen, M., Pflueger, M., Gano, K.A., Fagen, J.R., Drew, J.C., Brown, C.T., Kolaczkowski, B., Atkinson, M., Schatz, D., Bonifacio, E., Triplett, E.W., and Ziegler, A.G. (2014). Compromised gut microbiota networks in children with anti-islet cell autoimmunity. Diabetes 63, 2006–2014.
Estrada, E., and Rodríguez-Velázquez, J.A. (2005). Subgraph centrality in complex networks. Phys Rev E 71, 056103.
Fayad, R., Pini, M., Sennello, J.A., Cabay, R.J., Chan, L., Xu, A., and Fantuzzi, G. (2007). Adiponectin deficiency protects mice from chemically induced colonic inflammation. Gastroenterology 132, 601–614.
Freeman, L.C. (1977). A set of measures of centrality based on betweenness. Sociometry 40, 35–41.
Friedman, J., and Alm, E.J. (2012). Inferring correlation networks from genomic survey data. PLoS Comput Biol 8, e1002687.
Godon, J.J., Zumstein, E., Dabert, P., Habouzit, F., and Moletta, R. (1997). Molecular microbial diversity of an anaerobic digestor as determined by small-subunit rDNA sequence analysis. Appl Environ Microbiol 63, 2802–2813.
Håkansson, Å., Tormo-Badia, N., Baridi, A., Xu, J., Molin, G., Hagslätt, M.L., Karlsson, C., Jeppsson, B., Cilio, C.M., and Ahrné, S. (2015). Immunological alteration and changes of gut microbiota after dextran sulfate sodium (DSS) administration in mice. Clin Exp Med 15, 107–120.
Heimesaat, M.M., Fischer, A., Siegmund, B., Kupz, A., Niebergall, J., Fuchs, D., Jahn, H.K., Freudenberg, M., Loddenkemper, C., Batra, A., Lehr, H.A., Liesenfeld, O., Blaut, M., Göbel, U.B., Schumann, R.R., and Bereswill, S. (2007). Shift towards pro-inflammatory intestinal bacteria aggravates acute murine colitis via toll-like receptors 2 and 4. PLoS ONE 2, e662.
Hernandez-Chirlaque, C., Aranda, C.J., Ocon, B., Capitan-Canadas, F., Ortega-Gonzalez, M., Carrero, J.J., Suarez, M.D., Zarzuelo, A., Sanchez de Medina, F., and Martinez-Augustin, O. (2016). Germ-free and antibiotic-treated mice are highly susceptible to epithelial injury in DSS colitis. J Crohns Colitis 10, 1324–1335.
Justice, M.J., and Dhillon, P. (2016). Using the mouse to model human disease: increasing validity and reproducibility. Dis Model Mech 9, 101–103.
Lam, Y.Y., Ha, C.W.Y., Campbell, C.R., Mitchell, A.J., Dinudom, A., Oscarsson, J., Cook, D.I., Hunt, N.H., Caterson, I.D., Holmes, A.J., and Storlien, L.H. (2012). Increased gut permeability and microbiota change associate with mesenteric fat inflammation and metabolic dysfunction in diet-induced obese mice. PLoS ONE 7, e34233.
Macleod, M. (2011). Why animal research needs to improve. Nature 477, 511–511.
McGonigle, P., and Ruggeri, B. (2014). Animal models of human disease: challenges in enabling translation. Biochem Pharmacol 87, 162–171.
Mizoguchi, A. (2012). Animal models of inflammatory bowel disease. Prog Mol Biol Transl Sci 105, 263–320.
Nagalingam, N.A., Kao, J.Y., and Young, V.B. (2011). Microbial ecology of the murine gut associated with the development of dextran sodium sulfate-induced colitis. Inflamm Bowel Diss 17, 917–926.
Naqvi, A., Rangwala, H., Keshavarzian, A., and Gillevet, P. (2010). Network-based modeling of the human gut microbiome. Chem Biodiver 7, 1040–1050.
Nguyen, T.L., Vieira-Silva, S., Liston, A., and Raes, J. (2015). How informative is the mouse for human gut microbiota research? Dis Model Mech 8, 1–16.
Nishihara, T., Matsuda, M., Araki, H., Oshima, K., Kihara, S., Funahashi, T., and Shimomura, I. (2006). Effect of adiponectin on murine colitis induced by dextran sulfate sodium. Gastroenterology 131, 853–861.
Okayasu, I., Hatakeyama, S., Yamada, M., Ohkusa, T., Inagaki, Y., and Nakaya, R. (1990). A novel method in the induction of reliable experimental acute and chronic ulcerative colitis in mice. Gastroenterology 98, 694–702.
Prinz, F., Schlange, T., and Asadullah, K. (2011). Believe it or not: how much can we rely on published data on potential drug targets? Nat Rev Drug Discov 10, 712.
Shannon, P., Markiel, A., Ozier, O., Baliga, N.S., Wang, J.T., Ramage, D., Amin, N., Schwikowski, B., and Ideker, T. (2003). Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res 13, 2498–2504.
Siegmund, B., Lehr, H.A., Fantuzzi, G., and Dinarello, C.A. (2001). IL-1beta-converting enzyme (caspase-1) in intestinal inflammation. Proc Natl Acad Sci USA 98, 13249–13254.
Sivakumar, P.V., Westrich, G.M., Kanaly, S., Garka, K., Born, T.L., Derry, J.M., and Viney, J.L. (2002). Interleukin 18 is a primary mediator of the inflammation associated with dextran sulphate sodium induced colitis: blocking interleukin 18 attenuates intestinal damage. Gut 50, 812–820.
Takagi, H., Kanai, T., Okazawa, A., Kishi, Y., Sato, T., Takaishi, H., Inoue, N., Ogata, H., Iwao, Y., Hoshino, K., Takeda, K., Akira, S., Watanabe, M., Ishii, H., and Hibi, T. (2003). Contrasting action of IL-12 and IL-18 in the development of dextran sodium sulphate colitis in mice. Scand J Gastroenterol 38, 837–844.
ter Braak, C.J.F., and Smilauer, P.J. (1988). CANOCO Reference Manual and User’s Guide to Canoco for Windows: Software for Canonical Community Ordination (Version 4). (Texas Willis: Microcomputer Power), pp. 352.
Weiss, G.A., Chassard, C., and Hennet, T. (2014). Selective proliferation of intestinal Barnesiella under fucosyllactose supplementation in mice. Br J Nutr 111, 1602–1610.
Zaki, M.H., Boyd, K.L., Vogel, P., Kastan, M.B., Lamkanfi, M., and Kanneganti, T.D. (2010). The NLRP3 inflammasome protects against loss of epithelial integrity and mortality during experimental colitis. Immunity 32, 379–391.
Zhang, Q., Wu, Y., Wang, J., Wu, G., Long, W., Xue, Z., Wang, L., Zhang, X., Pang, X., Zhao, Y., Zhao, L., and Zhang, C. (2016). Accelerated dysbiosis of gut microbiota during aggravation of DSS-induced colitis by a butyrate-producing bacterium. Sci Rep 6, 27572.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (81401141), and Science and Technology Commission of Shanghai Municipality (14YF1402200).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Li, M., Wu, Y., Hu, Y. et al. Initial gut microbiota structure affects sensitivity to DSS-induced colitis in a mouse model. Sci. China Life Sci. 61, 762–769 (2018). https://doi.org/10.1007/s11427-017-9097-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-017-9097-0