Abstract
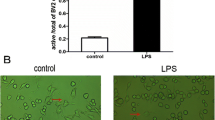
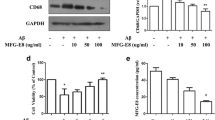
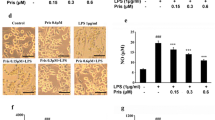
This paper aims to observe the changes of the inflammatory cytokines in microglial BV2 cells stimulated by apelin, and investigate the mechanism of inflammatory cytokines secretion after apelin stimulation. Immunofluorescence and quantitative real-time PCR were performed to observe expression of TNF-α, IL-1β, IL-10, MIP-1α, and MCP-1 in BV2 cells. Western blot was used to investigate the expression of phosphorylation PI-3K/Akt and phosphorylation Erk signaling pathways in BV2 cells after stimulation by apelin. Furthermore, PI-3K/Akt inhibitor (LY294402) and Erk inhibitor (U0126) were used as antagonists to detect the secretion mechanisms of cytokines in BV2 cells stimulated by apelin. Exogenous recombinant apelin activated the expression of TNF-α, IL-1β, MCP-1 and MIP-1α in BV2 cells by the detection of fluorescence expression and mRNA. Apelin also unregulated the protein expression of p-PI-3K/Akt and p-Erk in BV2 cells induced by apelin. LY294002 and U0126 inhibited activation of p-PI-3K/Akt and p-Erk expression by Western blot and attenuated the expression of inflammation factors in BV2 cells by fluorescence staining. This study demonstrates that apelin is a potential activator of inflammation factors through the PI3K/Akt and Erk signaling pathway and is potential therapeutically relevant to inflammatory responses of microglia cells.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
O’Dowd BF, Heiber M, Chan A, Heng HH, Tsui L-C, Kennedy JL, Shi X, Petronis A, George SR, Nguyen T. A human gene that shows identity with the gene encoding the angiotensin receptor is located on chromosome 11. Gene, 1993, 136: 355–360
Cox CM, D’Agostino SL, Miller MK, Heimark RL, Krieg PA. Apelin, the ligand for the endothelial G-protein-coupled receptor, APJ, is a potent angiogenic factor required for normal vascular development of the frog embryo. Dev Biol, 2006, 296: 177–189
Kasai A, Shintani N, Oda M, Kakuda M, Hashimoto H, Matsuda T, Hinuma S, Baba A. Apelin is a novel angiogenic factor in retinal endothelial cells. Biochem Biophys Res Commun, 2004, 325: 395–400
Kasai A, Ishimaru Y, Kinjo T, Satooka T, Matsumoto N, Yoshioka Y, Yamamuro A, Gomi F, Shintani N, Baba A, Maeda S. Apelin is a crucial factor for hypoxia-induced retinal angiogenesis. Arterioscl Throm Vasc Biol, 2010, 30: 2182–2187
Kasai A, Shintani N, Kato H, Matsuda S, Gomi F, Haba R, Hashimoto H, Kakuda M, Tano Y, Baba A. Retardation of retinal vascular development in apelin-deficient mice. Arterioscl Throm Vasc Biol, 2008, 28: 1717–1722
Hara C, Kasai A, Gomi F, Satooka T, Sakimoto S, Nakai K, Yoshioka Y, Yamamuro A, Maeda S, Nishida K. Laser-induced choroidal neovascularization in mice attenuated by deficiency in the apelin-APJ system. Invest Ophthalmol Vis Sci, 2013, 54: 4321–4329
Tao Y, Lu Q, Jiang YR, Qian J, Wang JY, Gao L, Jonas JB. Apelin in plasma and vitreous and in fibrovascular retinal membranes of patients with proliferative diabetic retinopathy. Invest Ophthalmol Vis Sci, 2010, 51: 4237–4242
Han S, Wang G, Qi X, Englander EW, Greeley Jr GH. Involvement of a Stat3 binding site in inflammation-induced enteric apelin expression. Am J Physiol Gastrointest Liver Physiol, 2008, 295: G1068
Karlstetter M, Ebert S, Langmann T. Microglia in the healthy and degenerating retina: insights from novel mouse models. Immunobiology, 2010, 215: 685–691
Lynch MA. The multifaceted profile of activated microglia. Mol Neurobiol, 2009, 40: 139–156
Krady JK, Basu A, Allen CM, Xu Y, LaNoue KF, Gardner TW, Levison SW. Minocycline reduces proinflammatory cytokine expression, microglial activation, and caspase-3 activation in a rodent model of diabetic retinopathy. Diabetes, 2005, 54: 1559–1565
Zeng HY, Green WR, Tso MO. Microglial activation in human diabetic retinopathy. Arch Ophthalmol, 2008, 126: 227–232
Zhang Y, Dong C. Regulatory mechanisms of mitogen-activated kinase signaling. Cell Mol Life Sci, 2007, 64: 2771–2789
Kim WK, Hwang SY, Oh ES, Piao HZ, Kim KW, Han IO. TGF-β represses activation and resultant death of microglia via inhibition of phosphatidylinositol 3-kinase activity. J Immunol, 2004, 172: 7015–7023
Zhou Y, Ling EA, Dheen ST. Dexamethasone suppresses monocyte chemoattractant protein-1 production via mitogen activated protein kinase phosphatase-1 dependent inhibition of Jun N-terminal kinase and p38 mitogen-activated protein kinase in activated rat microglia. J Neurochem, 2007, 102: 667–678
Park HY, Han MH, Park C, Jin CY, Kim GY, Choi IW, Kim ND, Nam TJ, Kwon TK, Choi YH. Anti-inflammatory effects of fucoidan through inhibition of NF-kappaB, MAPK and Akt activation in lipopolysaccharide-induced BV2 microglia cells. Food Chem Toxicol, 2011, 49: 1745–1752
Zhang Y, Jiang YR, Lu Q, Yin H, Tao Y. Apelin in epiretinal fibrovascular membranes of patients with retinopathy of prematurity and the changes after intravitreal bevacizumab. Retina, 2013, 33: 613–620
Tatemoto K, Takayama K, Zou MX, Kumaki I, Zhang W, Kumano K, Fujimiya M. The novel peptide apelin lowers blood pressure via a nitric oxide-dependent mechanism. Regul Peptides, 2001, 99: 87–92
Kawamata Y, Habata Y, Fukusumi S, Hosoya M, Fujii R, Hinuma S, Nishizawa N, Kitada C, Onda H, Nishimura O. Molecular properties of apelin: tissue distribution and receptor binding. Biochim Biophys Acta Mol Cell Res, 2001, 1538: 162–171
Chu J, Zhang H, Huang X, Lin Y, Shen T, Chen B, Man Y, Wang S, Li J. Apelin ameliorates TNF-a-induced reduction of glycogen synthesis in the hepatocytes through G protein-coupled receptor APJ. PLoS One, 2013, 8: e57231
Devic E, Rizzoti K, Bodin S, Knibiehler B, Audigier Y. Amino acid sequence and embryonic expression of msr/apj, the mouse homolog of Xenopus X-msr and human APJ. Mech Dev, 1999, 84: 199–203
Leeper NJ, Tedesco MM, Kojima Y, Schultz GM, Kundu RK, Ashley EA, Tsao PS, Dalman RL, Quertermous T. Apelin prevents aortic aneurysm formation by inhibiting macrophage inflammation. Am J Physiol Heart Circulat Physiol, 2009, 296: H1329
Hu PF, Chen WP, Tang JL, Bao JP, Wu LD. Apelin plays a catabolic role on articular cartilage: in vivo and in vitro studies. Int J Mol Med, 2010, 26: 357–363
Rothwell NJ, Luheshi GN. Interleukin 1 in the brain: biology, pathology and therapeutic target. Trends Neurosci, 2000, 23: 618–625
Kowluru RA, Odenbach S. Role of interleukin-1β in the development of retinopathy in rats: effect of antioxidants. Invest Ophthalmol Vis Sci, 2004, 45: 4161–4166
Rungger-Brändle E, Dosso AA, Leuenberger PM. Glial reactivity, an early feature of diabetic retinopathy. Invest Ophthalmol Vis Sci, 2000, 41: 1971–1980
John GR, Lee SC, Song X, Rivieccio M, Brosnan CF. IL-1-regulated responses in astrocytes: relevance to injury and recovery. Glia, 2005, 49: 161–176
Lu Q, Jiang YR, Qian J, Tao Y. Apelin-13 regulates proliferation, migration and survival of retinal Müller cells under hypoxia. Diabetes Res Clin Pract, 2013, 99: 158–167
Wang XL, Tao Y, Lu Q, Jiang YR. Apelin supports primary rat retinal Müller cells under chemical hypoxia and glucose deprivation. Peptides, 2012, 33: 298–306
Navarro-Gonzalez JF, Mora-Fernandez C, Muros de Fuentes M, Garcia-Perez J. Inflammatory molecules and pathways in the pathogenesis of diabetic nephropathy. Nat Rev Nephrol, 2011, 7: 327–340
Navarro-Gonzalez JF, Mora-Fernandez C. The role of inflammatory cytokines in diabetic nephropathy. J Am Soc Nephrol, 2008, 19: 433–442
Paine SK, Sen A Fau-Choudhuri S, Choudhuri S Fau-Mondal LK, Mondal Lk Fau-Chowdhury IH, Chowdhury Ih Fau-Basu A, Basu A Fau-Mukherjee A, Mukherjee A Fau-Bhattacharya B, Bhattacharya B. Association of tumor necrosis factor alpha, interleukin 6, and interleukin 10 promoter polymorphism with proliferative diabetic retinopathy in type 2 diabetic subjects. Retina, 2012, 32: 1197–1203
Joussen AM, Doehmen S, Le ML, Koizumi K, Radetzky S, Krohne TU, Poulaki V, Semkova I, Kociok N. TNF-a mediated apoptosis plays an important role in the development of early diabetic retinopathy and long-term histopathological alterations. Mol Vis, 2009, 15: 1418
Daviaud D, Boucher J, Gesta S, Dray C, Guigne C, Quilliot D, Ayav A, Ziegler O, Carpene C, Saulnier-Blache JS. TNFa up-regulates apelin expression in human and mouse adipose tissue. FASEB J, 2006, 20: 1528–1530
Jung WK, Lee DY, Park C, Choi YH, Choi I, Park SG, Seo SK, Lee SW, Yea SS, Ahn SC. Cilostazol is anti-inflammatory in BV2 microglial cells by inactivating nuclear factor-kappaB and inhibiting mitogen-activated protein kinases. British J Pharmacol, 2010, 159: 1274–1285
Rankine E, Hughes P, Botham M, Perry V, Felton L. Brain cytokine synthesis induced by an intraparenchymal injection of LPS is reduced in MCP-1-deficient mice prior to leucocyte recruitment. Eur J Neurosci, 2006, 24: 77–86
Nawaz M, Van Raemdonck K, Mohammad G, Kangave D, Van Damme J, Abu El-Asrar A, Struyf S. Autocrine CCL2, CXCL4, CXCL9 and CXCL10 signal in retinal endothelial cells and are enhanced in diabetic retinopathy. Exp Eye Res, 2013, 109: 67–76
de Waal Malefyt R, Abrams J, Bennett B, Figdor CG, de Vries JE. Interleukin 10 (IL-10) inhibits cytokine synthesis by human monocytes: an autoregulatory role of IL-10 produced by monocytes. J Exp Med, 1991, 174: 1209–1220
Kremlev SG, Palmer C. Interleukin-10 inhibits endotoxin-induced pro-inflammatory cytokines in microglial cell cultures. J Neuroimmunol, 2005, 162: 71–80
Tang J, Kern TS. Inflammation in diabetic retinopathy. Prog Retinal Eye Res, 2011, 30: 343–358
Coorey NJ, Shen W, Chung SH, Zhu L, Gillies MC. The role of glia in retinal vascular disease. Clin Exp Optometry, 2012, 95: 266–281
Cantley LC. The phosphoinositide 3-kinase pathway. Science, 2002, 296: 1655–1657
Fruman DA, Meyers RE, Cantley LC. Phosphoinositide kinases. Ann Rev Biochem, 1998, 67: 481–507
Hirsch E. Central role for G protein-coupled phosphoinositide 3-kinase gamma in Inflammation. Science, 2000, 287: 1049–1053
Johnson GL, Lapadat R. Mitogen-activated protein kinase pathways mediated by ERK, JNK, and p38 protein kinases. Science, 2002, 298: 1911–1912
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at link.springer.com
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Chen, L., Tao, Y. & Jiang, Y. Apelin activates the expression of inflammatory cytokines in microglial BV2 cells via PI-3K/Akt and MEK/Erk pathways. Sci. China Life Sci. 58, 531–540 (2015). https://doi.org/10.1007/s11427-015-4861-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-015-4861-0