Abstract
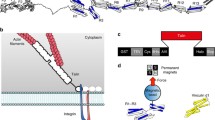
Talin is an integrin-binding protein located at focal adhesion site and serves as both an adapter and a force transmitter. Its integrin binding activity is regulated by the intramolecular autoinhibition interaction between its F3 and RS domains. Here, we used atomic force microscopy to measure the strength of talin autoinhibition complex. Our results suggest that the lifetime of talin autoinhibition complex shows weak catch bond behavior and does not change significantly at smaller forces, while it drops rapidly at larger forces (>10 pN). Moreover, besides the complex conformation revealed by crystal structure, our molecular dynamics (MD) simulations indicate the possible existence of another stable conformation. Further analysis indicates that forces may regulate the equilibrium of the two stable binding states and result in the non-exponential force dependence of the binding lifetime. Our findings reveal a negative regulation mechanism on talin activation and provide a new point of view on the function of talin in focal adhesion.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Critchley DR. Biochemical and structural properties of the integrin-associated cytoskeletal protein talin. Annu Rev Biophys, 2009, 38: 235–254
Di Paolo G, Pellegrini L, Letinic K, Cestra G, Zoncu R, Voronov S, Chang SH, Guo J, Wenk MR, De Camilli P. Recruitment and regulation of phosphatidylinositol phosphate kinase type 1 gamma by the ferm domain of talin. Nature, 2002, 420: 85–89
Lee HS, Bellin RM, Walker DL, Patel B, Powers P, Liu HJ, Garcia-Alvarez B, de Pereda JM, Liddington RC, Volkmann N, Hanein D, Critchley DR, Robson RM. Characterization of an actin-binding site within the talin ferm domain. J Mol Biol, 2004, 343: 771–784
Goldfinger LE, Ptak C, Jeffery ED, Shabanowitz J, Han JW, Haling JR, Sherman NE, Fox JW, Hunt DF, Ginsberg MH. An experimentally derived database of candidate ras-interacting proteins. J Proteome Res, 2007, 6: 1806–1811
Goksoy E, Ma YQ, Wang XX, Kong XM, Perera D, Plow EF, Qin J. Structural basis for the autoinhibition of talin in regulating integrin activation. Mol Cell, 2008, 31: 124–133
Burridge K, Mangeat P. An interaction between vinculin and talin. Nature, 1984, 308: 744–746
Gilmore AP, Burridge K. Regulation of vinculin binding to talin and actin by phosphatidyl-inositol-4–5-bisphosphate. Nature, 1996, 381: 531–535
Calderwood DA, Ginsberg MH. Talin forges the links between integrins and actin. Nat Cell Biol, 2003, 5: 694–697
Sun N, Critchley DR, Paulin D, Li ZL, Robson RM. Identification of a repeated domain within mammalian alpha-synemin that interacts directly with talin. Exp Cell Res, 2008, 314: 1839–1849
Zhang X, Jiang G, Cai Y, Monkley SJ, Critchley DR, Sheetz MP. Talin depletion reveals independence of initial cell spreading from integrin activation and traction. Nat Cell Biol, 2008, 10: 1062–1068
Wegener KL, Partridge AW, Han J, Pickford AR, Liddington RC, Ginsberg MH, Campbell ID. Structural basis of integrin activation by talin. Cell, 2007, 128: 171–182
Tadokoro S, Shattil SJ, Eto K, Tai V, Liddington RC, de Pereda JM, Ginsberg MH, Calderwood DA. Talin binding to integrin beta tails: a final common step in integrin activation. Science, 2003, 302: 103–106
Giannone G, Sheetz MP. Substrate rigidity and force define form through tyrosine phosphatase and kinase pathways. Trends Cell Biol, 2006, 16: 213–223
Jiang GY, Giannone G, Critchley DR, Fukumoto E, Sheetz MP. Two-piconewton slip bond between fibronectin and the cytoskeleton depends on talin. Nature, 2003, 424: 334–337
Giannone G, Jiang G, Sutton DH, Critchley DR, Sheetz MP. Talin1 is critical for force-dependent reinforcement of initial integrin-cytoskeleton bonds but not tyrosine kinase activation. J Cell Biol, 2003, 163: 409–419
Calderwood DA, Zent R, Grant R, Rees DJG, Hynes RO, Ginsberg MH. The talin head domain binds to integrin beta subunit cytoplasmic tails and regulates integrin activation. J Biol Chem, 1999, 274: 28071–28074
Song XQ, Yang J, Hirbawi J, Ye S, Perera HD, Goksoy E, Dwivedi P, Plow EF, Zhang RG, Qin J. A novel membrane-dependent on/off switch mechanism of talin ferm domain at sites of cell adhesion. Cell Res, 2012, 22: 1533–1545
Goult BT, Bate N, Anthis NJ, Wegener KL, Gingras AR, Patel B, Barsukov IL, Campbell ID, Roberts GCK, Critchley DR. The structure of an interdomain complex that regulates talin activity. J Biol Chem, 2009, 284: 15097–15106
Lee HS, Lim CJ, Puzon-McLaughlin W, Shattil SJ, Ginsberg MH. Riam activates integrins by linking talin to ras gtpase membrane-targeting sequences. J Biol Chem, 2009, 284: 5119–5127
Han JW, Lim CJ, Watanabe N, Soriani A, Ratnikov B, Calderwood DA, Puzon-McLaughlin W, Lafuente EM, Boussiotis VA, Shattil SJ, Ginsberg MH. Reconstructing and deconstructing agonist-induced activation of integrin alpha iib beta 3. Curr Biol, 2006, 16: 1796–1806
Yang J, Zhu L, Zhang H, Hirbawi J, Fukuda K, Dwivedi P, Liu J, Byzova T, Plow EF, Wu J, Qin J. Conformational activation of talin by riam triggers integrin-mediated cell adhesion. Nat Commun, 2014, 5: 5880
Barsukov IL, Prescot A, Bate N, Patel B, Floyd DN, Bhanji N, Bagshaw CR, Letinic K, Di Paolo G, De Camilli P, Roberts GCK, Critchley DR. Phosphatidylinositol phosphate kinase type 1 gamma and beta(1)-integrin cytoplasmic domain bind to the same region in the talin ferm domain. J Biol Chem, 2003, 278: 31202–31209
Burridge K, Connell L. A new protein of adhesion plaques and ruffling membranes. J Cell Biol, 1983, 97: 359–367
del Rio A, Perez-Jimenez R, Liu R, Roca-Cusachs P, Fernandez JM, Sheetz MP. Stretching single talin rod molecules activates vinculin binding. Science, 2009, 323: 638–641
Grashoff C, Hoffman BD, Brenner MD, Zhou RB, Parsons M, Yang MT, McLean MA, Sligar SG, Chen CS, Ha T, Schwartz MA. Measuring mechanical tension across vinculin reveals regulation of focal adhesion dynamics. Nature, 2010, 466: 263–266
Roca-Cusachs P, del Rio A, Puklin-Faucher E, Gauthier NC, Biais N, Sheetz MP. Integrin-dependent force transmission to the extracellular matrix by alpha-actinin triggers adhesion maturation. Proc Natl Acad Sci USA, 2013, 110: E1361–E1370
Wang XF, Ha T. Defining single molecular forces required to activate integrin and notch signaling. Science, 2013, 340: 991–994
Deng QP, Huo YQ, Luo JC. Endothelial mechanosensors: the gatekeepers of vascular homeostasis and adaptation under mechanical stress. Sci China Life Sci, 2014, 57: 755–762
Kong F, García AJ, Mould AP, Humphries MJ, Zhu C. Demonstration of catch bonds between an integrin and its ligand. J Cell Biol, 2009, 185: 1275–1284
Ebner A, Hinterdorfer P, Gruber HJ. Comparison of different aminofunctionalization strategies for attachment of single antibodies to afm cantilevers. Ultramicroscopy, 2007, 107: 922–927
Sarangapani KK, Marshall BT, McEver RP, Zhu C. Molecular stiffness of selectins. J Biol Chem, 2011, 286: 9567–9576
Kong F, Li ZH, Parks WM, Dumbauld DW, Garcia AJ, Mould AP, Humphries MJ, Zhu C. Cyclic mechanical reinforcement of integrin-ligand interactions. Mol Cell, 2013, 49: 1060–1068
Zhang XH, Wojcikiewicz E, Moy VT. Force spectroscopy of the leukocyte function-associated antigen-1/intercellular adhesion molecule-1 interaction. Biophys J, 2002, 83: 2270–2279
Klein DCG, Ovrebo KM, Latz E, Espevik T, Stokke BT. Direct measurement of the interaction force between immunostimulatory cpg-DNA and tlr9 fusion protein. J Mol Recognit, 2012, 25: 74–81
Yan C, Yersin A, Afrin R, Sekiguchi H, Ikai A. Single molecular dynamic interactions between glycophorin a and lectin as probed by atomic force microscopy. Biophys Chem, 2009, 144: 72–77
Evans E. Probing the relation between force-lifetime-and chemistry in single molecular bonds. Ann Rev Biophys Biomol Struct, 2001, 30: 105–128
Chen W, Lou J, Zhu C. Forcing switch from short- to intermediate- and long-lived states of the alphaa domain generates LFA-1/ICAM-1 catch bonds. J Biol Chem, 2010, 285: 35967–35978
Bell GI. Models for the specific adhesion of cells to cells. Science, 1978, 200: 618–627
Phillips JC, Braun R, Wang W, Gumbart J, Tajkhorshid E, Villa E, Chipot C, Skee RD, Kalé L, Schulten K. Scalable molecular dynamics with namd. J Comput Chem, 2005, 26: 1781–1802
MacKerell A, Jr., Bashford D, Bellott M, Dunbrack RLJ, Evanseck J, Field MJ, Fischer S, Gao J, Guo H, Ha S, Joseph D, Kuchnir L, Kuczera K, Lau FTK, Mattos C, Michnick S, Ngo T, Nguyen DT, Prodhom B, Reiher IWE, Roux B, Schlenkrich M, Smith J, Stote R, Straub J, Watanabe M, Wiorkiewicz-Kuczera J, Yin D, Karplus M. All-atom empirical potential for molecular modeling and dynamics studies of proteins. J Phys Chem B, 1998, 102: 3586–3616
Humphrey W, Dalke A, Schulten K. VMD: visual molecular dynamics. J Mol Graphics, 1996, 14: 33–38
Lee CK, Wang YM, Huang LS, Lin SM. Atomic force microscopy: determination of unbinding force, off rate and energy barrier for protein-ligand interaction. Micron, 2007, 38: 446–461
Evans E, Ritchie K. Dynamic strength of molecular adhesion bonds. Biophys J, 1997, 72: 1541–1555
Merkel R, Nassoy P, Leung A, Ritchie K, Evans E. Energy landscapes of receptor-ligand bonds explored with dynamic force spectroscopy. Nature, 1999, 397: 50–53
Marshall BT, Long M, Piper JW, Yago T, McEver RP, Zhu C. Direct observation of catch bonds involving cell-adhesion molecules. Nature, 2003, 423: 190–193
Chen W, Lou J, Evans EA, Zhu C. Observing force-regulated conformational changes and ligand dissociation from a single integrin on cells. J Cell Biol, 2012, 199: 497–512
Yago T, Lou J, Wu T, Yang J, Miner JJ, Coburn L, Lopez JA, Cruz MA, Dong JF, McIntire LV, McEver RP, Zhu C. Platelet glycoprotein ibalpha forms catch bonds with human wt vwf but not with type 2b von willebrand disease vwf. J Clin Invest, 2008, 118: 3195–3207
Lee CY, Lou J, Wen KK, McKane M, Eskin SG, Ono S, Chien S, Rubenstein PA, Zhu C, McIntire LV. Actin depolymerization under force is governed by lysine 113:glutamic acid 195-mediated catch-slip bonds. Proc Natl Acad Sci USA, 2013, 110: 5022–5027
Guo B, Guilford WH. Mechanics of actomyosin bonds in different nucleotide states are tuned to muscle contraction. Proc Natl Acad Sci USA, 2006, 103: 9844–9849
Michael Feig, John Karanicolas, III CLB. Mmtsb tool set: enhanced sampling and multiscale modeling methods for applications in structural biology. J Mol Graphics Model, 2004, 22: 377–395
Author information
Authors and Affiliations
Corresponding authors
Additional information
Contributed equally to this work
This article is published with open access at link.springer.com
Electronic supplementary material
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Zeng, Y., Zhang, Y., Song, X. et al. The conformational states of talin autoinhibition complex and its activation under forces. Sci. China Life Sci. 58, 694–703 (2015). https://doi.org/10.1007/s11427-015-4873-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-015-4873-9